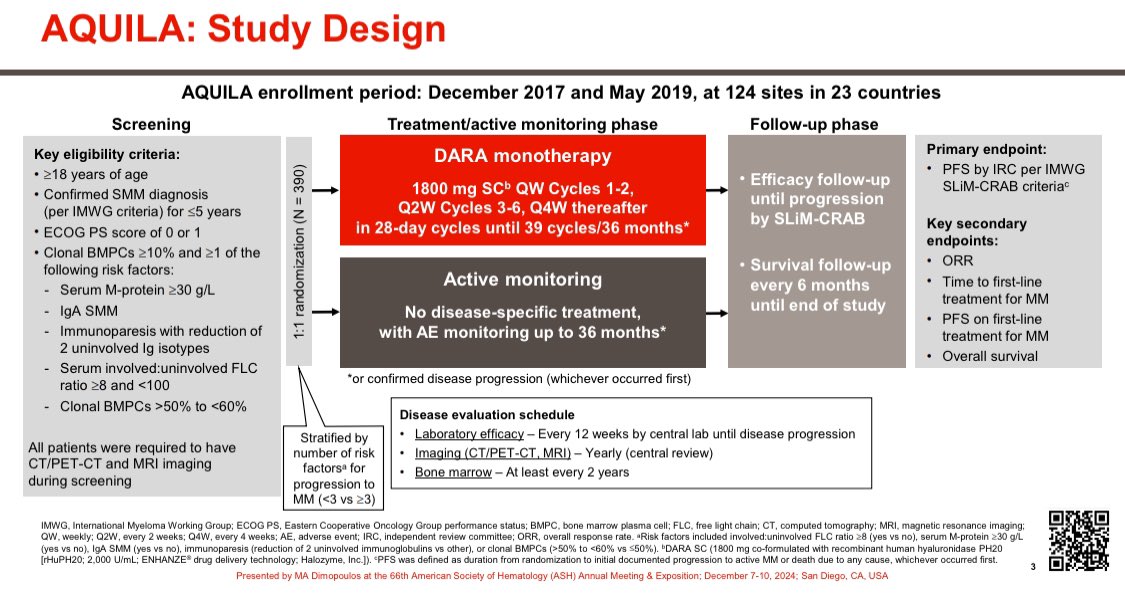
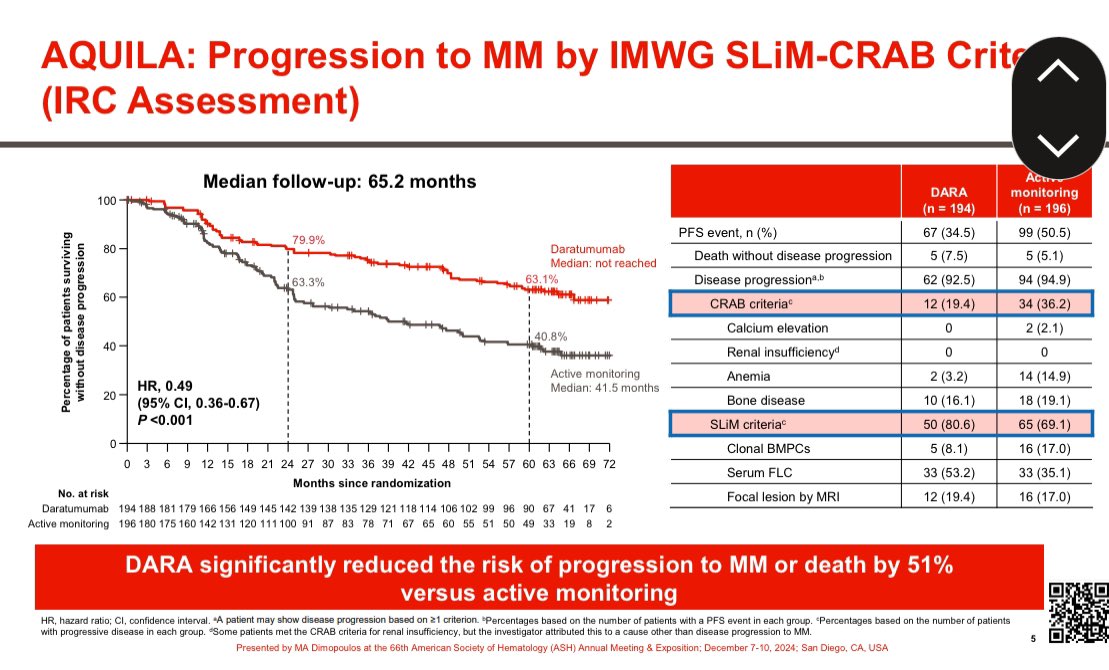
Our study shows that approximately 10% of patients undergoing auto transplant for myeloma have an exceptional response (PFS > 8 years without maintenance therapy). Median survival ~20 years; 25% never achieved CR. #BCJ @MayoCancerCare @MorieGertz @Rfonsi1 nature.com/articles/s4140…
This doesn’t mean we don’t recommend maintenance. We do for all patients. But an attempt to determine the baseline proportion of patients who achieve extraordinary outcome with just induction and transplant. It’s hard to predict who they will be. Almost all had standard risk MM.
It also shows the incredible value of high dose melphalan in myeloma therapy.
The first author, Dr. Ashley Paquin was a medical student @MayoClinicSOM when she did this project. @VisramAlissa greatly helped complete the project.
• • •
Missing some Tweet in this thread? You can try to
force a refresh