Live tweeting the VRBPAC meeting in this thread. See here for materials and meeting live:
fda.gov/advisory-commi…
Also I have an errand to run during the break/public comment period so I can't cover that.
fda.gov/advisory-commi…
Also I have an errand to run during the break/public comment period so I can't cover that.
Dr. Arnold Monto will not be able to chair today's meeting due to unforeseen circumstances; Dr. Stanley Perlman will chair it instead:
medicine.uiowa.edu/microbiology/p…
medicine.uiowa.edu/microbiology/p…
They are currently taking roll; this is the roster for today's meeting:
fda.gov/media/164701/d…
and this is the draft agenda:
fda.gov/media/164698/d…
As a reminder, Dr. Monto is not present.
fda.gov/media/164701/d…
and this is the draft agenda:
fda.gov/media/164698/d…
As a reminder, Dr. Monto is not present.
Peter Marks, Director of CBER is beginning with a moment of remembrance of Dr. Ovetta Fuller, who died in November 2022 and was a voting member of VRBPAC through all prior meetings.
medicine.umich.edu/dept/microbiol…
I appreciated her contributions.
medicine.umich.edu/dept/microbiol…
I appreciated her contributions.
Dr. David Kaslow is the newly appointed Director for CBER's Office of Vaccine Research and Review from PATH (path.org) where he was chief scientific officer and he will run future VRBPAC meetings for the FDA.
We are now in a place where, despite the successes of the vaccines, we can reflect on their merits and deficiencies of our vaccination programme and see if we can simplify things to make the programme work better basically.
Here is a summary of the current status of COVID-19 vaccination. Having a different booster and primary series composition is cumbersome, and we have a model for influenza that works well for adjusting for risk and updates to new emergent strains. 

Once more, the major meeting objectives. In essence, it probably makes sense to offer risk-based recommendations. There is the possibility of convening ad hoc VRBPAC meetings for variants that merit adjustment to vaccination strategies. 

Presumably, at this point in the pandemic, most of the US has been exposed to spike protein such that a single dose of vaccine would be adequate to induce or restore protection for most people. Some age-based data will be presented to discuss this as well as risk-based adjustment
(while we say presumably, seroprevalence of nucleocapsid in the US is extremely high, so it isn't really much of a presumption)
Questions from the committee now.
Meissner: Is it reasonable to make the decision in June in regard to composition of the vaccine for the following Fall? That's much less time than an influenza vaccine, selected in February.
Meissner: Is it reasonable to make the decision in June in regard to composition of the vaccine for the following Fall? That's much less time than an influenza vaccine, selected in February.
Kaslow: More details will be given from manufacturers regarding feasibility; we should return to this after Dr. Weir's presentation re what it takes to get there.
Levy: Example provided in the briefing documents of using bivalent vaccines as annual booster- is today's discussion from FDA open to other options or are they primarily considering this possibility?
Kaslow: this is framed as a discussion topic, rather than a voting topic and is quite interested in input from the advisory committee on this point
Reingold: When would it be optimal to administer a booster- September? October? November? Depends on how long we think protection lasts, when most infections and illnesses occurring.
Dr. Heather Scobie will now present for CDC's CDR regarding the current epidemiology of COVID-19 and variants.
fda.gov/media/164814/d…
fda.gov/media/164814/d…
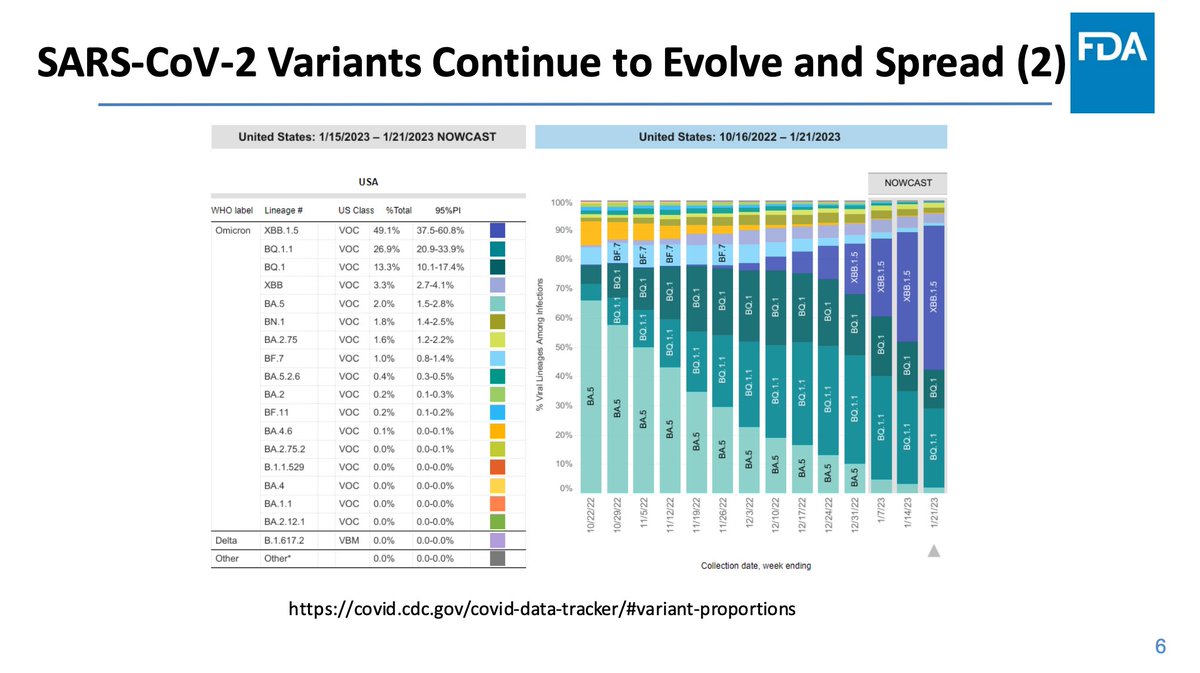
Here is the Nowcast for current variant composition in the US. Omicron isolates have accounted for >99% of sequences for many months now. XBB.1.5 is dominant and growing. 
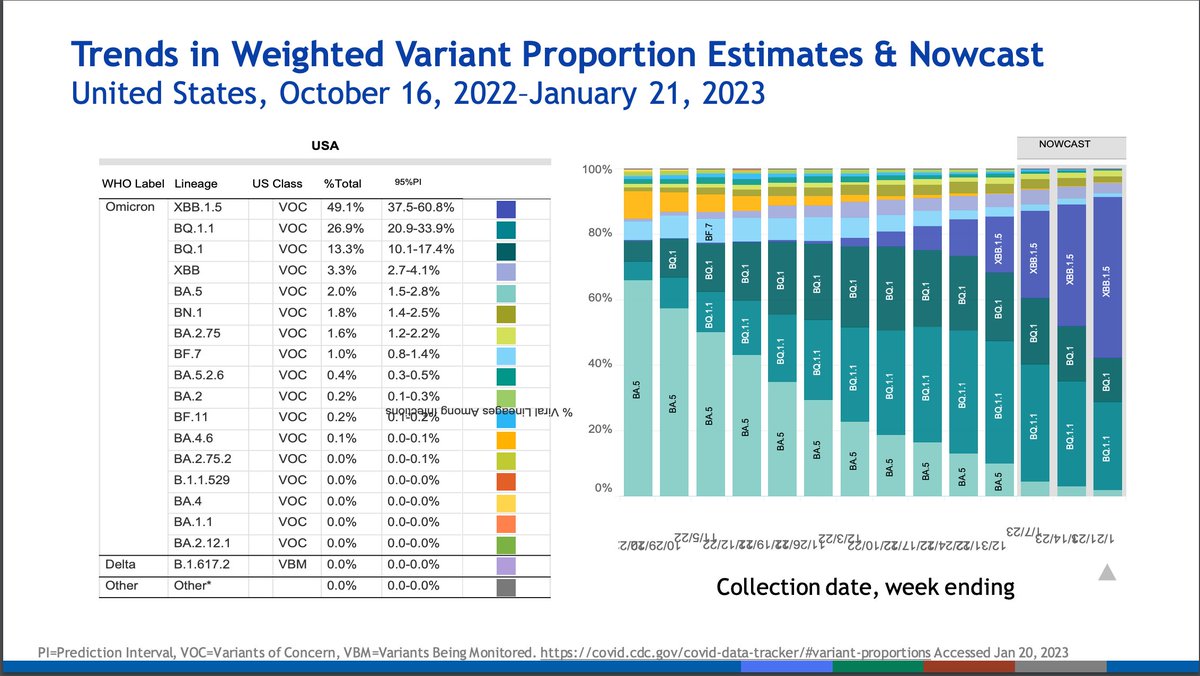
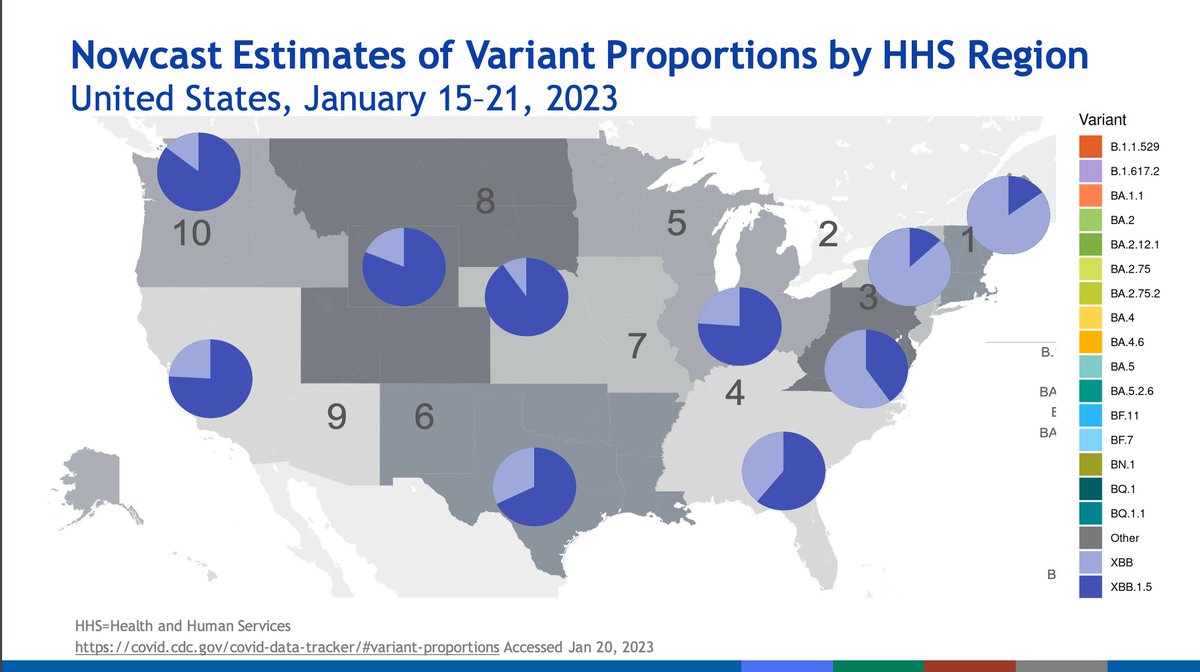
XBB lineages are mostly competing with the BQ lineages. Both are related to BA.5, and have evolved convergently to escape antibodies in the population with similar mutations. Red mutations affect monoclonal antibodies. 


