The high cost of prescription drugs and what we need to do about it. Why nothing ever gets done.
A full 360 of the issue using cancer drugs and insulin as examples. Check it out.
1/
A full 360 of the issue using cancer drugs and insulin as examples. Check it out.
1/
2/ I'm not an enemy of Pharma. I understand the value of innovation first hand. I've led numerous clinical trials, and continue to work with Pharma on new drugs.
My interest started with the recognition that every drug in my field is priced sky high. Unsustainable.👇
My interest started with the recognition that every drug in my field is priced sky high. Unsustainable.👇

3/ Prescription drug prices are generally higher in the US than in every other country. Simple drugs. Complex drugs. New drugs. Old drugs. Everything. 

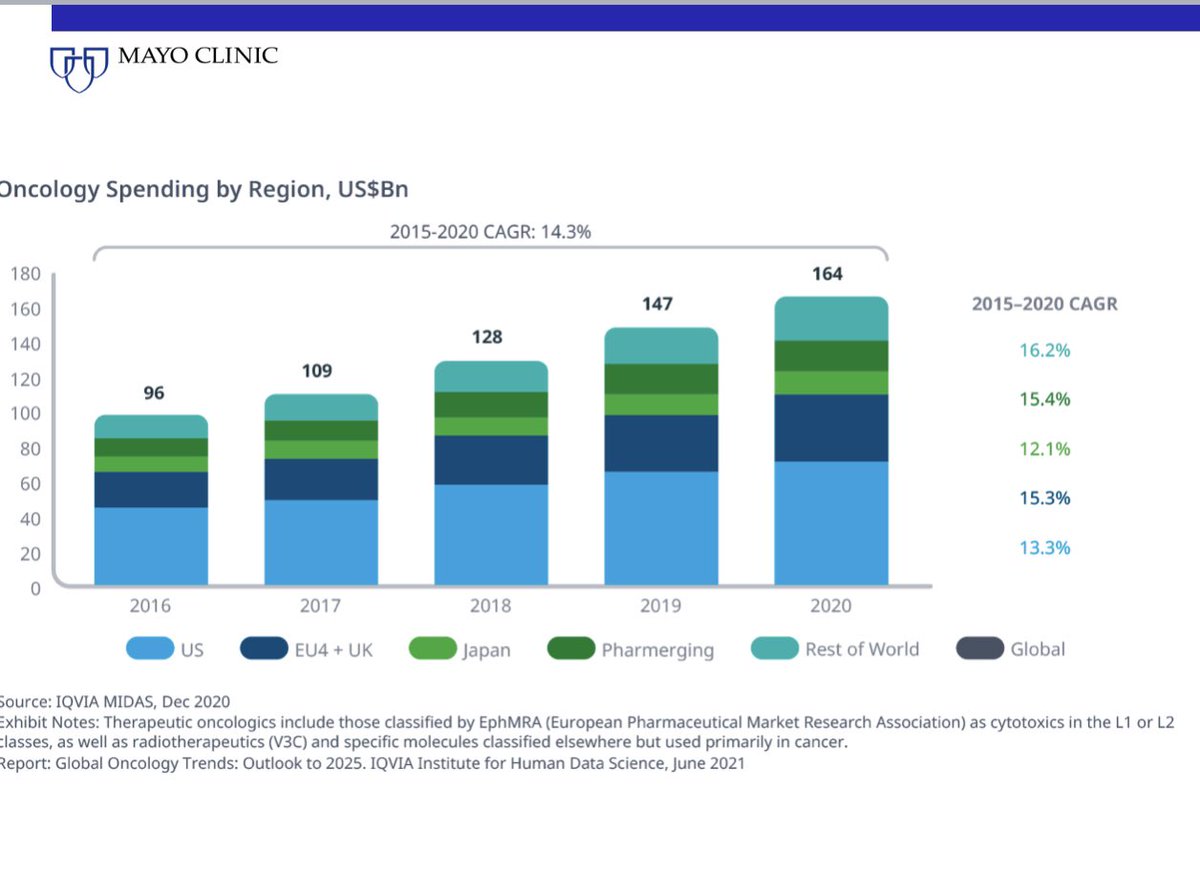
4/ We spend almost the same on prescription drugs for drugs to treat serious illnesses like cancer as all other countries combined. 
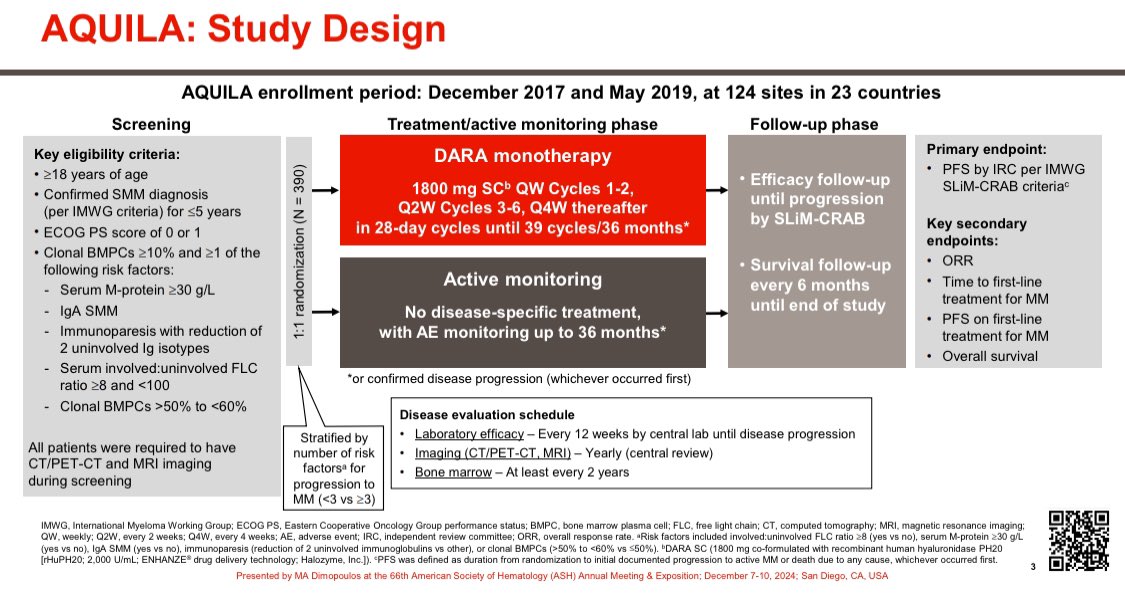
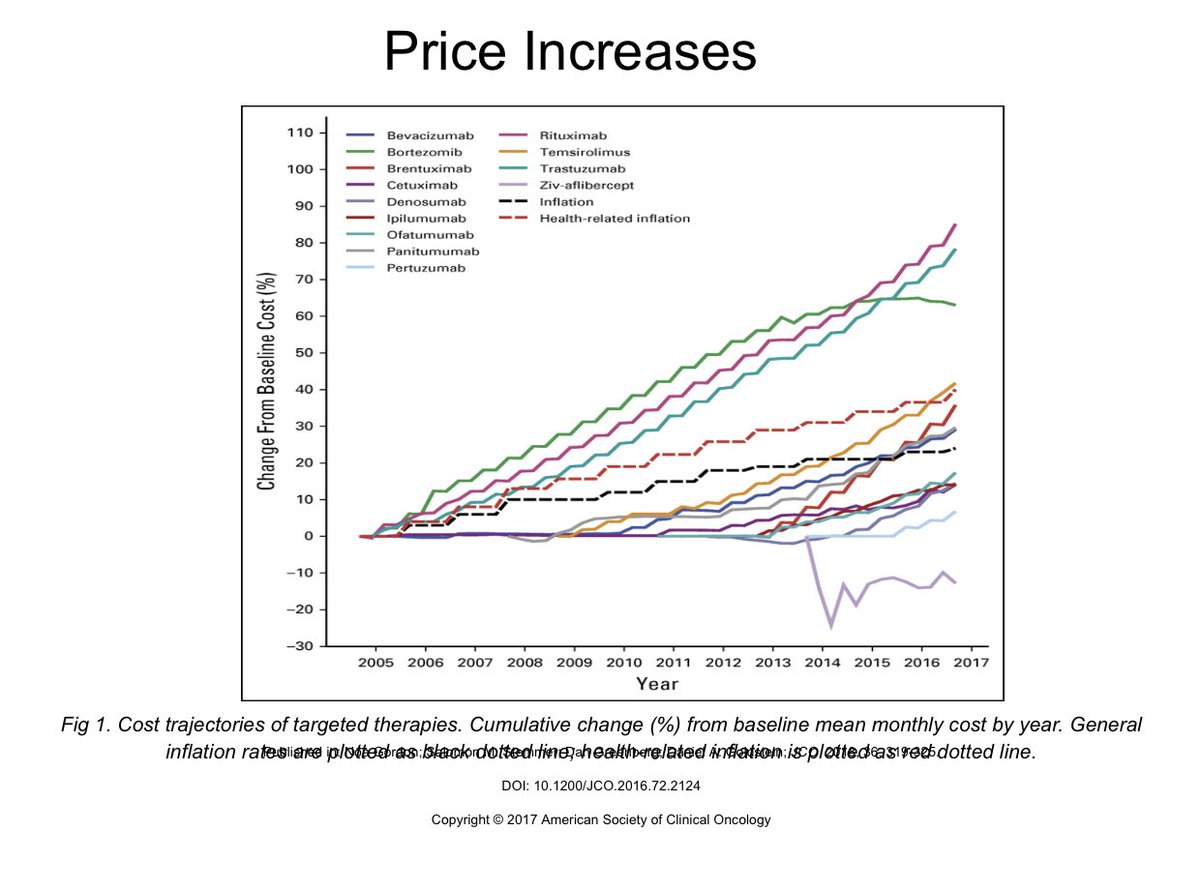
5/ Another unique craziness in the US is that not only is the launch price high, but prices of new drugs increase each year for no reason. Way more than inflation. So while prices go down in other countries, we pay more. 
6/ Why are prescription drug prices so high and so out of control? Many reasons which I list below.
But #1 is that prescription drugs are not luxury items that we can live without.
👇👇
But #1 is that prescription drugs are not luxury items that we can live without.
👇👇

