This issue is, appropriately, contentious. As a vaccinologist - & citizen & relative of people in at-risk groups - I fully support the UK decision to increase dose intervals of both our Ox/AZ product and the Pfizer product. I'd happily receive either with a >8w gap. Here's why 🧵
https://twitter.com/drmarkporter/status/1344926628393181184
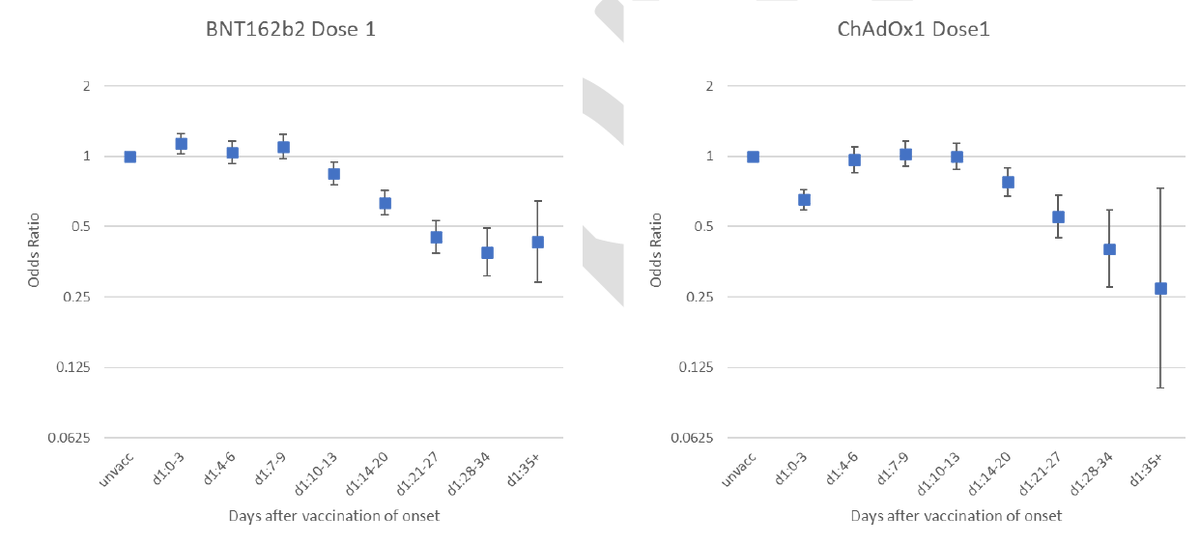
For the Ox/AZ vaccine, it's fairly simple. The trial demonstrated efficacy at a range of dose intervals. Antibody responses after the boost were significantly stronger with longer intervals - see table 3.
gov.uk/government/pub…
gov.uk/government/pub…
(so in response to @drmarkporter's point, higher immune responses with a longer interval is proven & now public. I haven't seen a similar analysis for efficacy against disease but the data exists and I suspect the regulators & JCVI committee have)
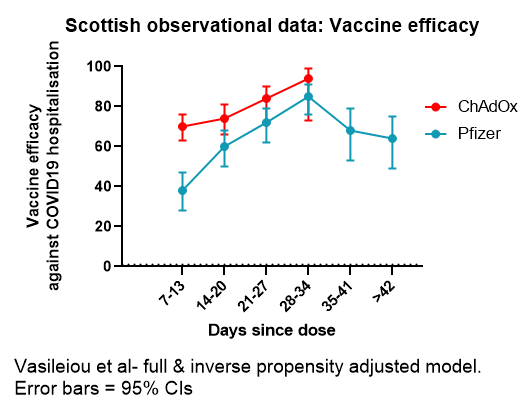
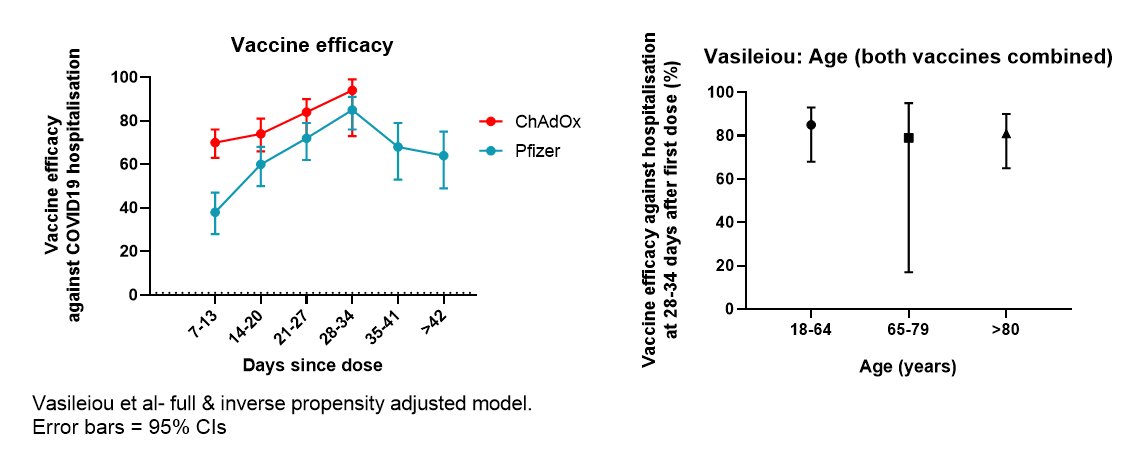
For Pfizer, there isn't direct evidence of efficacy with a >3wk interval. But as widely publicised, efficacy in the period from 14 days after first dose to 21 days is high. 