More recent variants are on the far right of the graph; they have the largest relative decreases in neutralization. The bivalent booster does help some with neutralization. XBB and XBB.1 had the largest reduction in neutralization. 
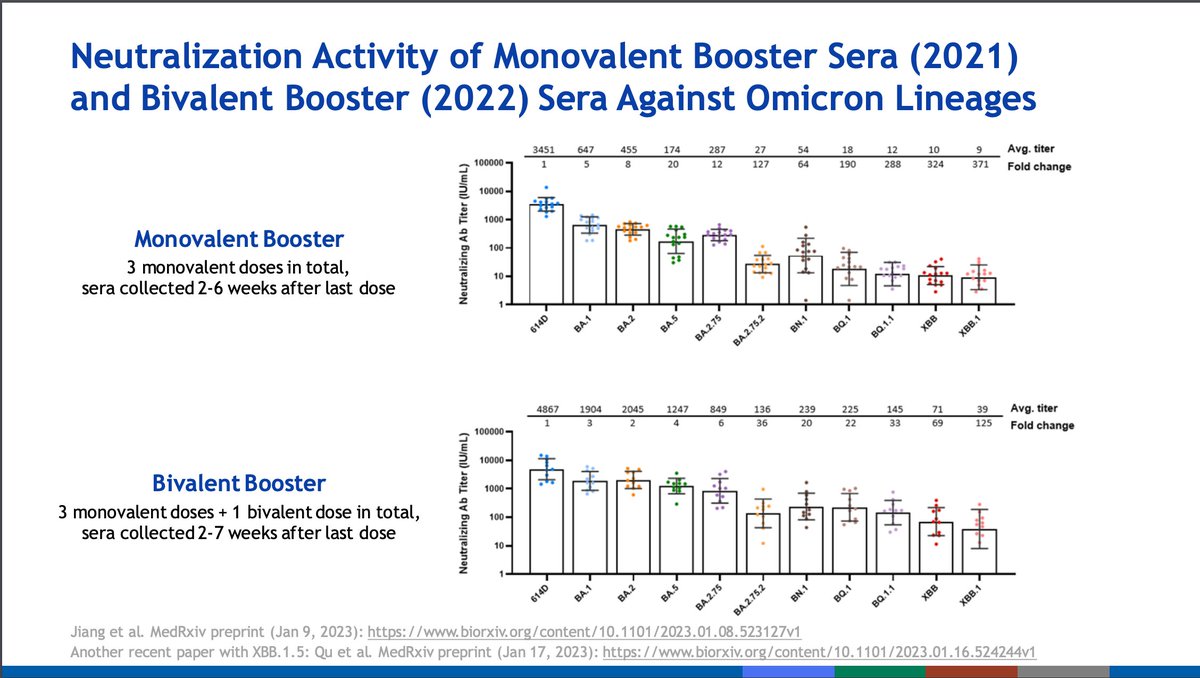
XBB.1.5 has shown similar neutralization titer to XBB.1 based on recent publications.
Because of increases in home testing, cases are underreported today and do not capture the full scale of transmission. 
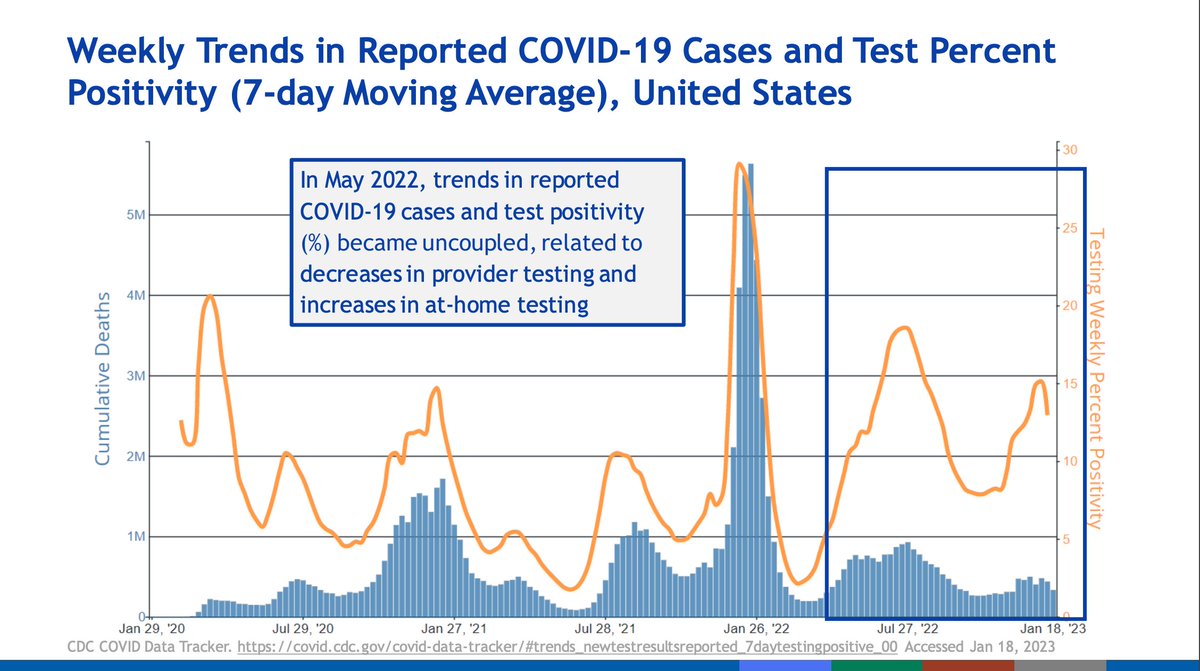
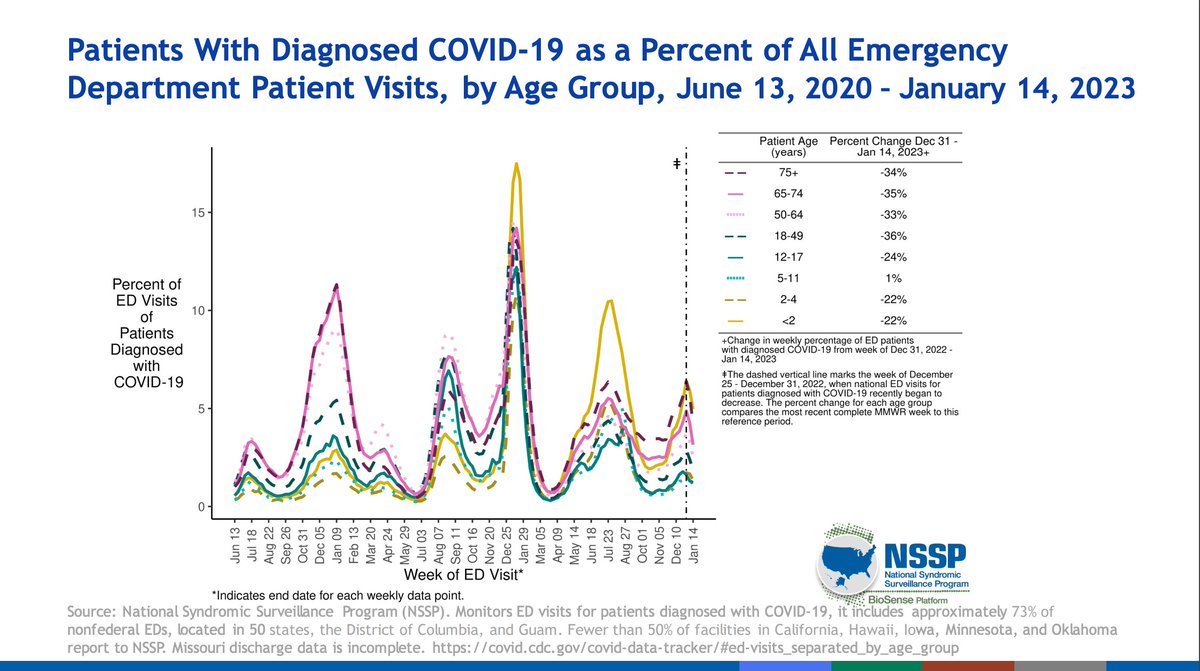
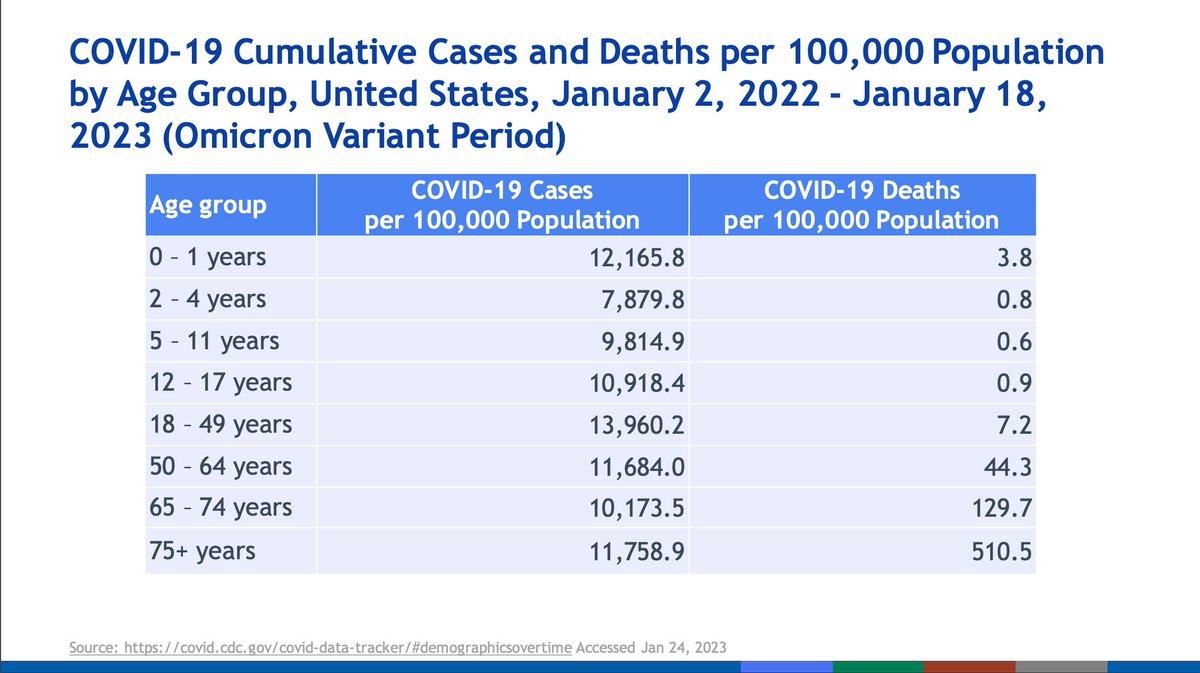
Recently older ages appear to bear more of a burden of cases but this can also reflect differences in testing practices. Young children under age 2 had the highest usage of emergency departments in recent times. 


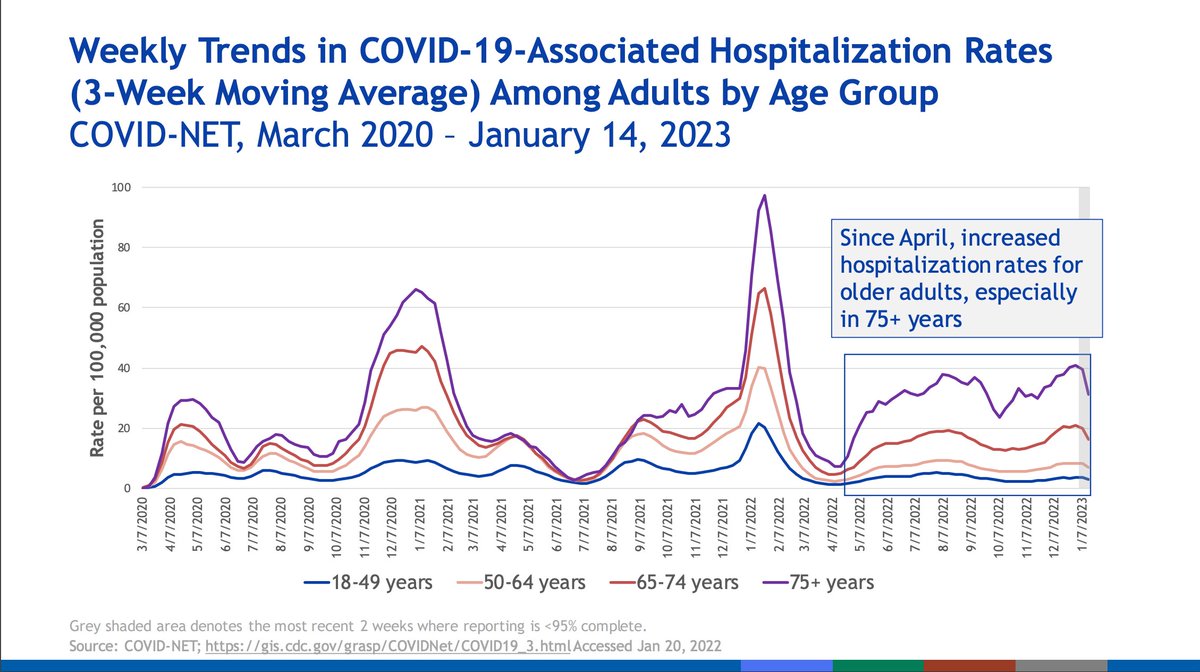
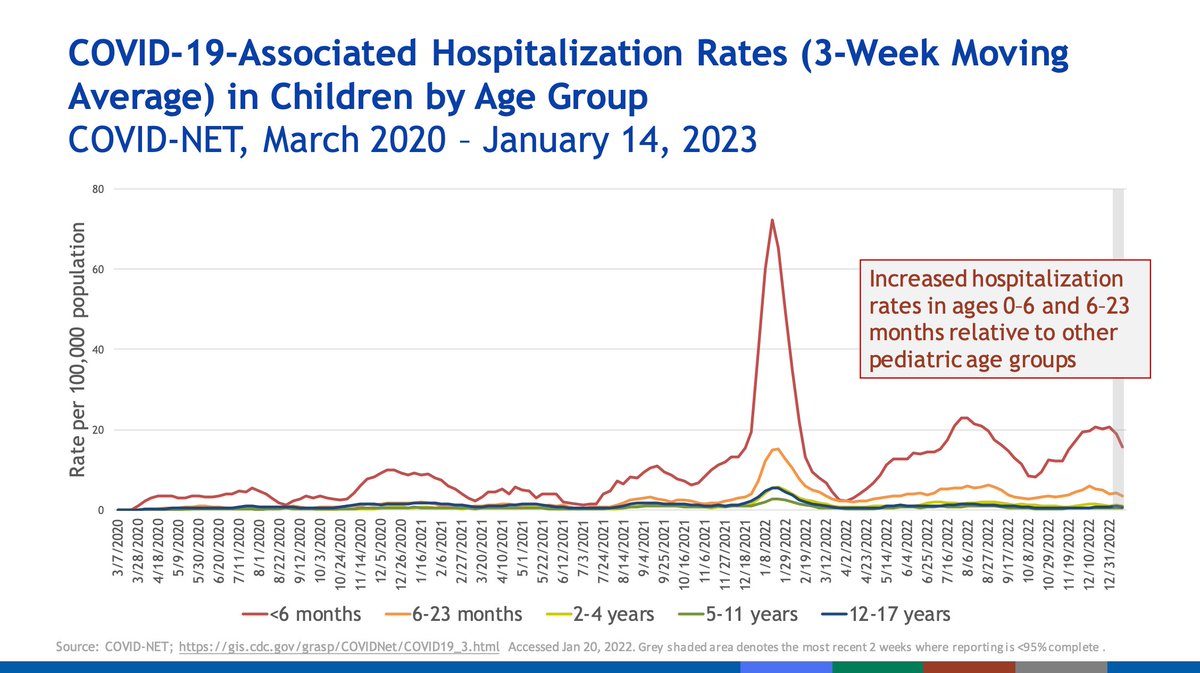
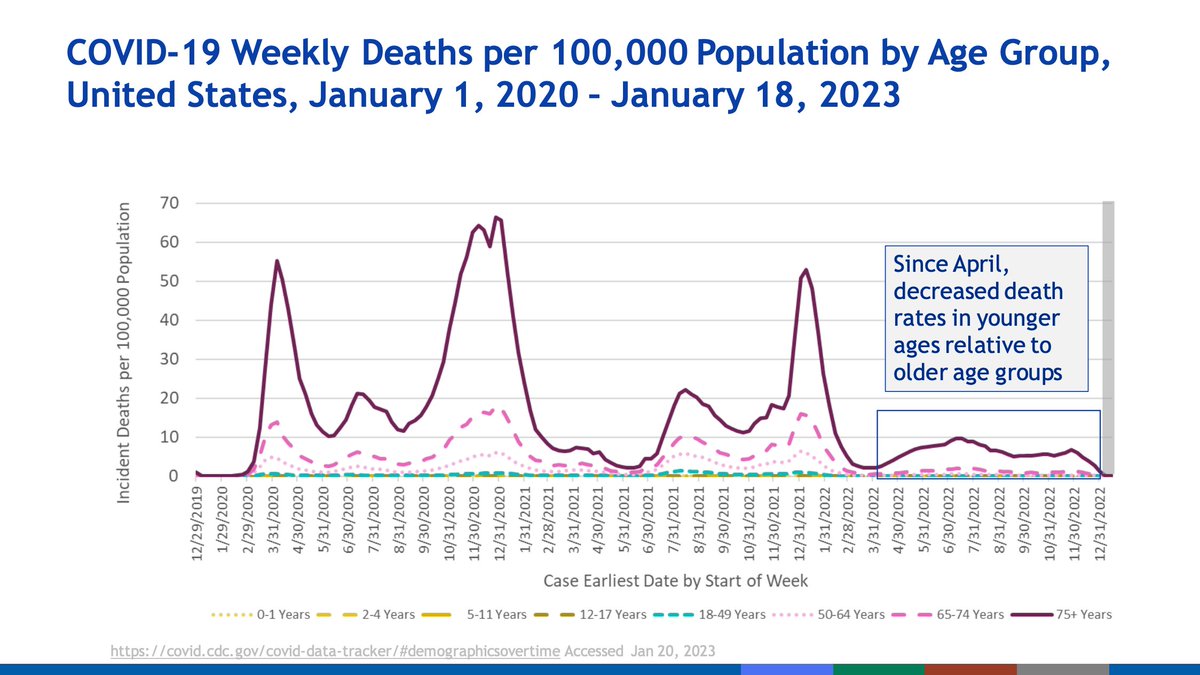
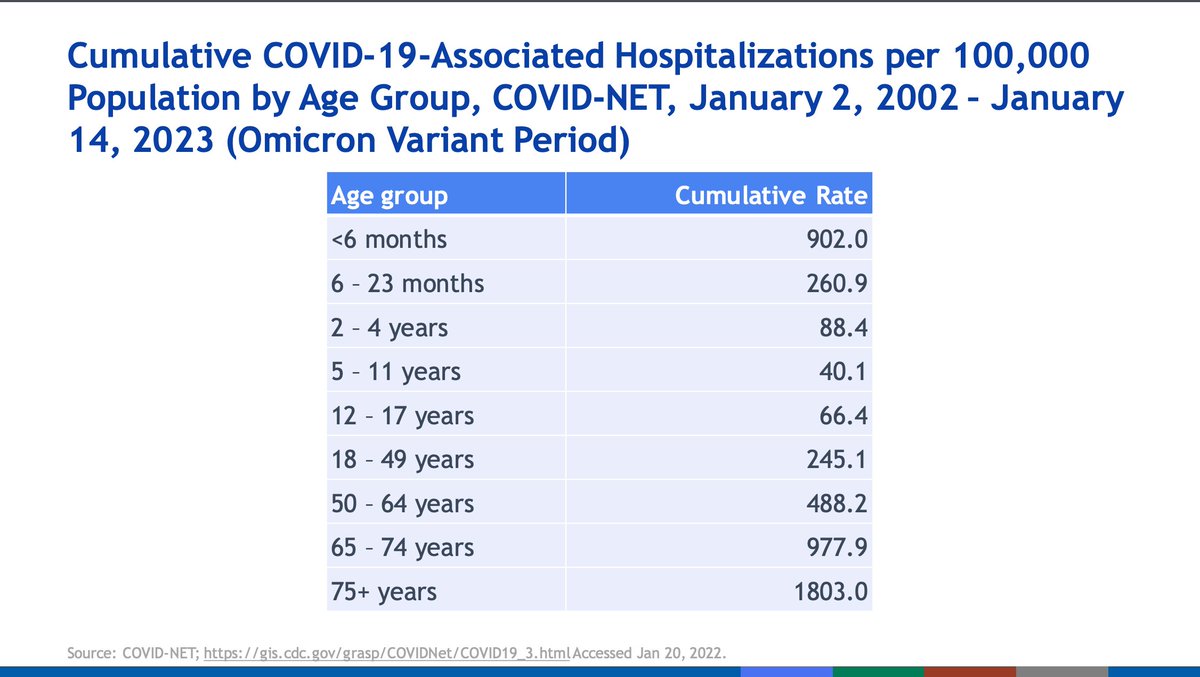
Older age groups, particularly those 75+, have had disproportionately high hospitalization rates compared with other groups. Omicron surges triggered massive surges in pediatric hospitalizations, especially children < 6 months of age who could not be vaccinated. 