7/ Second it's a MONOPOLY. The seller can dictate the price. When the monopoly is on a life saving drug, they really have you.
Even when there are many drugs to treat the same disease, because each drug may work only for a while, especially in cancer, each drug is a monopoly.
Even when there are many drugs to treat the same disease, because each drug may work only for a while, especially in cancer, each drug is a monopoly.

8/ Third, they have perfected the art of making patents last on and on and on. From filing newer patents on same drug to frivolous lawsuits to making slightly better versions of their drug and then filing new patents on them. 

9/ Fourth, just like in tech, even when the current drug works well, it will be made to look obsolete by marketing. So the promise of low prices as drugs go generic may not be realized coz by then the drug is made to look old fashioned and obsolete. 

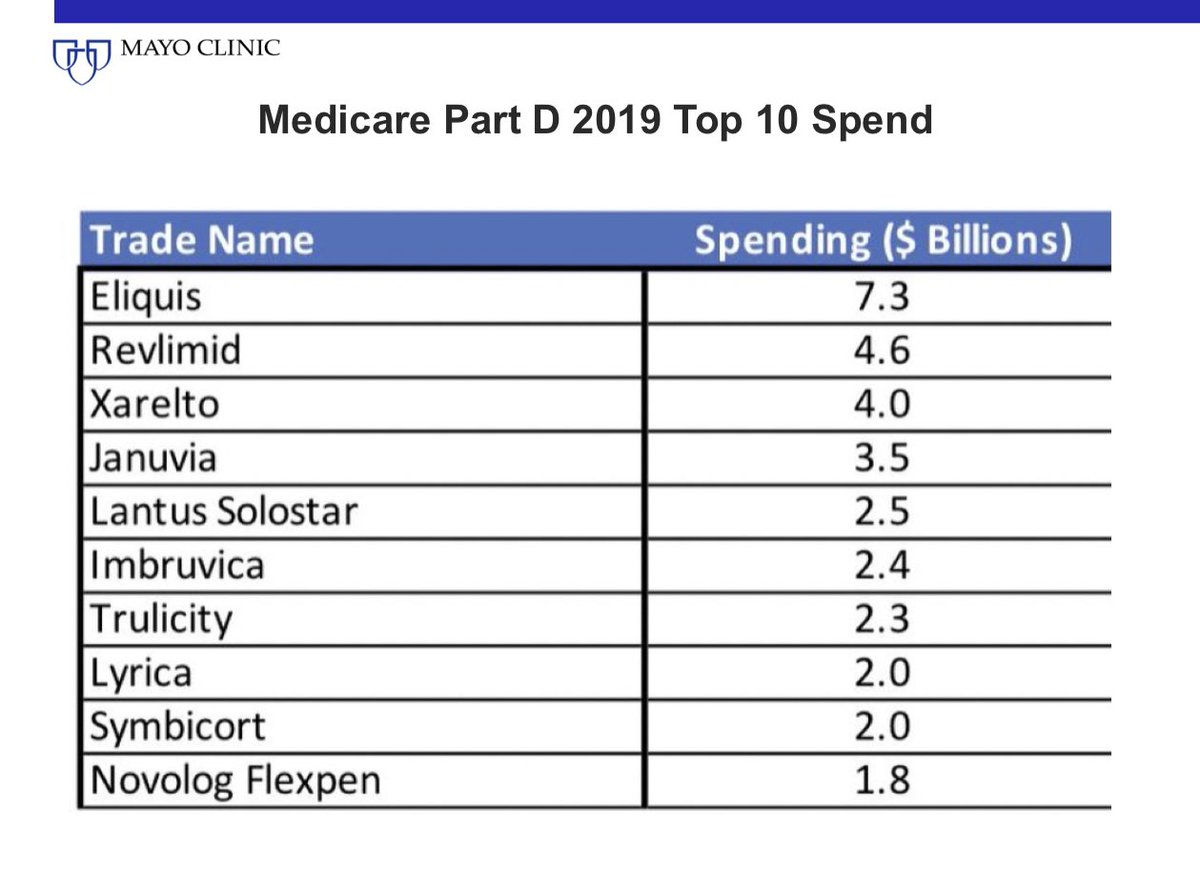
10/ No wonder 2 of the top 3 drugs Medicare spends the most money on are new oral blood thinners. 4 of top 10 are diabetes meds.
These are better than older ones but don't fall for the prices will come down with time trick. They may, but not for the drug you are prescribed.
These are better than older ones but don't fall for the prices will come down with time trick. They may, but not for the drug you are prescribed.
11/ Fifth, we have many regulatory and legal barriers for generics & biosimilars to enter. Even if a generic enters, getting market share is not easy in our system where contracts may go to who can offer the best profit margin than to who has the lowest price product. 

