Doc 1: There’s a big swing on their A-line, shall I give more fluids?
Doc 2: Well given that they’re spontaneously breathing, there’s no real evidence to support PPV here 🤷♂️
- A recently overheard conversation, prompting this 🧵 on PPV in the spontaneously breathing patient.
Doc 2: Well given that they’re spontaneously breathing, there’s no real evidence to support PPV here 🤷♂️
- A recently overheard conversation, prompting this 🧵 on PPV in the spontaneously breathing patient.
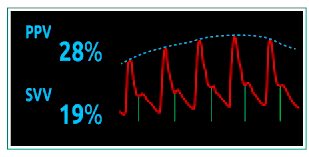
The ∆ between systolic & diastolic pressures is the pulse pressure (PP) & it is determined by the compliance of the aorta and the ventricular stroke volume (SV).
Whilst aortic compliance reduces with age, beat-to-beat changes in PP occur predominately due to changes in SV.
Whilst aortic compliance reduces with age, beat-to-beat changes in PP occur predominately due to changes in SV.

When asking if a pt is fluid responsive (FR), we’re asking if ⬆️ preload will ⬆️ SV. Observing a ∆PP with a ∆preload can help us try to answer this.
Pulse pressure variation (PPV) is the diff. between the max/min PP (as a % of the mean), occurring over a respiratory cycle.
Pulse pressure variation (PPV) is the diff. between the max/min PP (as a % of the mean), occurring over a respiratory cycle.

Ventilation requires changes in intra-thoracic pressure (ITP), and this ∆ITP, in turn affects preload.
PPV allows us to see if these ventilation-induced changes lead to a change in SV, hinting at where pts lie on their Starling curve, and if they are likely to be FR.
PPV allows us to see if these ventilation-induced changes lead to a change in SV, hinting at where pts lie on their Starling curve, and if they are likely to be FR.

Spontaneous ventilation (SpV) causes a -ve ITP & tends to ⬆️ preload; mechanical ventilation (MV) causes a +ve ITP & tends to ⬇️preload.
In each case, the size of the ∆preload, relates to the size of the ∆ITP.
This needs to be considered when interpreting PPV in practice.
In each case, the size of the ∆preload, relates to the size of the ∆ITP.
This needs to be considered when interpreting PPV in practice.

During MV, a PPV >12% has been shown to accurately predict FR (sens 88%; spec 89%) as long as:
🔹Vt >8ml/kg
🔹Sinus rhythm (SR)
🔹No spontaneous breathing
Understanding the physiology behind PPV, we can see why these preconditions must be met.
ccforum.biomedcentral.com/articles/10.11…
🔹Vt >8ml/kg
🔹Sinus rhythm (SR)
🔹No spontaneous breathing
Understanding the physiology behind PPV, we can see why these preconditions must be met.
ccforum.biomedcentral.com/articles/10.11…
🔹Spontaneous breathing alters the ∆ITP unpredictably making interpreting PPV 😵💫
🔹In arrhythmias (i.e. AF) preload & thus PP can vary significantly beat-to-beat leading to false +ve’s
🔹Smaller Vt ➡️ Smaller ∆ITP leading to false -ve’s
🔹In arrhythmias (i.e. AF) preload & thus PP can vary significantly beat-to-beat leading to false +ve’s
🔹Smaller Vt ➡️ Smaller ∆ITP leading to false -ve’s

Another important ⛔️ is pts with ⬇️ chest wall compliance (i.e. IAH). Here, the same Vt ➡️ a larger ∆ITP, causing a false +ve PPV.
Unrecognised this can lead to IV boluses, which can worsen CW compliance, exaggerating the PPV, leading to a 🌀 of crystalloid water-boarding 😨
Unrecognised this can lead to IV boluses, which can worsen CW compliance, exaggerating the PPV, leading to a 🌀 of crystalloid water-boarding 😨

PPV has also been studied in SpV where it’s been shown to have a diminished sensitivity but good specificity (see table from linked systematic review)
Sens will ⬆️ with forced insp efforts, or when there is increased WOB with larger -∆ITP
ncbi.nlm.nih.gov/pmc/articles/P…
Sens will ⬆️ with forced insp efforts, or when there is increased WOB with larger -∆ITP
ncbi.nlm.nih.gov/pmc/articles/P…

Two of the mechanisms for ⬇️ sensitivity are:
1) In health the resp system is very compliant, so tidal breathing may cause inadequate ∆ITP
2) In hypovolaemic states, a ⬇️ITP may not ⬆️preload due to collapse of the great veins (maximal venous return is already achieved)
1) In health the resp system is very compliant, so tidal breathing may cause inadequate ∆ITP
2) In hypovolaemic states, a ⬇️ITP may not ⬆️preload due to collapse of the great veins (maximal venous return is already achieved)
A final caveat… for both MV & SpV: in the presence of RV/LV impairment, ventilation can significantly affect SV irrespective of preload conditions, leading to high PPV/false +ve’s
Specificity is thus significantly reduced and PPV may be of limited value 😕
Specificity is thus significantly reduced and PPV may be of limited value 😕
So back to the original question… in the absence of RV/LV dysfunction, a high PPV in a SpV pt means it’s highly likely they’ll be FR.
Of course the likes of @msiuba @icmteaching & @ThinkingCC won’t forgive me if I don’t mention that being FR doesn’t necessarily = needs fluid!
Of course the likes of @msiuba @icmteaching & @ThinkingCC won’t forgive me if I don’t mention that being FR doesn’t necessarily = needs fluid!
Hope this is of use, & may even help people avoid pitfalls I know I’ve fallen for!
For far more detailed explanations please read @PrXaMonnet review article:
annalsofintensivecare.springeropen.com/articles/10.11…
& @heart_lung ‘s book (everything worth knowing about Heart-Lung interactions I learnt here!)
For far more detailed explanations please read @PrXaMonnet review article:
annalsofintensivecare.springeropen.com/articles/10.11…
& @heart_lung ‘s book (everything worth knowing about Heart-Lung interactions I learnt here!)
• • •
Missing some Tweet in this thread? You can try to
force a refresh