The oldest in our population face disproportionately high mortality from COVID-19 compared with other age groups, particularly from age 75.
This underscores the importance of vaccination in pregnancy as well (see <6 months old incidences of hospitalization).


This underscores the importance of vaccination in pregnancy as well (see <6 months old incidences of hospitalization).
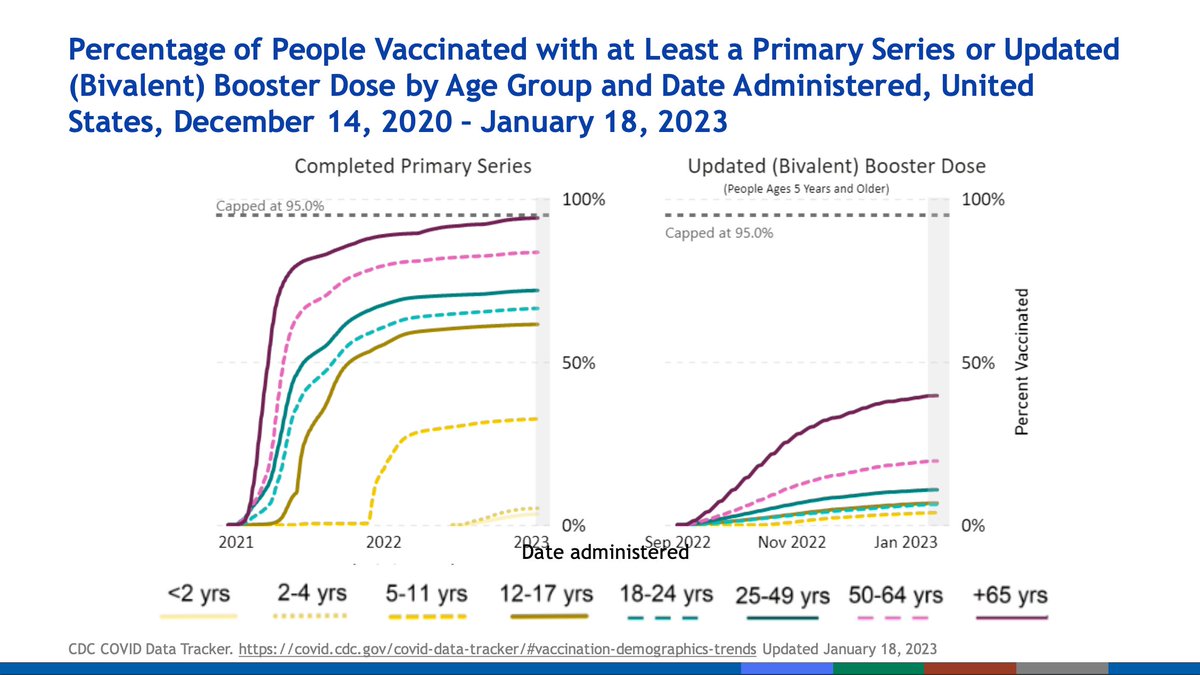
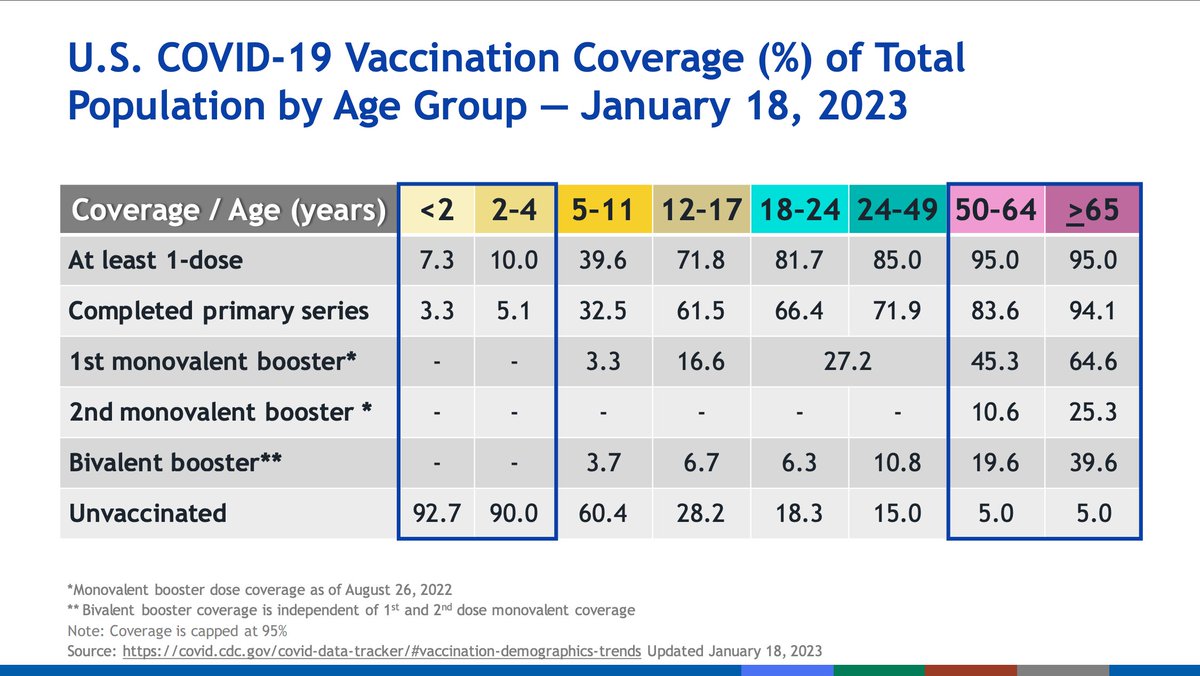
Bivalent booster uptake has lagged. 73% of the US has received a primary series (which at this point in the pandemic is pathetic). Coverage of vaccines is higher in older age gorups, which is of relative comfort. 



Higher proportions of infection-induced immunity only are seen in younger adults over time, whereas vaccine induced immunity is increased in older adults over time. 

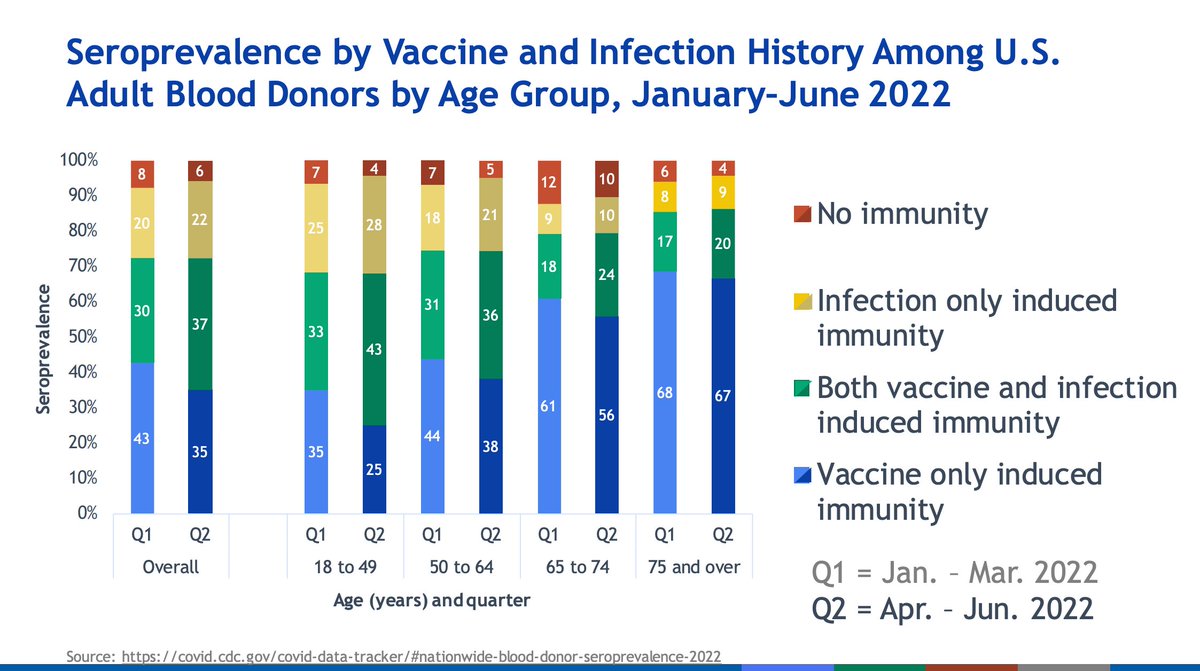
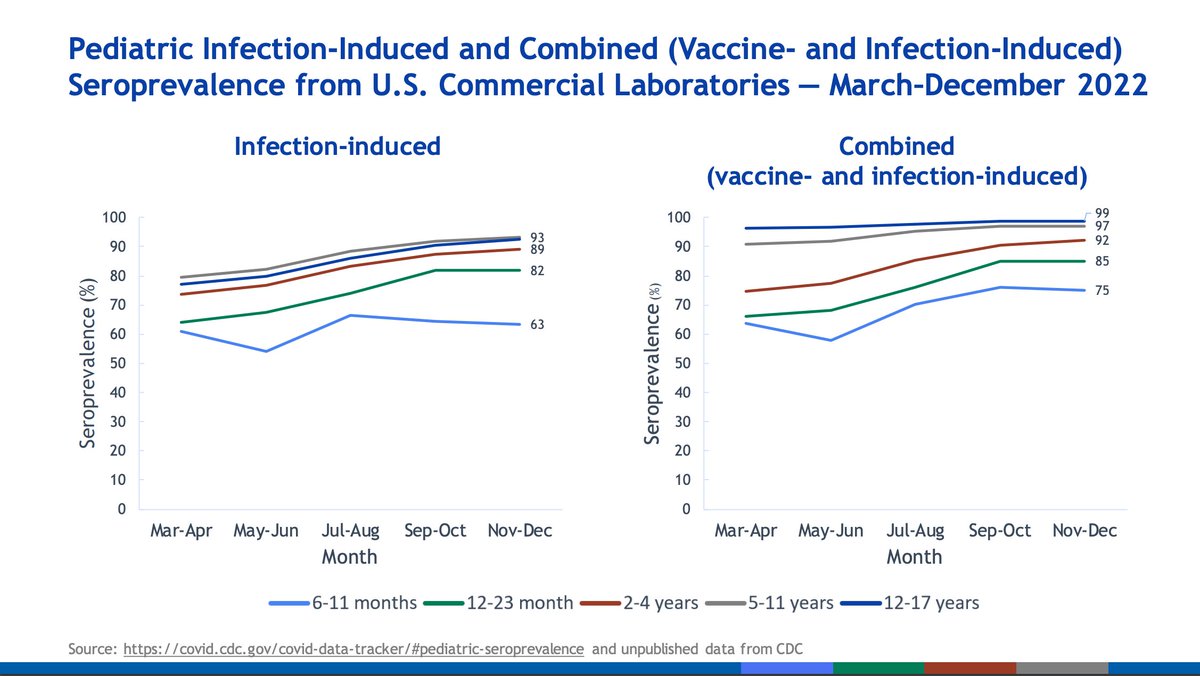
Note this is only up to June and December 2022 respectively; they are therefore likely underestimates (at least for adults for certain).
Hospitalizations have consistently been substantially higher for unvaccinated than vaccinated people in the US throughout the entire pandemic. The bivalent booster enhances protection that might decrease over time due to waning or evolution of SARS-CoV-2. 