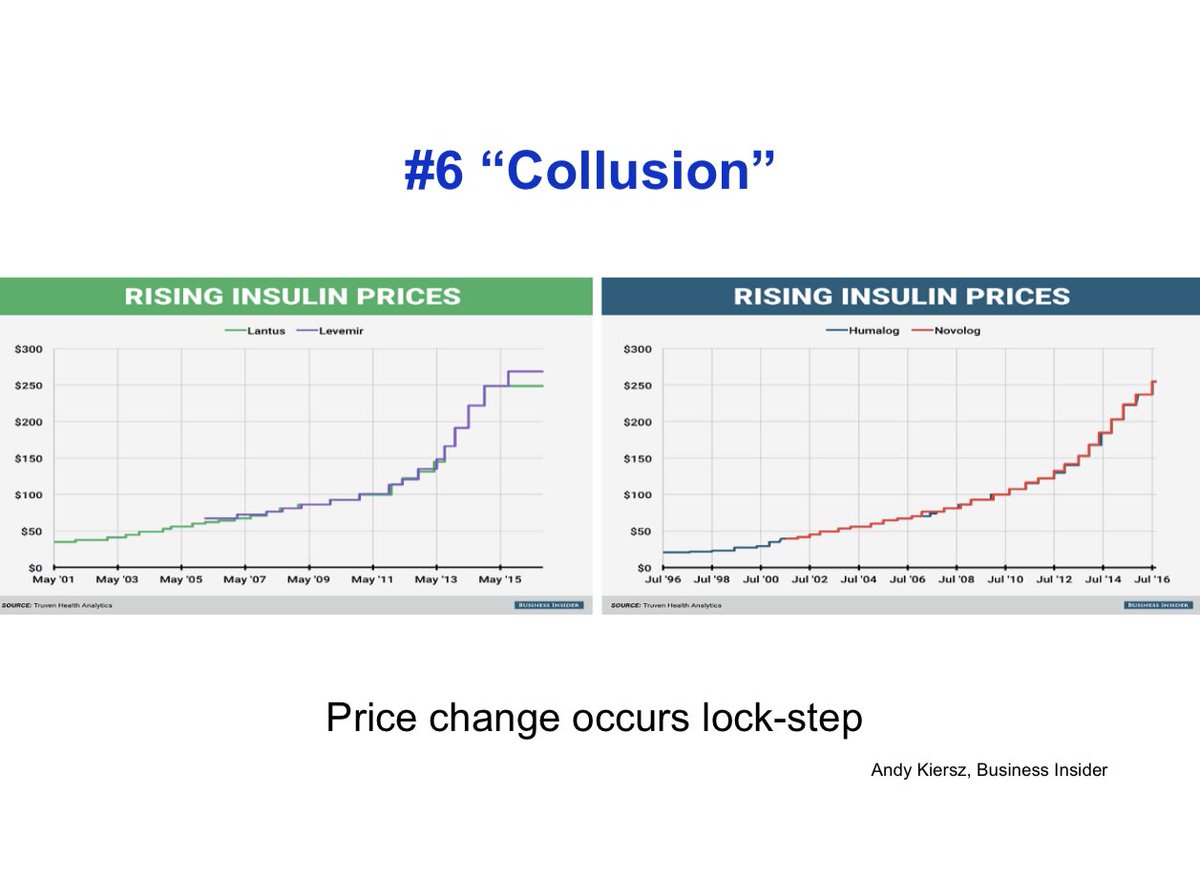
12/ Sixth is the wink wink understanding among drug manufacturers to promptly increase price when a competitor increases their price. Collusion without collusion. See below with insulin. An astonishing lockstep price increase over decades. 
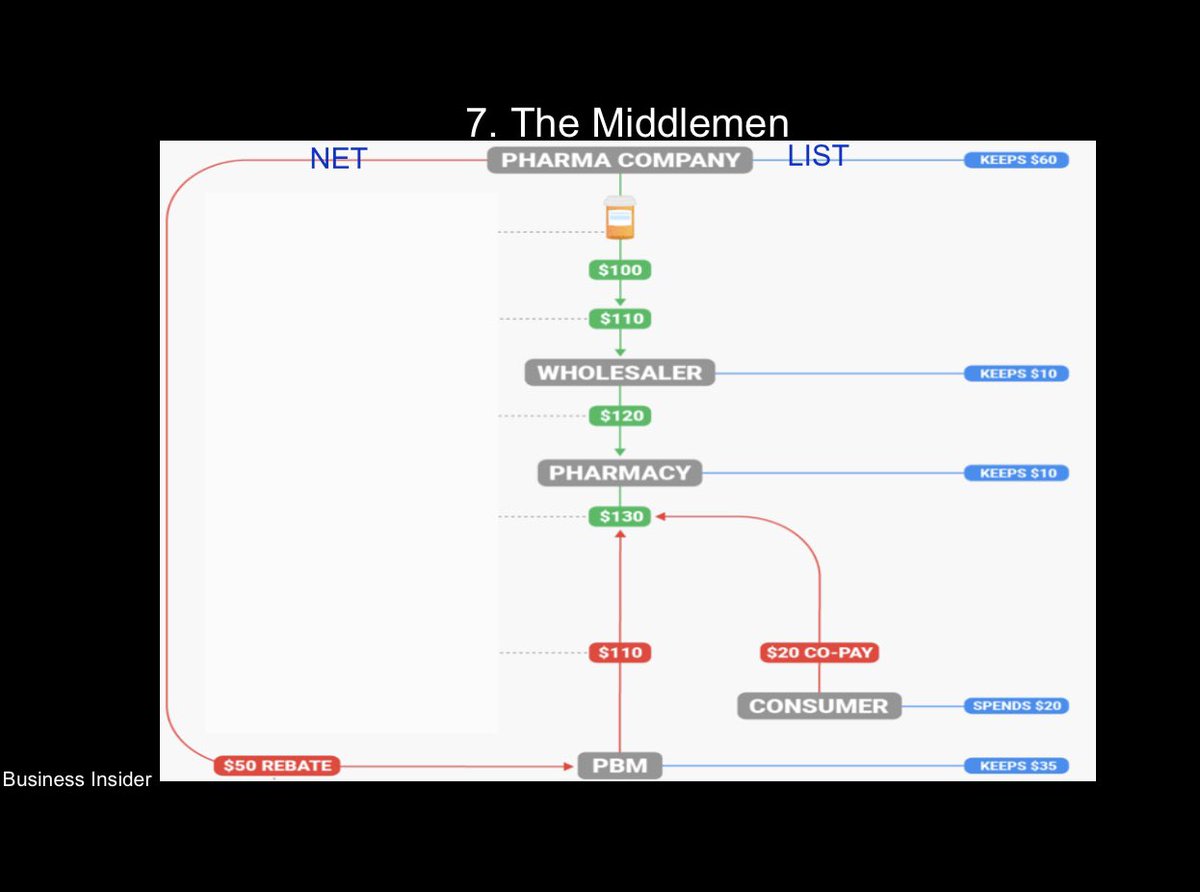
13/ Seventh, the middlemen. Everyone in the chain benefits from a high list price (profit as a % of list price) except the patient.
Leads to ever rising drug prices.
Leads to ever rising drug prices.
14/ Eighth, Pharma lobbying power and money is hard to overcome. So despite overwhelming public support to change things, NOTHING gets done. 