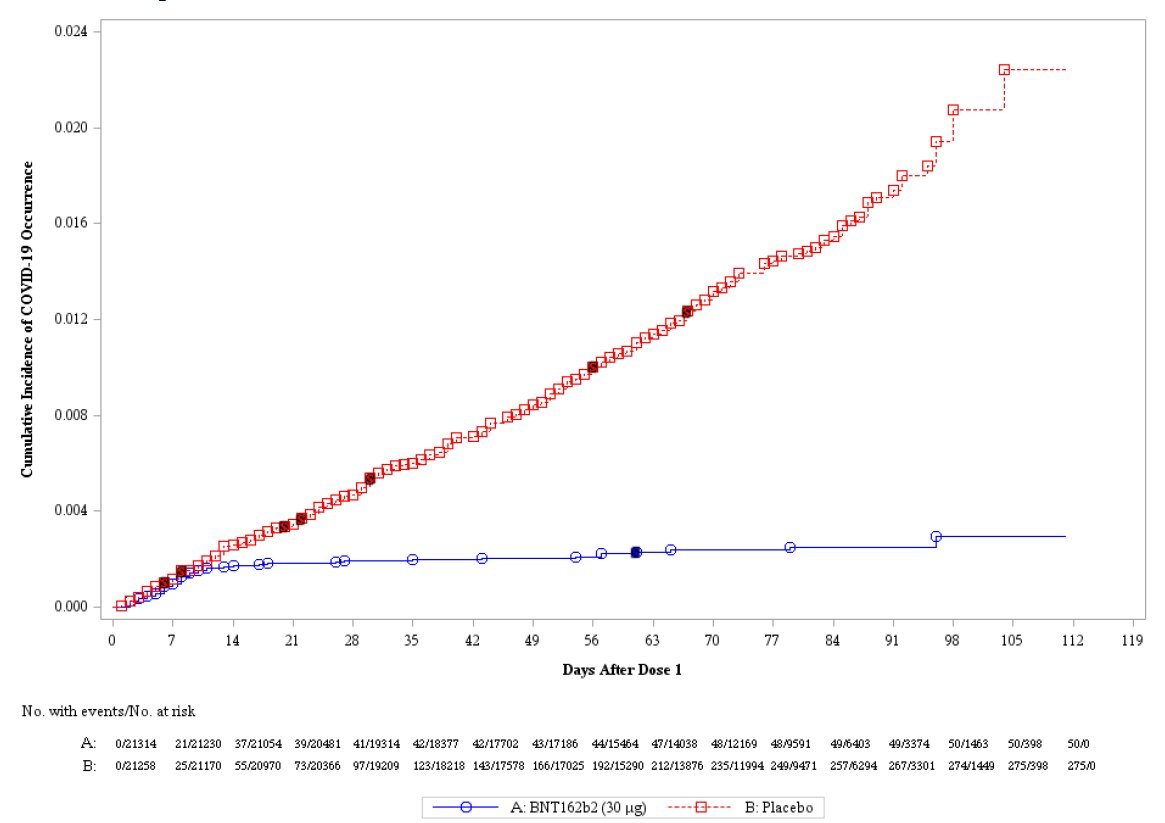
Can we extrapolate from this to a longer interval? It's a judgment call. On one hand is evidence-based medicine's scepticism of anything not directly proven 'beyond reasonable doubt' in an RCT; on the other is a 'balance of probabilities' approach based upon the biology.
Based upon the biology, I'd eat my hat if the Pfizer vaccine is substantially less effective with a longer dose interval. Most vaccines induce stronger immune responses with longer intervals. A couple of examples below. There are more. 
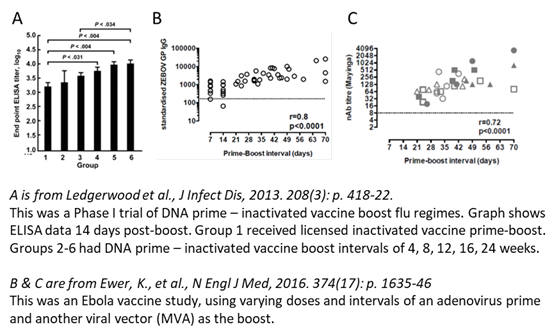
Regimes like the Ox/AZ use the same adenoviral vector to prime & boost so face 'anti-vector immunity' (immunity from the first dose to the viral 'postman' which must delivers the spike protein 'message' for the boost). This favours longer intervals specifically for Ad/Ad but...
...the above shows that longer intervals are better for regimes with different viral vectors, DNA priming, inactivated virus boosting - this isn't just an adeno effect. It's v rare for a 3wk interval to give stronger responses than 8+ wks (I can't think of examples, can you?)
Mechanistically, at 3w, the immune response to prime isn't complete- it hasn't yet produced all the memory B cells which give the best response to the boost. Once they are made, the memory cells last years! They won't forget how to respond to a boost in a few months.
I appreciate mechanistic arguments often prove to be wrong, and RCT evidence with Pfizer at longer intervals should definitely be produced ASAP... but as @zeynep has written in an excellent article today:
theatlantic.com/science/archiv…
theatlantic.com/science/archiv…

There is a good debate:
@Bob_Wachter has written thoughtfully
@trishgreenhalgh @EricTopol @nataliexdean have all argued the other way from me - would welcome their thoughts on the above 🧵
@Bob_Wachter has written thoughtfully
https://twitter.com/Bob_Wachter/status/1344667655324585986
@trishgreenhalgh @EricTopol @nataliexdean have all argued the other way from me - would welcome their thoughts on the above 🧵
I haven't yet seen a vaccine immunologist who has personally done experiments giving vaccines at different intervals and who is concerned about the longer interval... but I look forward to hearing that view too...
• • •
Missing some Tweet in this thread? You can try to
force a refresh