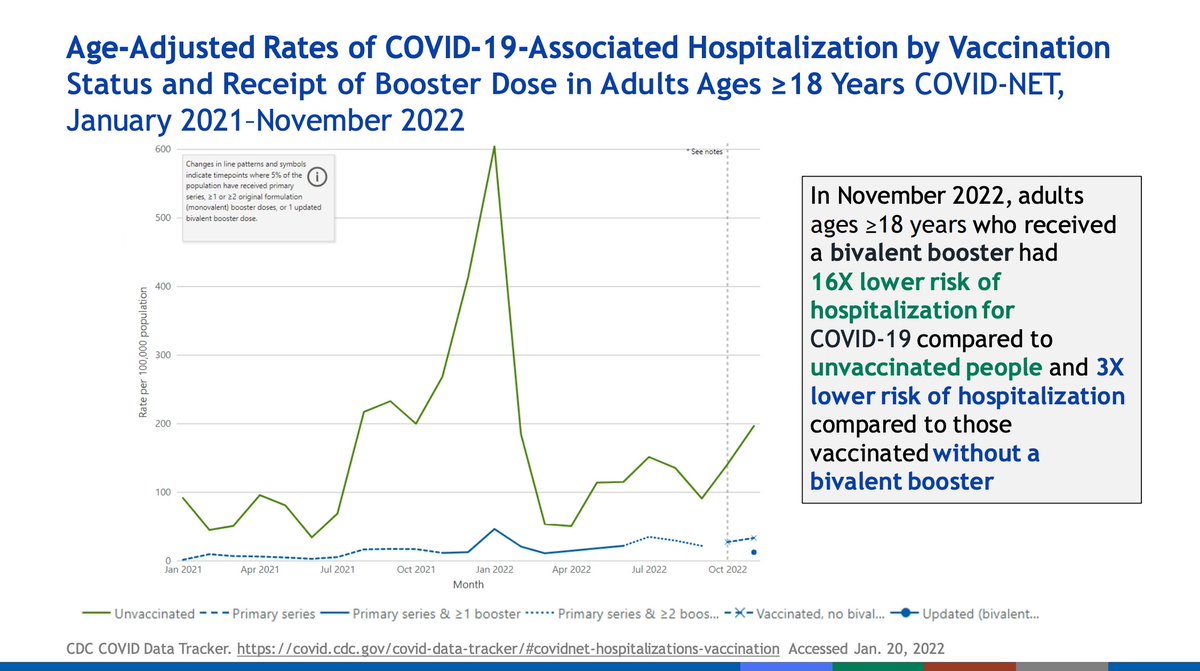
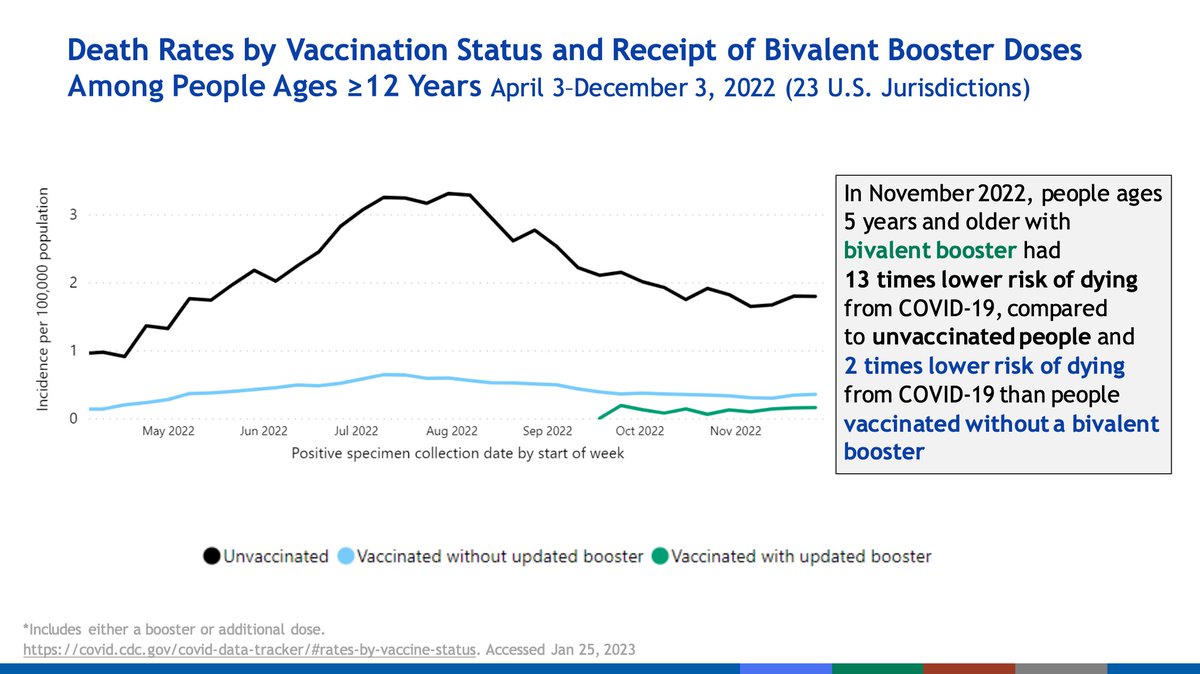

When vaccinated people develop severe COVID-19, it is overwhelmingly individuals who have multiple serious comorbidities. Antivirals are important in these individuals, REGARDLESS OF VACCINATION STATUS.
Summary slide from CDC I think says it well. Older adults and the youngest face disproportionate burdens compared with their neighboring age groups. Therapeutics are available and should be used as indicated. 

Perlman: [serum] neutralizing antibodies as a sole correlate of protection might need to be updated given protection is robust despite low titers to novel subvariants
Gans: Showed variant range by geography over a single time point- would be more helpful to look at trends over time. Important because if there is a notion to change based on evidence, geographic variation may be a consideration as well. Notion that sever disease can be...
impacted by the medications available, but we should be mindful that many of these medications are NOT available for children. Agrees with Perlman re neutralizing titers as a marker of efficacy.
Scobie: Regional variation in variants can be viewed by week on CDC site. Early on before Delta became predominant, we did observe regional circulation of some lineages e.g. Epsilon on the west coast, which did not attain predominance. We have since observed that if variants...
have a [meaningful] advantage, it sweeps throughout the world regardless of its origin. This will likely occur with XBB.1.5 as well. Time will tell. We are in a new era with variants ("soup"), they often differ by only 1 or 2 amino acids in spike, so we haven't had a clear...
winner for a while with selective advantage, but XBB.1.5 seems poised to take over now.
Hawkins: Home testing and the impact on variant composition data- COVID-19 cases are underestimated due to home testing. How does variant composition data correct for this?
Scobie: They are not currently incorporated into genomic surveillance. There are efforts to incorporate them (make my test count initiative- makemytestcount.org). CDC plans to make those data public but this is not currently part of the framework.
Scobie: People who do home tests in general are lower acuity in terms of COVID-19 severity, so if we were worried about catching all of those cases, we have a problem but current surveillance captures the variants causing higher acuity illness.
Gellin: Timing and duration of protection- what assumption are we using for how long people are protected?
Scobie: Protected against what outcome?
Gellin: Both.
Scobie: Dr. Link-Gelles will present on that question; in general, vaccines protect pretty well for 3 months...
Scobie: Protected against what outcome?
Gellin: Both.
Scobie: Dr. Link-Gelles will present on that question; in general, vaccines protect pretty well for 3 months...
against [mild/asymptomatic] infection and protect far longer against severe infection.
Meissner: In Massachusetts, they distinguish between severe disease based on whether COVID-19 is incidental or causative to a patient's hospitalization [by Dexamethasone use]. 33% were hospitalized last week BECAUSE of COVID vs. all positives. More important as we progress...
in the pandemic. If we introduce vaccines with a novel composition, it would be helpful to know who is hospitalized BECAUSE of COVID-19 rather than With. Is it possible for CDC could generate those data in addition to Massachusetts?
Scobie: COVID-NET has published papers...
Scobie: COVID-NET has published papers...
using an algorithm that looks at diagnoses and divides people out based on likely scenarios (accident, obstetric care) can provide a percentage of cases they think are there because of COVID vs. incidentally. Incidental cases increased over time. It's not broken down in...
surveillance data, but they use a reasonable case definition i.e. people tested positive within some period of time into or before admission are called COVID-associated cases. These are the data presented today. A reasonable issue to raise but the trends seen are real.
Meissner: it would be helpful if CDC incorporated dexamethasone use, it would be helpful.
Mcinnes: 101.9 million cases noted by CDC; case ascertainment mechanism was what?
Scobie: Cases reported to CDC.
Mcinnes: If these are measured cases,...
Mcinnes: 101.9 million cases noted by CDC; case ascertainment mechanism was what?
Scobie: Cases reported to CDC.
Mcinnes: If these are measured cases,...
what do you speculate the number of cases have been that did not present for care.
Scobie: Seroprevalence data were presented regarding exposure, which address this point. The vast majority of the US population has been infected, vaccinated, or both.
Scobie: Seroprevalence data were presented regarding exposure, which address this point. The vast majority of the US population has been infected, vaccinated, or both.
Jefferson Jones from CDC: In Q2 of 2022, there had been 22% of adults had infection-only immunity, 37% with hybrid immunity based on nucleocapsid serology. Likely continued to increase but there is a lot of variation by age.
There have been large increases in hybrid immunity prevalence over time; the data cannot identify reinfections but first infections have increased in prevalence greatly over time.
Dr. Link-Gelles will now present on vaccine effectiveness.
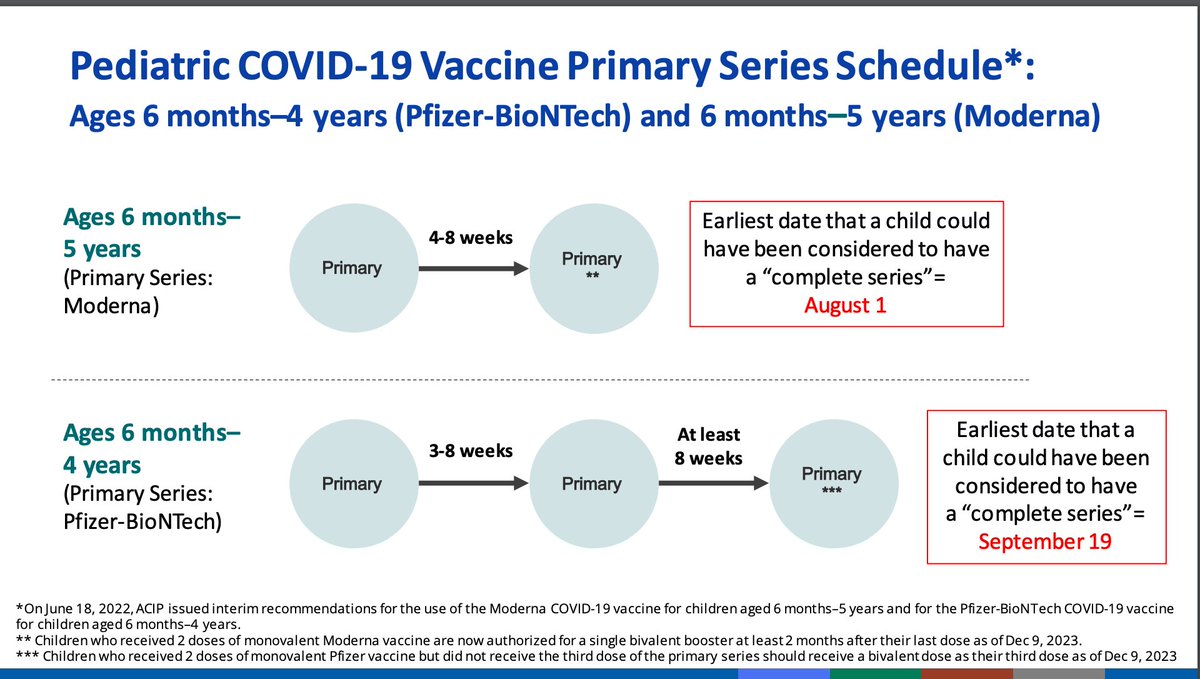
The third dose for Pfizer was changed in December to the bivalent; the analysis today focuses on the monovalent third dose. Youngest children have the lowest vaccine coverage of the entire US. Low coverage can cause fluctuations in effectiveness estimates. 
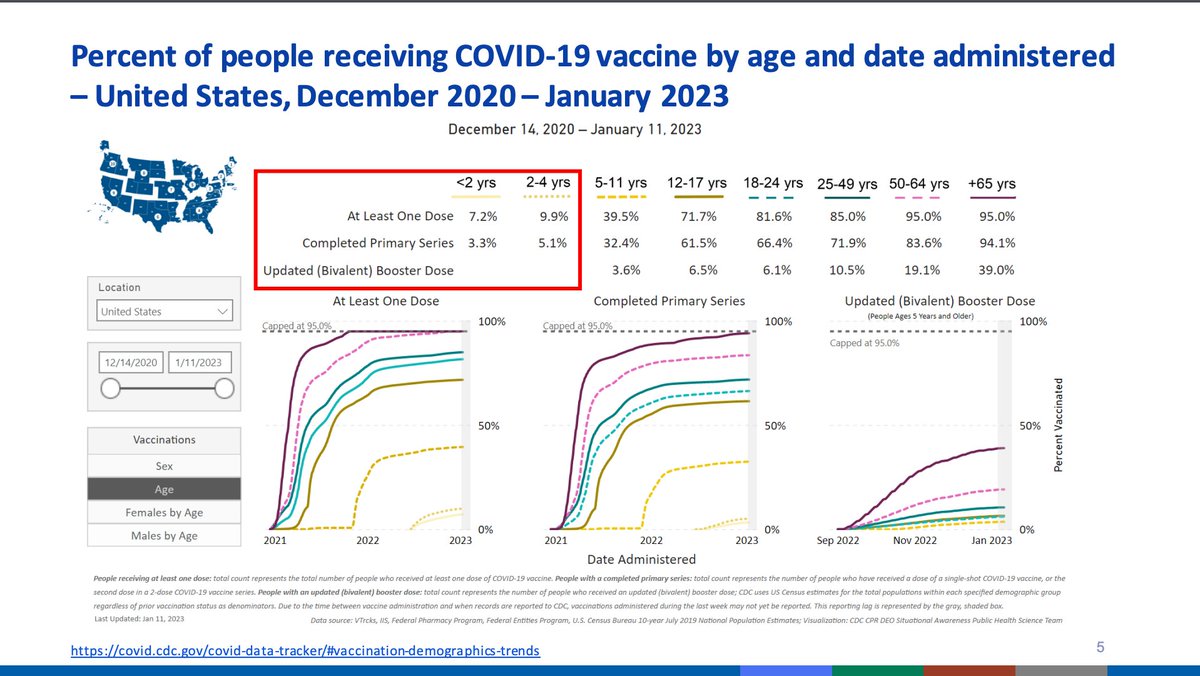
ICATT has been used to monitor VE for symptomatic infection via a test-negative design with self-reported vaccine history. This includes children as young as 3 years of age; data do not include immunocompromised individuals. 