16/ Finally there are few allies in the fight to lower drug prices. Very few speak up for measures that truly reduce list price. They often support Pharma friendly policy measures or stay silent. 

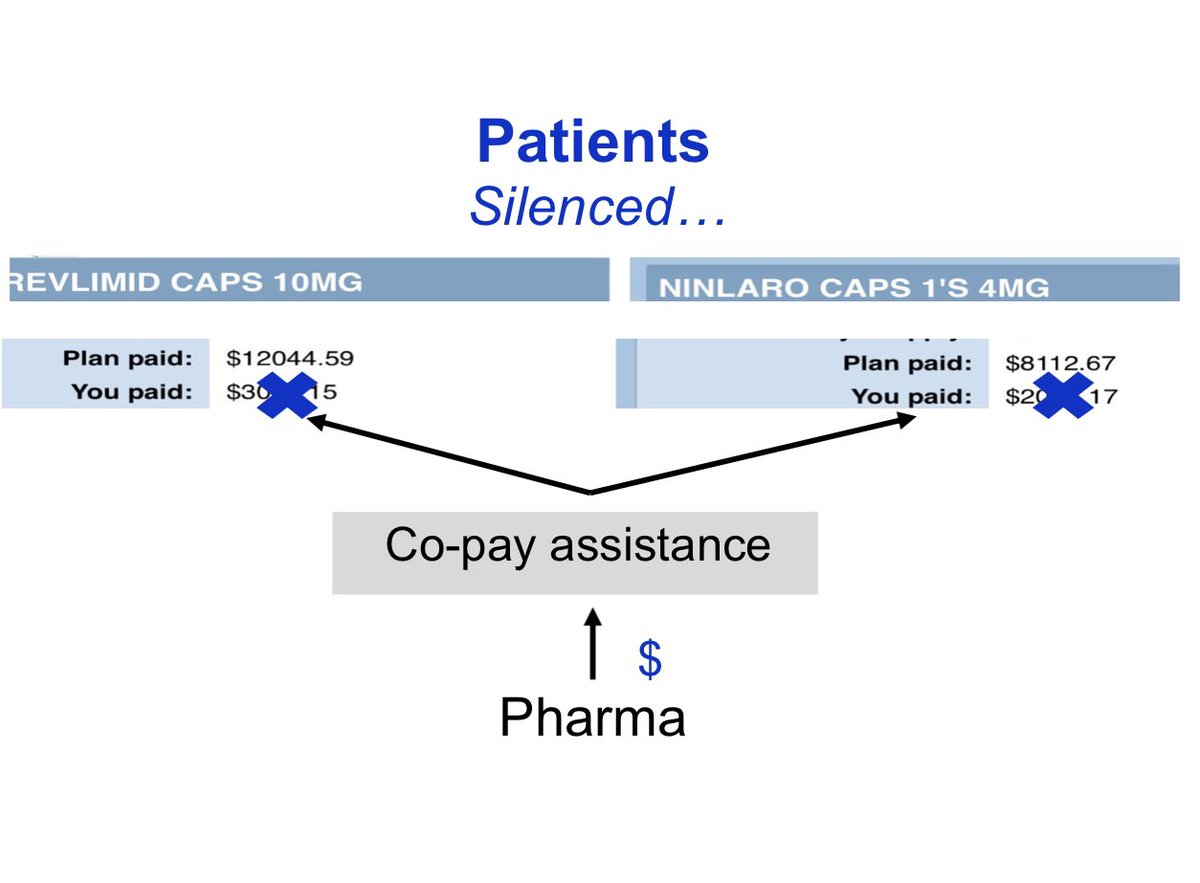
17/ By providing copay assistance through Pharma funded copay relief organizations, the patient voice is silenced. Pharma channels a few thousand to get tens of thousands back, and can now keep increasing prices without patients protesting. @NorthTxMSG 
18/ So what can we do? What should we do.
First, we must have an organization that sets a value based price for all new drugs that derives legitimacy and is endorsed by Medicare and the federal government. Similar to other developed countries.
First, we must have an organization that sets a value based price for all new drugs that derives legitimacy and is endorsed by Medicare and the federal government. Similar to other developed countries.

