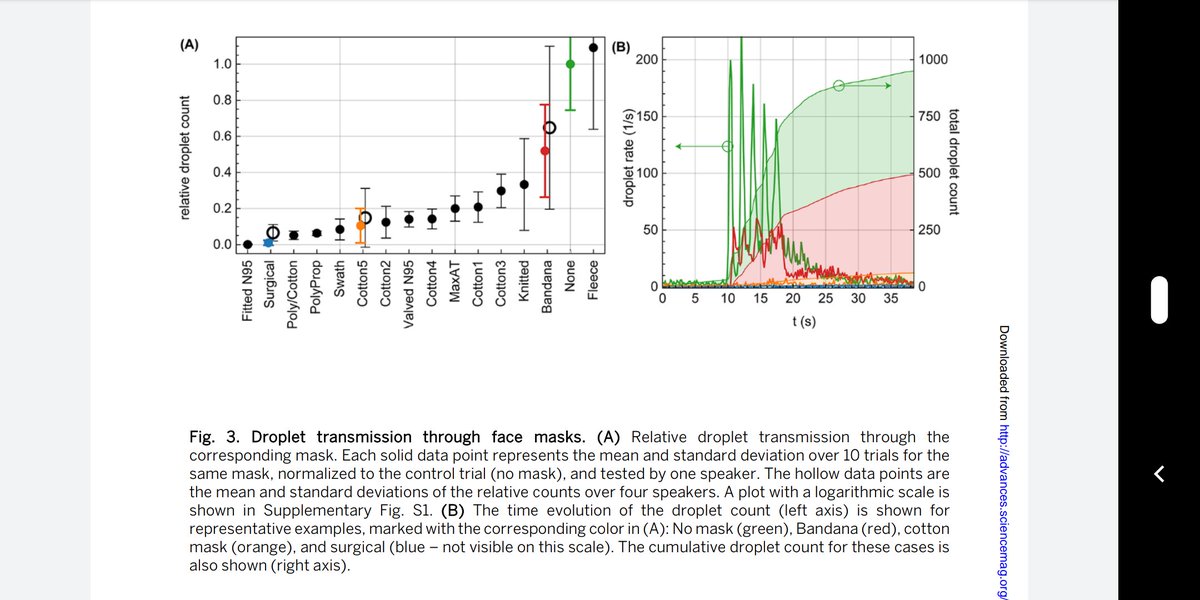
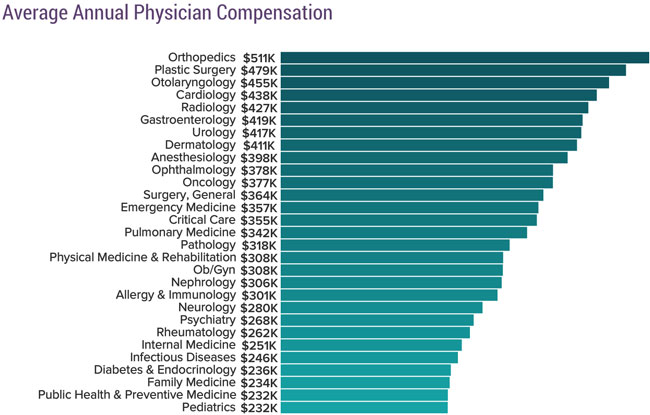
Closing the barn door 6 months after the horse left the barn, and 3 months after she moved to a different barn!
Trump administration bars FDA from regulating some laboratory tests, including for coronavirus
Let's review-
washingtonpost.com/health/2020/08…
ON THE SECOND DAY OF FEBRUARY
We wasted WEEKS in a critical phase of the outbreak, overly reliant on the flawed CDC tests, but by March the EUAs were rolling out finally
the department has determined that the Food and Drug Administration ("FDA") will not require premarket review of laboratory developed tests ("LDT") absent notice-and-comment rulemaking

@scottjbecker CEO of Public Health Lab Assoc (@APHL) nails it. “I’m not certain why this reversal by HHS was done now, given the oversight process seemed to be working”
Bizarre
*Expertise Matters (listen to people like @ScottGottliebMD)
*Regulatory oversight sucks, lack of regulatory oversight sucks more
*One day we need to re-architect the patchwork oversight of labs (and food, and animal disease)- but thoughtfully, not like this