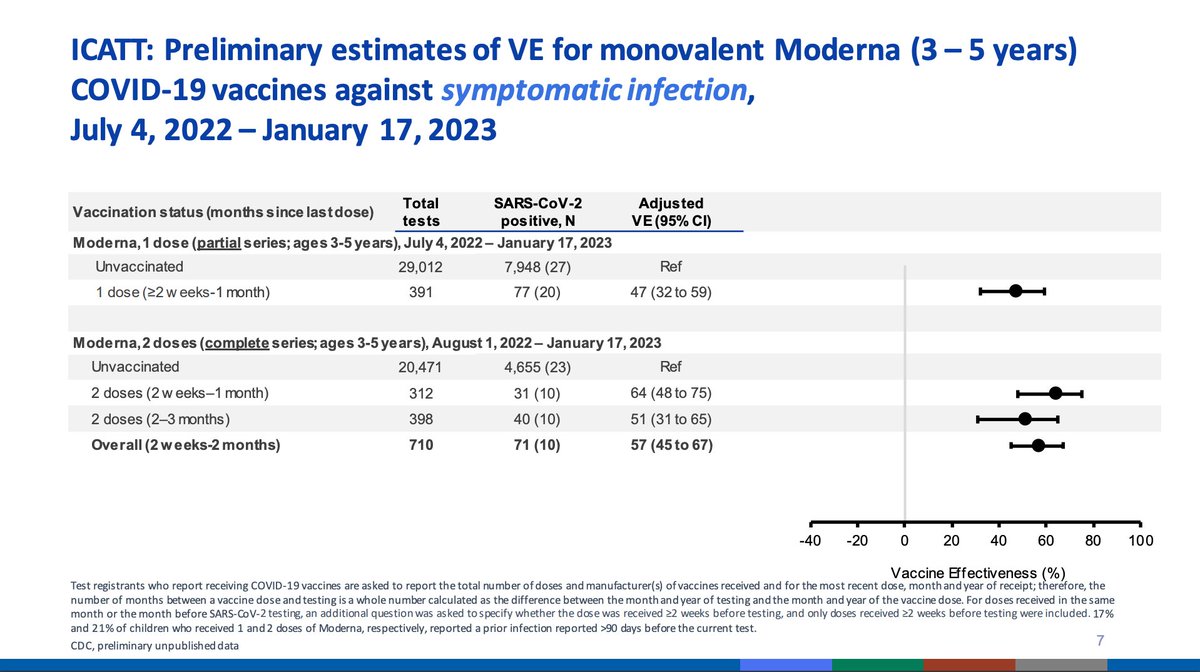
VE for symptomatic infection for monovalent Moderna series looks... okay. Not great but not zero. Effectiveness for Pfizer are harder to interpret. A single dose is not sufficient for protection from symptomatic infection. 2 doses had about 39% effectiveness but the primary... 
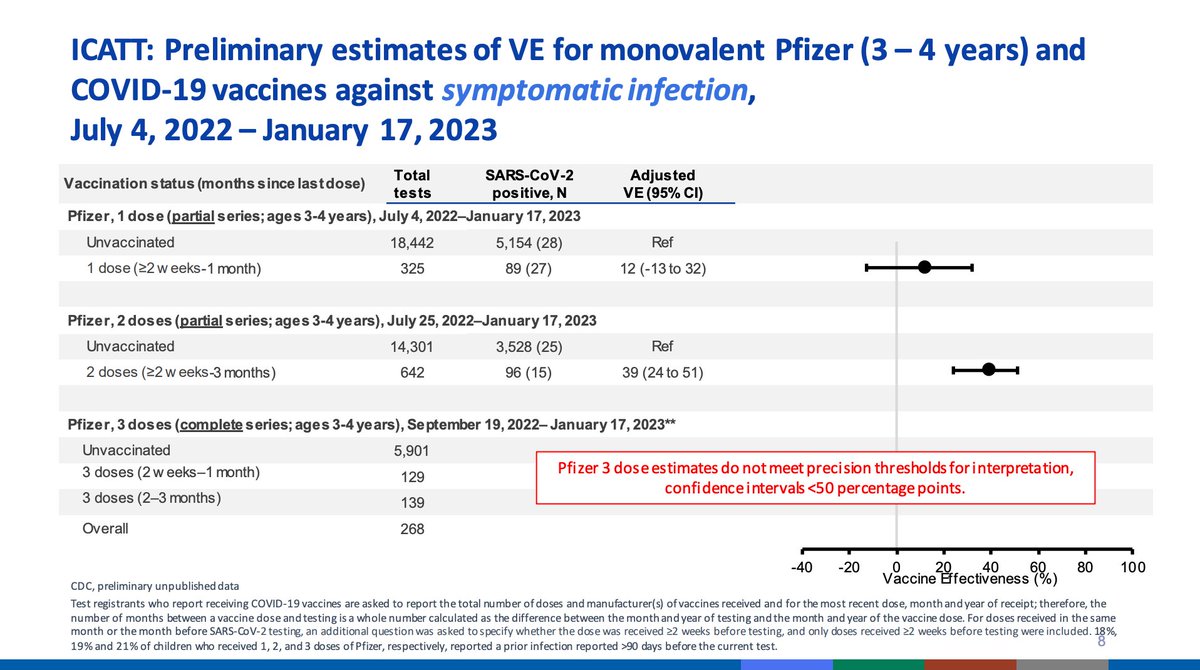
series is 3 doses, but too few cases were seen to provide reliable estimates.
Bivalent vaccine data for adults with COVID-like illness with SGTF to distinguish between subvariants. These are RELATIVE vaccine effectiveness vs no booster. VE is similar for subvariants.
Bivalent vaccine data for adults with COVID-like illness with SGTF to distinguish between subvariants. These are RELATIVE vaccine effectiveness vs no booster. VE is similar for subvariants.
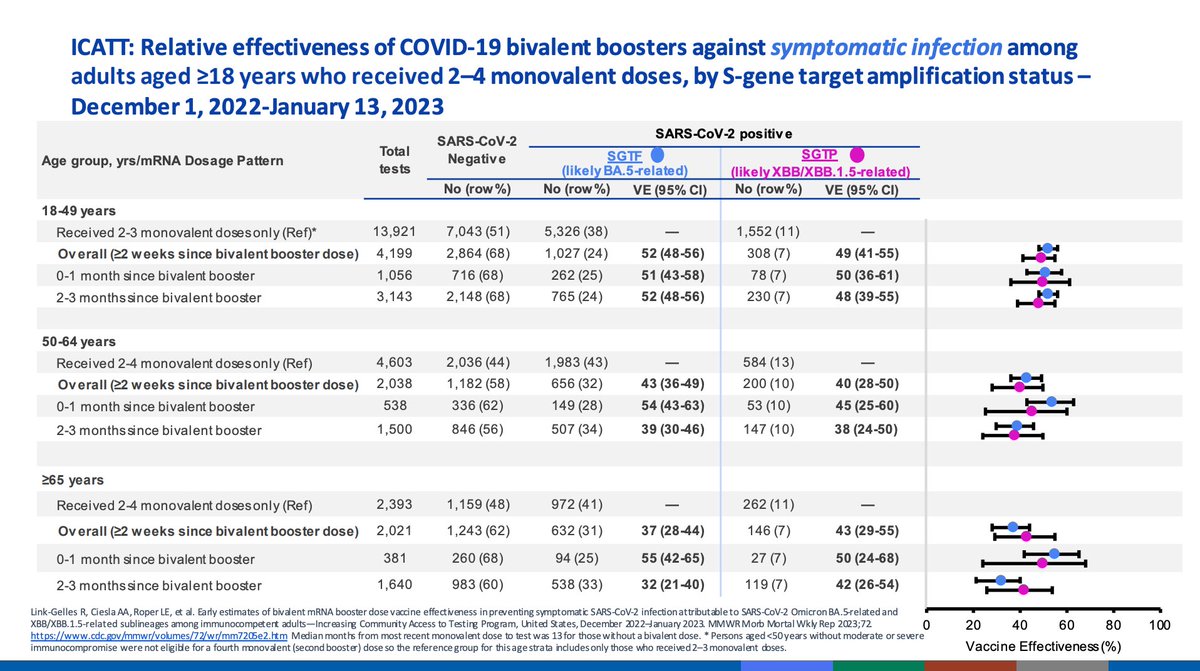
No substantial evidence of waning as of 3 months post-vaccination but continued monitoring is obviously needed.
Here is an update to a recent MMWR containing 1 additional month of data. Tbh these look very good (consider infection prevalence in the US).
Here is an update to a recent MMWR containing 1 additional month of data. Tbh these look very good (consider infection prevalence in the US).

Summary: Primary series of vaccines for young children look okay vs. symptomatic infection (not great but hell of a lot better than nothing). Bivalent vaccines greatly enhance protection from hospitalization including against XBB and BA.5 sublineages. 

Before continuing with questions, we will review safety data from CDC by Drs. Shimabukuro and Klein.
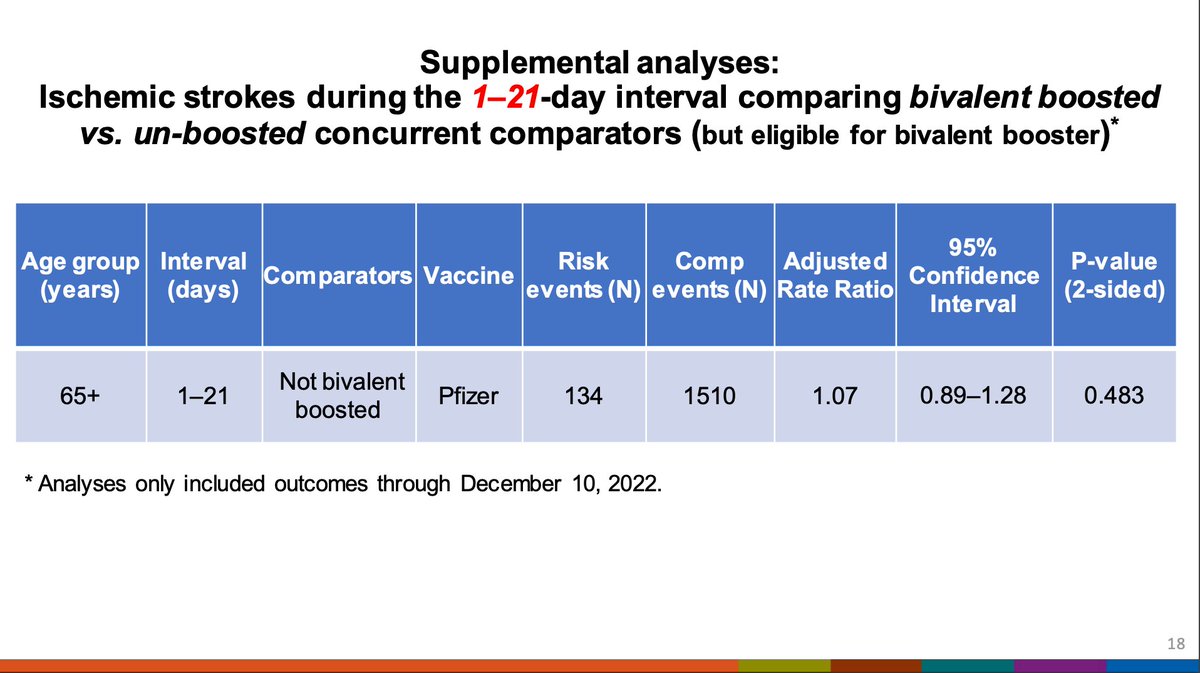
This principally concerns the finding of an increased risk of ischemic strokes in the VSD by the Pfizer bivalent vaccine (which was weird, because the signal wasn't reproduced anywhere). 

The VSD allows for rapid analyses across the US by electronic health data across all sites. It covers 12.5 million people which makes it a very powerful tool for assessing safety issues of vaccines in the US, especially rare adverse events. 

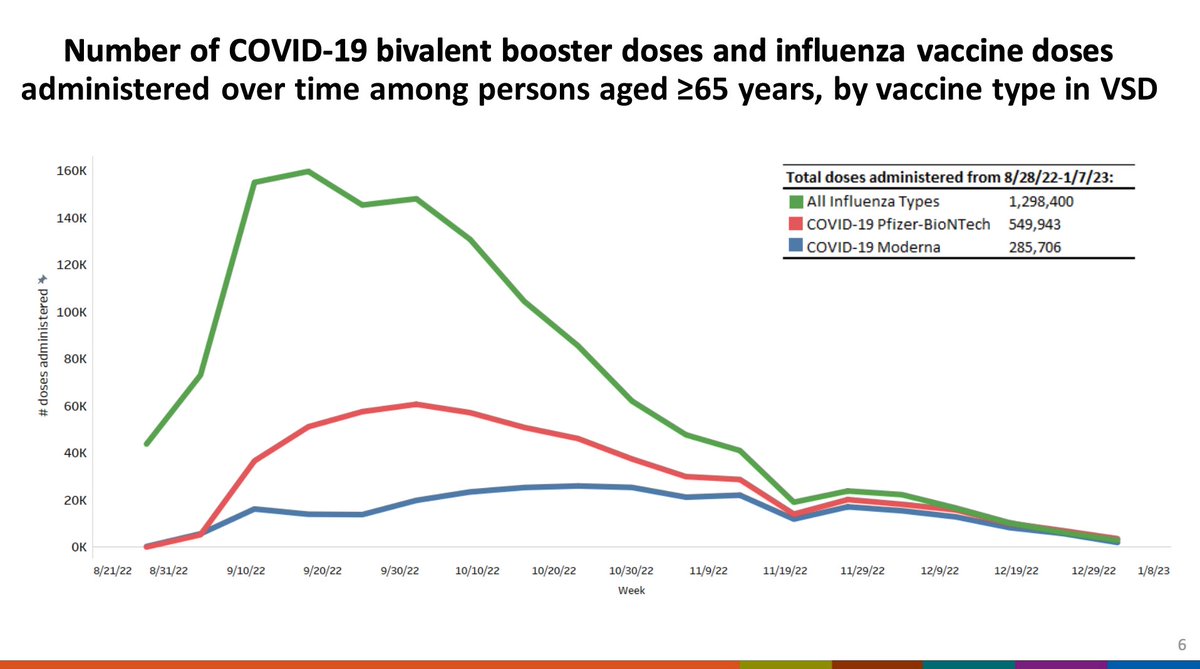
Here are the doses of flu and bivalent vaccines given via the VSD centers over time. Many more Pfizer bivalents were given than Moderna in VSD centers.
You can do nearly real-time analyses to examine comprehensive medical records to estimate risks in vaccinated vs. unvaccinated