19/ Second, Medicare must be authorized to negotiate for prescription drugs. It's foolish to mandate Medicare to cover prescription drug costs and simultaneously order them to pay up whatever Pharma wants without negotiation. 

20/ More than 80-90% of the public supports Medicare negotiation. Other developed countries negotiate prices. The fact that we don't negotiate affects the ability of other countries to negotiate prices because Pharma can walk away and make up loss by increasing prices in the US. 

21/ See below how the price of the same drug can be increased year after year in the US. From insulin to Revlimid. 
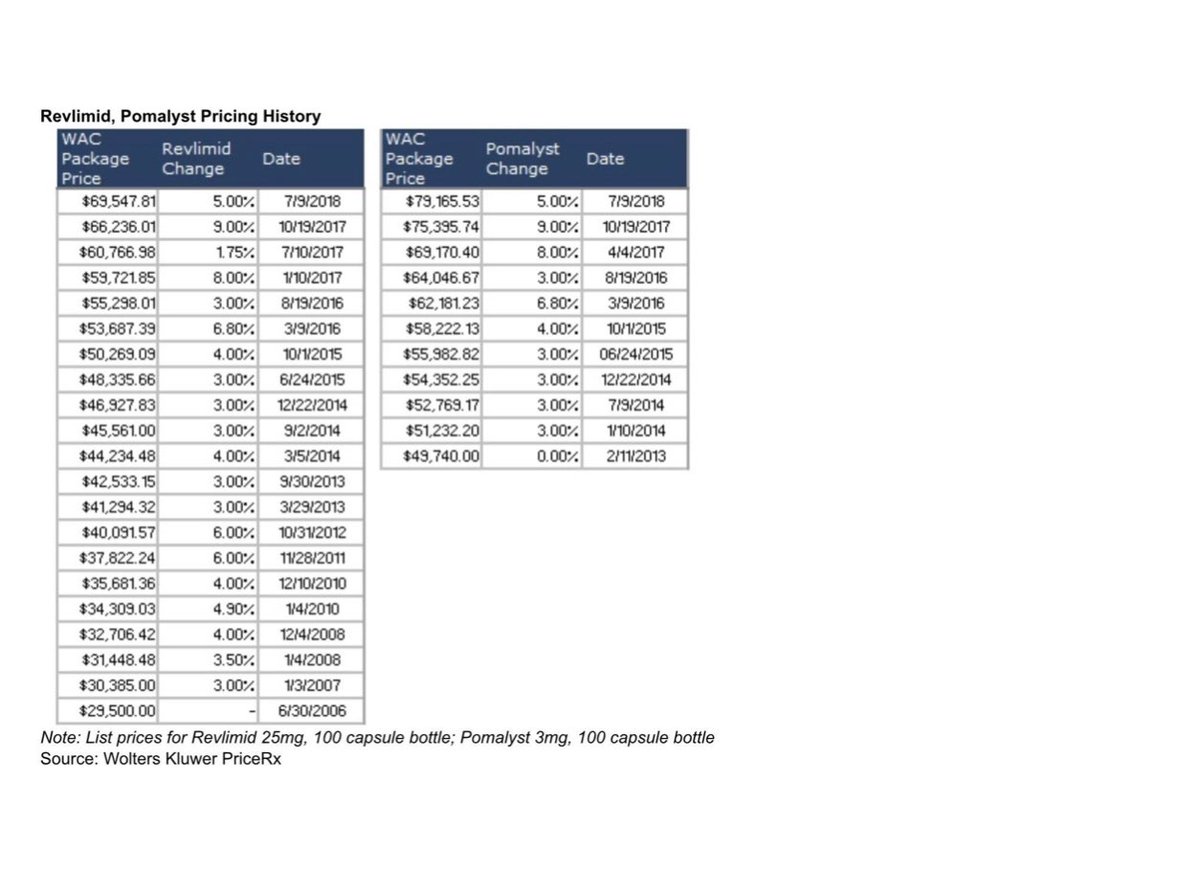
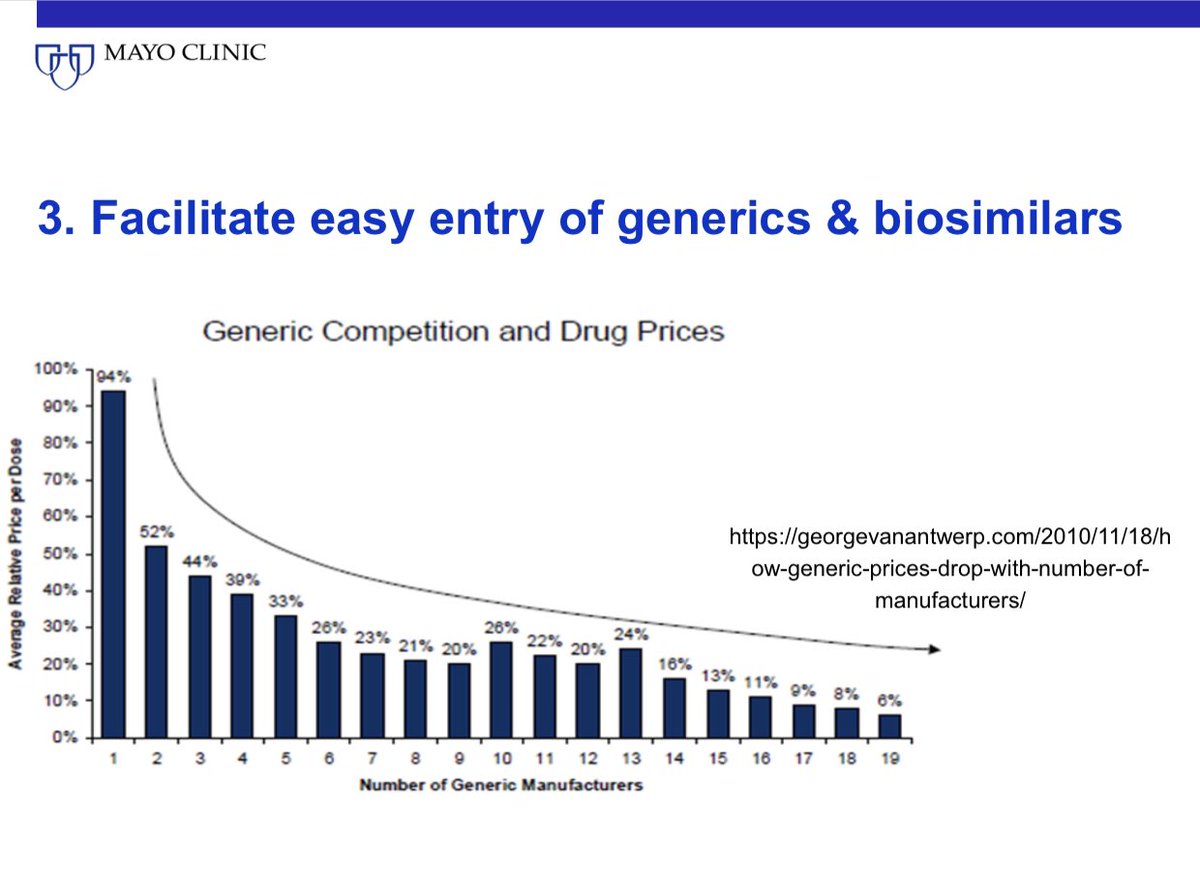
22/ Third, we need to make it far easier and competitive for generic and biosimilar manufacturers to enter the US market. At least 4 generics for each brand name drug is needed to have an effect on prices. 
25/ Fourth we need to reduce the cost of clinical trials. This puristic drive for 100% monitoring and perfection makes our trials extraordinarily expensive. Revise FDA guidelines. 

26/ Fifth, we need serious patent reform. 7-10 years and done. Or similar. @pritikrishtel @IMAKglobal 

28/ First, we must have strong National treatment guidelines and pathways that take cost into account. Guidelines that recommend two dramatically differently priced drugs as equivalent do not help. 

29/ So the 2 drugs below are not similar just because the trial found them similar. They are not similar in price. Since our system rewards the higher priced drug with more reimbursement it is an invitation to prescribe the higher price drug if we say both are reasonable options. 

30/ We must conduct strategic trials that reduce cost. Testing fixed duration therapy. Clever dosing schedules. Etc. 

31/ Always support the lower lost price option even if your profit margin is higher with the brand name drug due to rebates or other deals. Always support generics and biosimilars. 

32/ Always learn the cost of drugs. Learn how to help your patient get lowest price drugs. @costplusdrugs @GoodRx 

33/ Advocacy is important. There are few organizations seriously trying to lower drug costs. Find a way to engage and support. @AARP @policydeb @DavidP4AD @P4AD_ @IMAKglobal
34/ Talk to your patients about access and affordability.
35/ For a talk in which I discuss most of this, check out.
• • •
Missing some Tweet in this thread? You can try to
force a refresh