You can do nearly real-time analyses to examine comprehensive medical records to estimate risks in vaccinated vs. unvaccinated

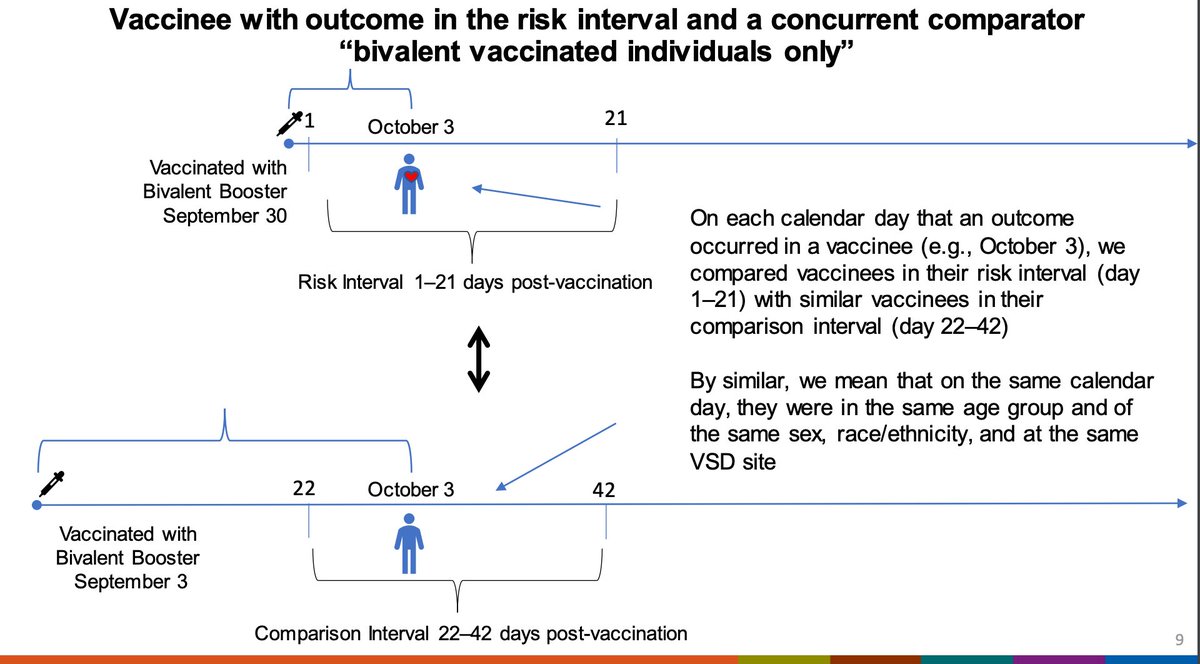
Here is a summary of how Rapid Cycle Analysis works using the VSD. A risk interval is defined and then compared with a comparator interval in matched vaccinees at the same VSD site (adjusted for age, sex, race, calendar time, etc.) 


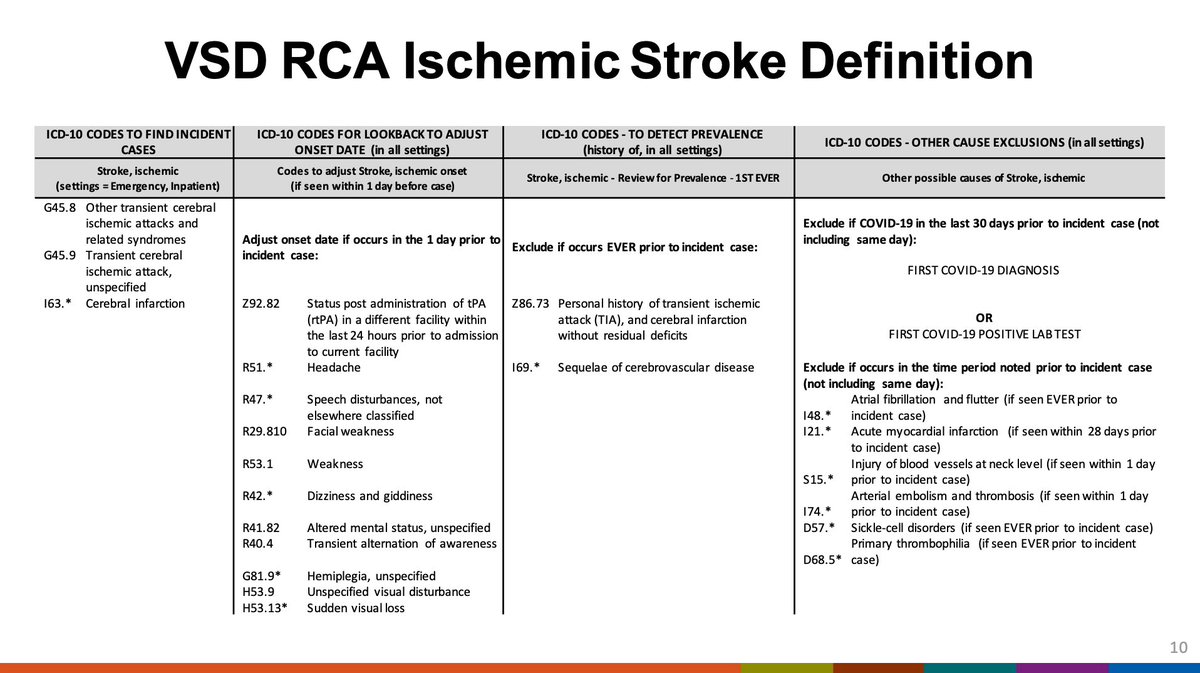
These are the case definitions for the RCA ischemic stroke analyses (error in the slide- not first COVID but any COVID) and here are the data for Pfizer and Moderna in VSD. There is 1.47 (1.11-1.95) times measured risk of ischemic stroke for the Pfizer bivalent 

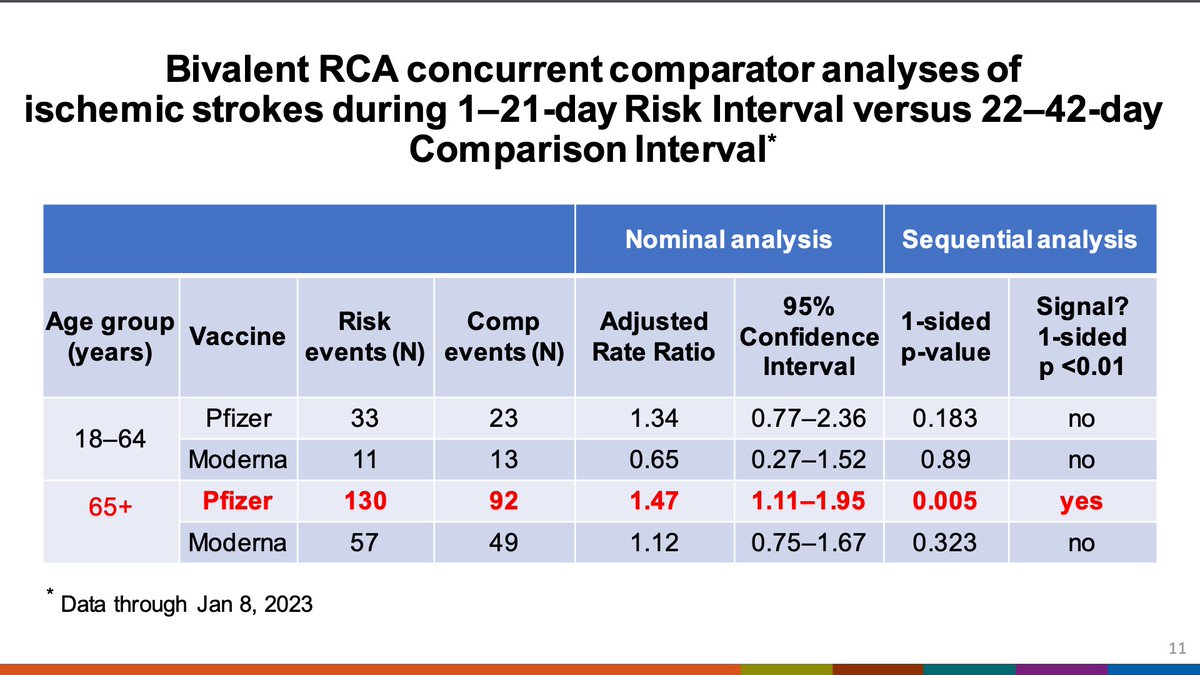
When the VSD detects a signal for RCA it undergoes a procedure to confirm the validity of the signal including data quality assessment, chart review, and further epidemiological investigations. 

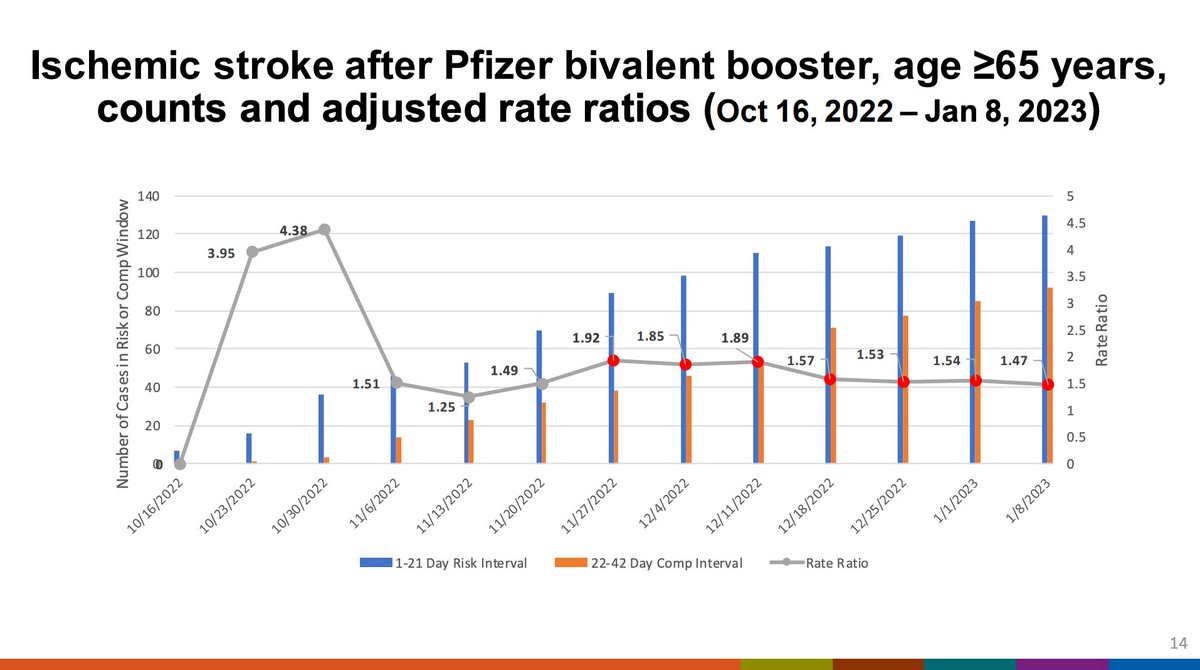
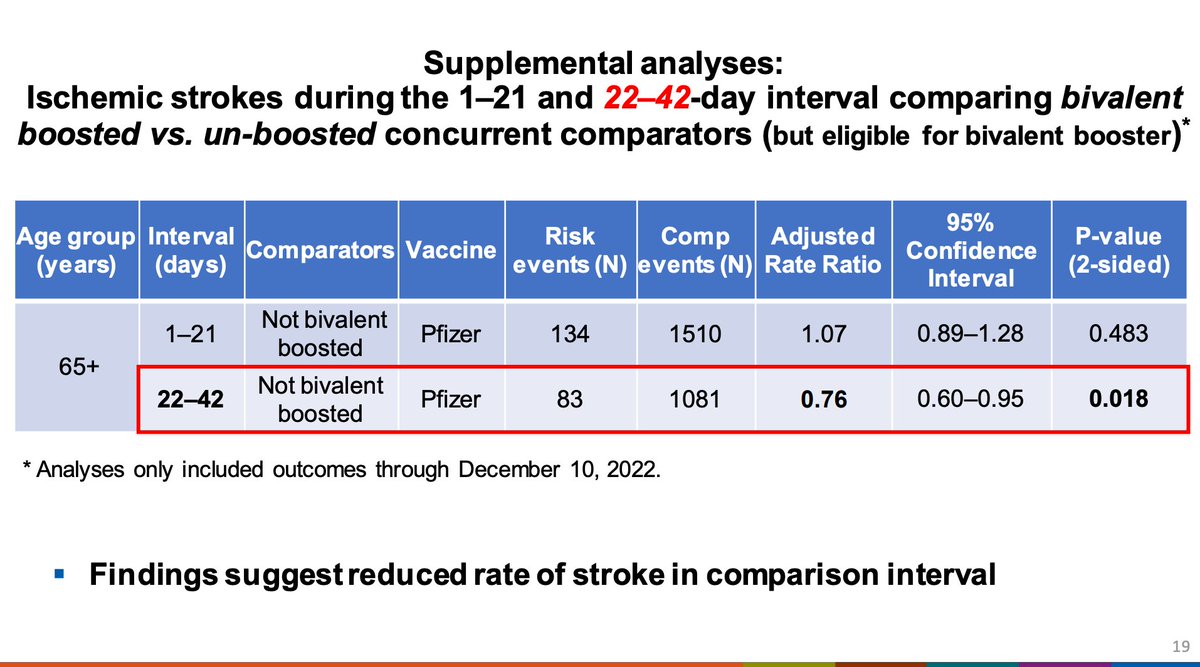
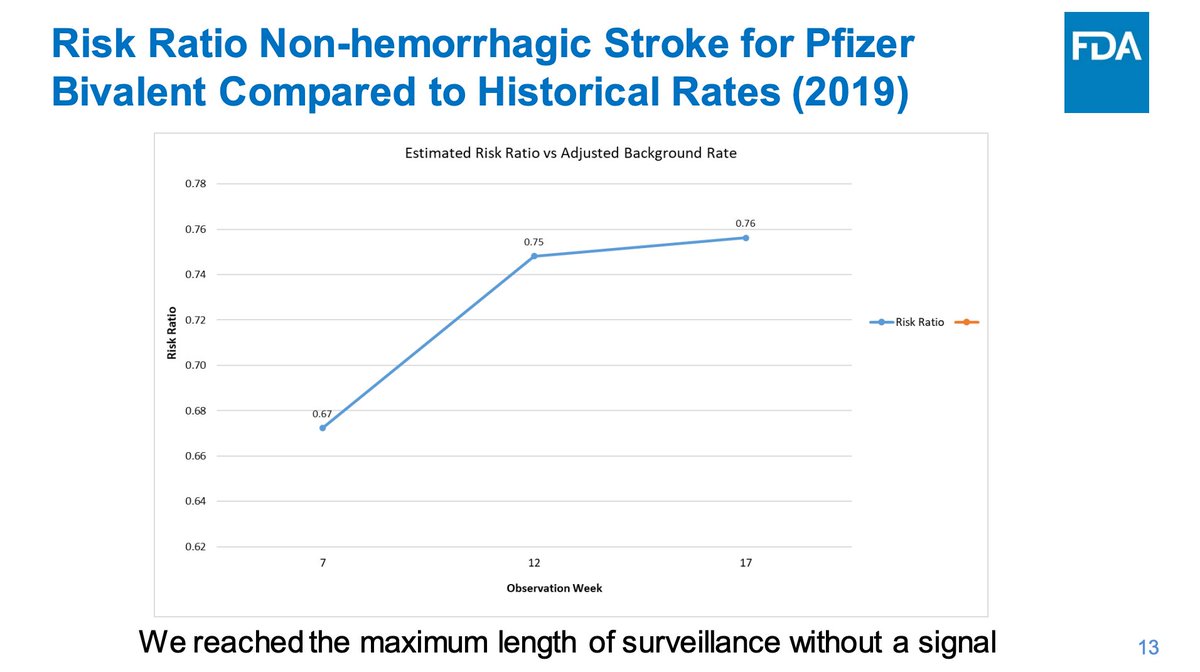
This is what the Pfizer stroke risk in older adults looks like graphically. The signal has been attenuating over time compared when it was first detected. The number of cases in the comparator interval have been increasing over time. 



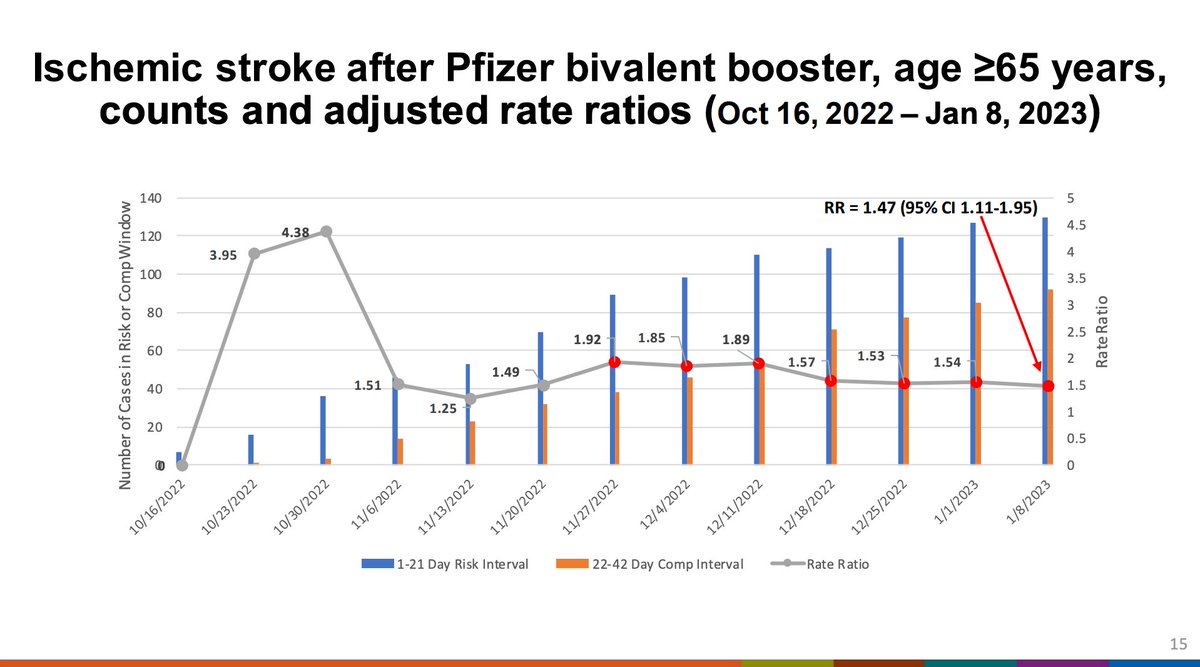
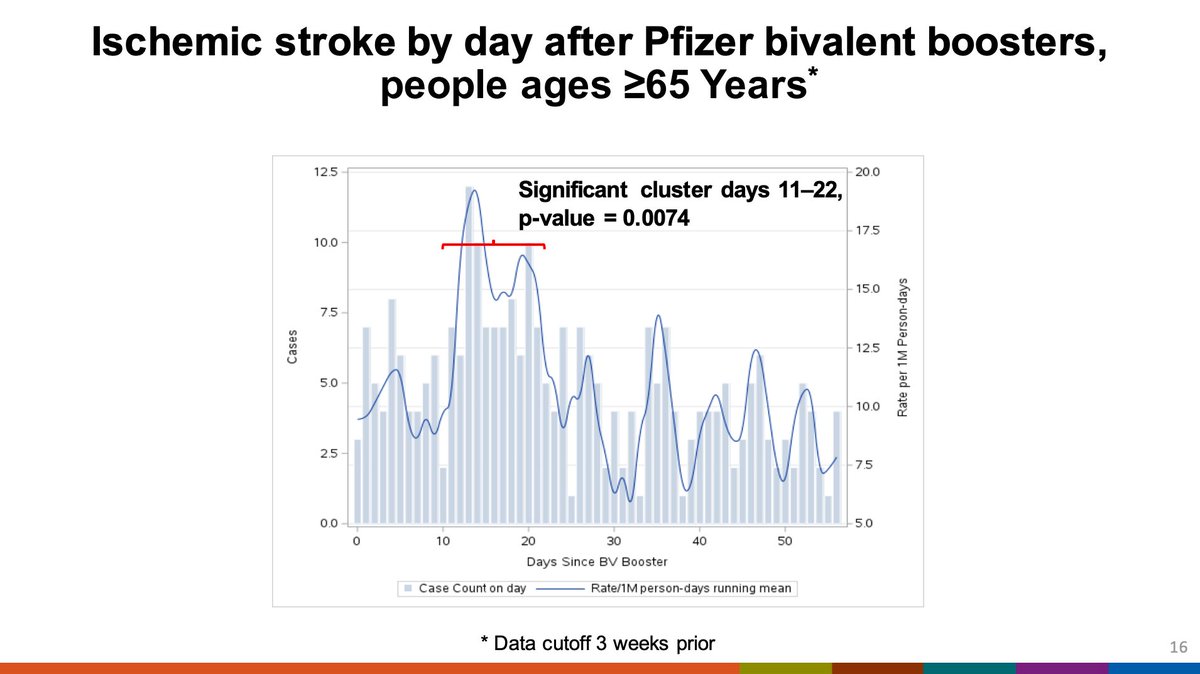

Interestingly, further analysis shows that the comparator interval seems to have a reduced risk of stroke in the comparator interval compared to what might be expected at baseline, which may be driving the signal. 

Further analysis implicated the risk was coming from those who received both Pfizer and a high-dose or adjuvanted flu vaccine. Without the flu vaccine, this signal is not apparent. Again, unusually low risk in comparator is seen. 

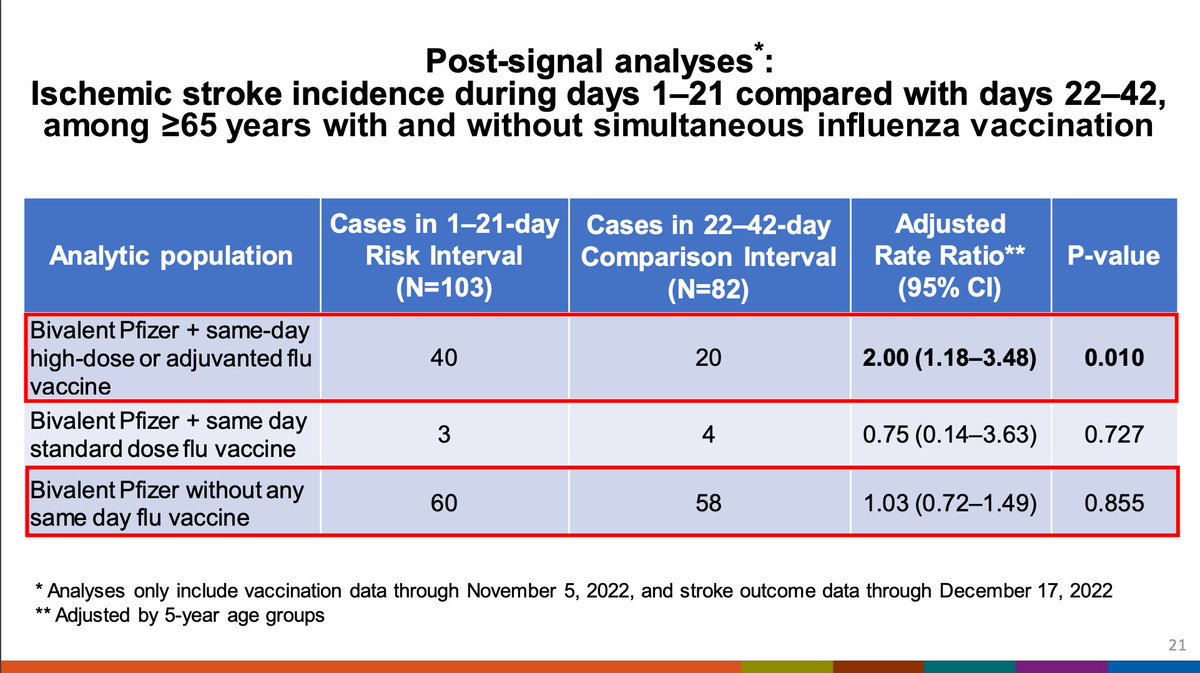

This past week, the signal attenuated to no longer being apparent per Dr. Klein. This signal is weird because it is seen only for stroke but not heart attack. Possible roles of SARS-CoV-2 infection may help explain it but it's hard to tell 



Note from me (not the meeting): a recent large analysis of the safety of the bivalent was published as a preprint in Israel and found no increased risk in older adults:
papers.ssrn.com/sol3/papers.cf…
papers.ssrn.com/sol3/papers.cf…
CDC will continue to investigate this and in particular the role of concomitant administration of high dose/adjuvanted flu vaccine and the bivalent. 



Gans- What other safety signals are being monitored routinely and on what schedule are they reviewed?
Shimabukuro refers to this slide with specific adverse events of interest and the settings they are looked in:
Shimabukuro refers to this slide with specific adverse events of interest and the settings they are looked in:

Gans- how are we monitoring long COVID e.g. VAERS?
Shimabukuro- VAERS is a spontaneous reporting system; it is challenging to monitor long COVID because it would be multiple sign and symptom codes. Some outcomes are difficult to monitor by RCA.
Shimabukuro- VAERS is a spontaneous reporting system; it is challenging to monitor long COVID because it would be multiple sign and symptom codes. Some outcomes are difficult to monitor by RCA.
Klein- the type of outcomes best suited for RCA have to have clear diagnostic codes, close proximity to vaccination (days to weeks, rather than weeks to months). We don't want to be constantly examining false signals. VSD can investigate long covid but not by RCA.
Reingold: People with flu have an increased risk of stroke and heart attack for a while after recovery. If this is true for COVID-19, consider how much COVID you prevent and how much of those outcomes you prevent IF there is an increased risk of stroke with vaccination.
Shimabukuro- Good point, will be discussed in that context in future ACIP meetings.
Forshee will now present the FDA's surveillance data for the safety and effectiveness of the bivalent vaccine via the BEST (that is an acronym) surveillance system. 
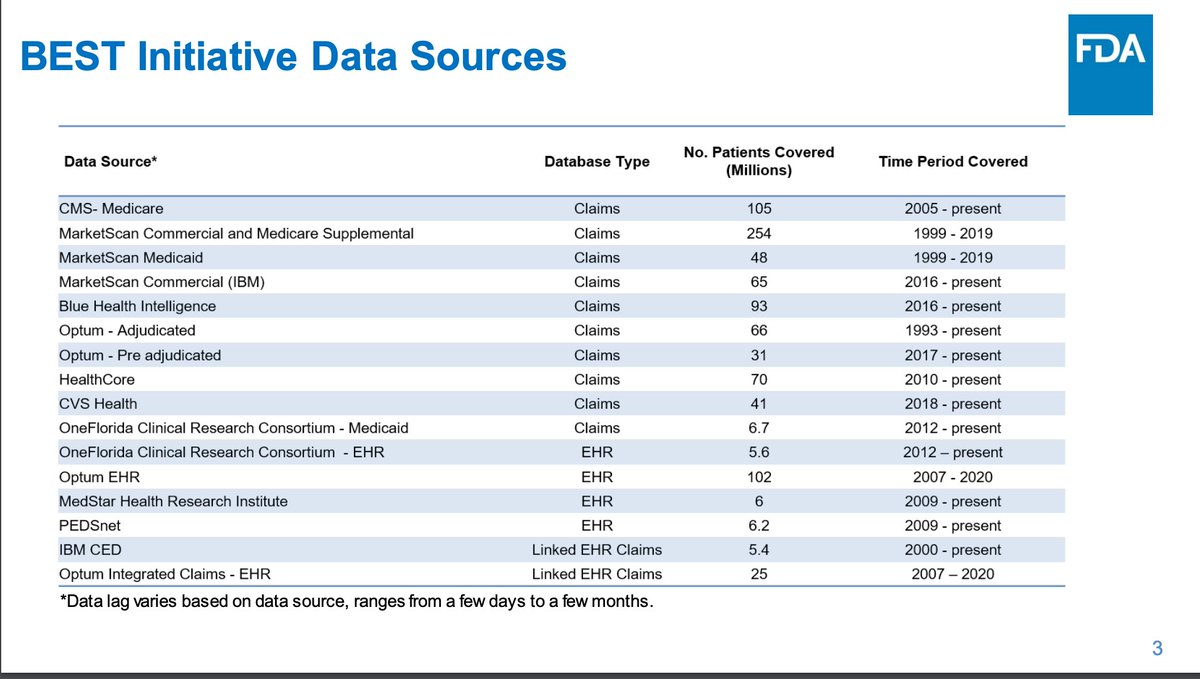
BEST RCA included 36 million individuals 65+ via CMS/Medicare. There is a concern that the vaccines were not always reported in health claims databases. To help address this, IISes have been used to supplement the data. 



There is a process to active surveillance of vaccine safety. 7.3 million bivalent vaccine doses have been given, 4.2 million of which are from Pfizer which can be analyzed. Rapid cycle analysis cannot establish a causal association (it is signal detection, not evaluation). 





FDA monitoring of outcomes of interest is similar to CDC's. FDA's definition excludes TIAs (ministrokes) but CDC's does not. No signal is apparent for non-hemorrhagic stroke. Only myocarditis/pericarditis has shown a signal of the outcomes of interest. 


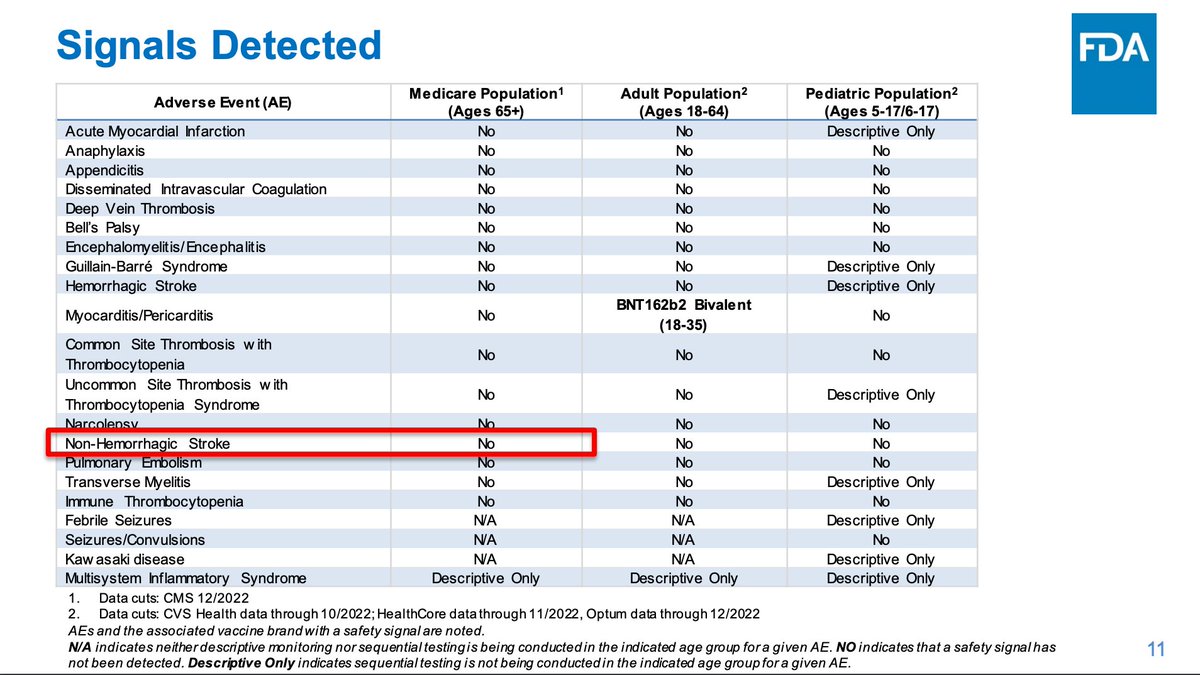
These outcomes have completed surveillance without a signal- note that neither Pfizer or Moderna have produced a signal for non-hemorrhagic stroke. 


Re concomitant flu vaccination in this age group, which was emphasized in the last presentation, a signal for non-hemorrhagic stroke was not seen. Further investigation is being done by influenza vaccine type to confirm. 

This is the summary of safety for current safety findings from FDA BEST for the vaccine. A signal is seen for the bivalent and myocarditis in males 18-35 (which is not surprising given the history of mRNA vaccines) but otherwise no additional risks are seen. 



FDA has also done surveillance specific to nursing home residents to understand effectiveness of vaccines in this patient population given their extraordinarily heightened risk. This is about the Delta period though so unsure why we're talking about it now. 




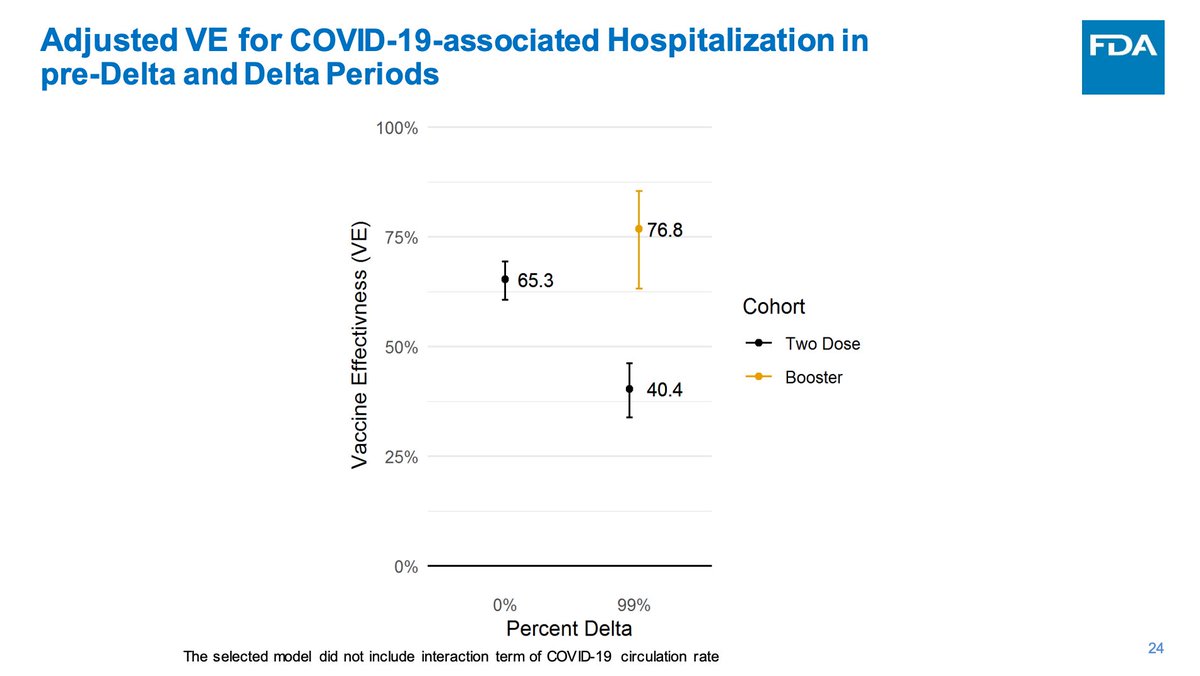
In essence, the booster really mattered during the delta period for nursing homes and still does (roughly 2-fold reduction in risk of death with 2 doses to roughly 10-fold reduction with the booster; roughly 2-fold to 4-fold with hospitalization). 




Chatterjee- Ischemic vs non-hemorrhagic stroke definition (VSD used ischemic, FDA used non-hemorrhagic); what's the difference?
Forshee- VSD included TIAs, FDA did not.
Chatterjee- Study FDA is planning, with the signal from VSD RCA would it be prudent to separate...
Forshee- VSD included TIAs, FDA did not.
Chatterjee- Study FDA is planning, with the signal from VSD RCA would it be prudent to separate...
the doses of flu vaccine and flu vaccines for those 65+. How will FDA investigate this?
Forshee- self-controlled analysis with defined risk and control windows after COVID-19 vaccination and specifically address concomitant vaccination by looking at individuals receiving...
Forshee- self-controlled analysis with defined risk and control windows after COVID-19 vaccination and specifically address concomitant vaccination by looking at individuals receiving...
adjuvanted or high-dose flu vaccine on the same day as their COVID bivalent boosters.
Chatterjee- unclear on whether the study is a passive or active analysis?
Forshee- Study will use CMS health claims data (active surveillance) but no intervention is required.
Chatterjee- unclear on whether the study is a passive or active analysis?
Forshee- Study will use CMS health claims data (active surveillance) but no intervention is required.
Reingold- positive SARS-CoV-2 tests incidentally; could this lead you to underestimate VE?
Forshee- yes.
McInnes- Plea for consistency of language usage; coadministration has a different context- can be pulling up 2 vaccines in the same syringe which is distinct from...
Forshee- yes.
McInnes- Plea for consistency of language usage; coadministration has a different context- can be pulling up 2 vaccines in the same syringe which is distinct from...
concomitant administration of vaccines (meaning 2 separate vaccines given at the same visit), so please harmonize the language.
Nelson- agree with Gans re important to report negative findings with signals. Identified a myo/pericarditis signal. How does that compare to...
Nelson- agree with Gans re important to report negative findings with signals. Identified a myo/pericarditis signal. How does that compare to...
other studies done by FDA to do deeper investigation across studies? Are there privacy issues?
Forshee: Follow up studies are done with any signals detected, medical records requested. Association seen with mRNA vaccines and in other systems. This is considered throughout...
Forshee: Follow up studies are done with any signals detected, medical records requested. Association seen with mRNA vaccines and in other systems. This is considered throughout...
decision-making for approvals for vaccines. Some of those studies are published.
Dr. Beigel of NIAID will discuss how to evaluate next-gen COVID-19 vaccines. The prior discussions established the need for next-generation vaccines. To help frame how we think about this: current strategy is to use antigenically diverse strains via CoVAIL study. 



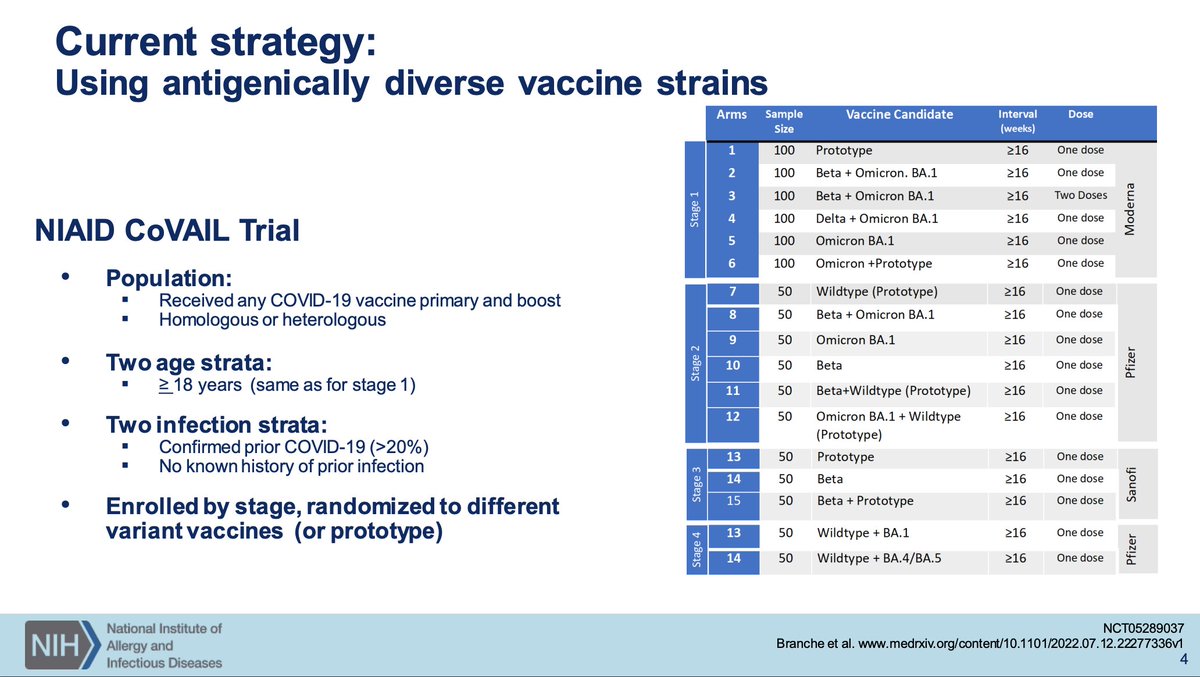
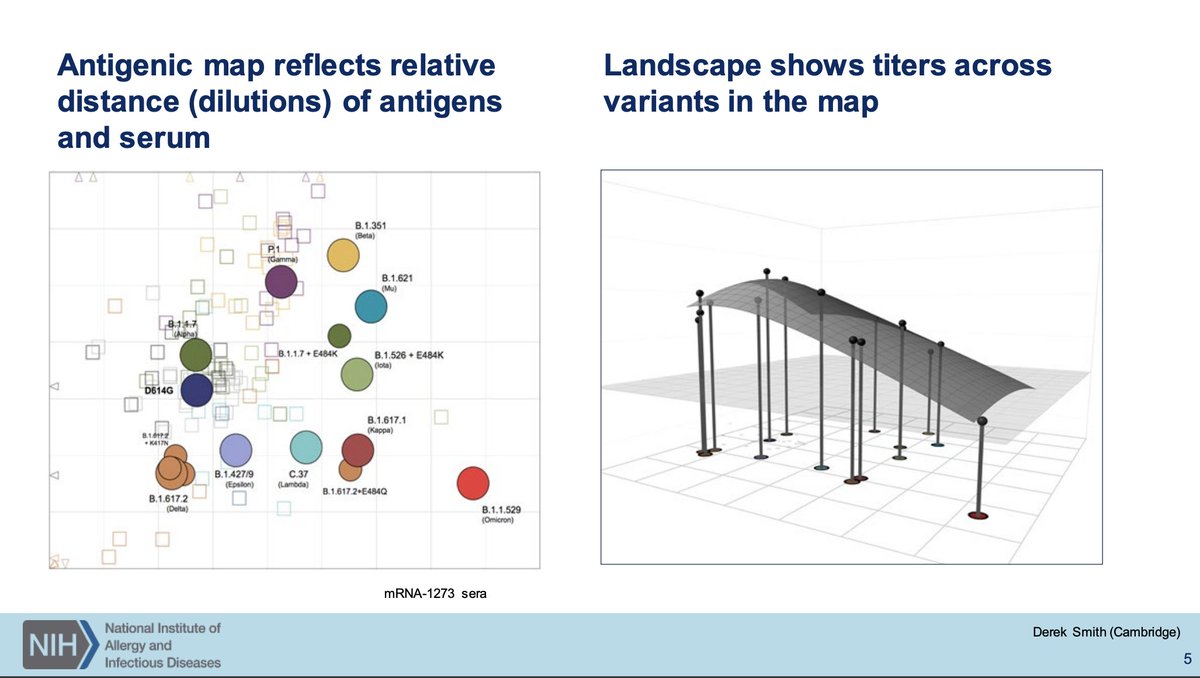
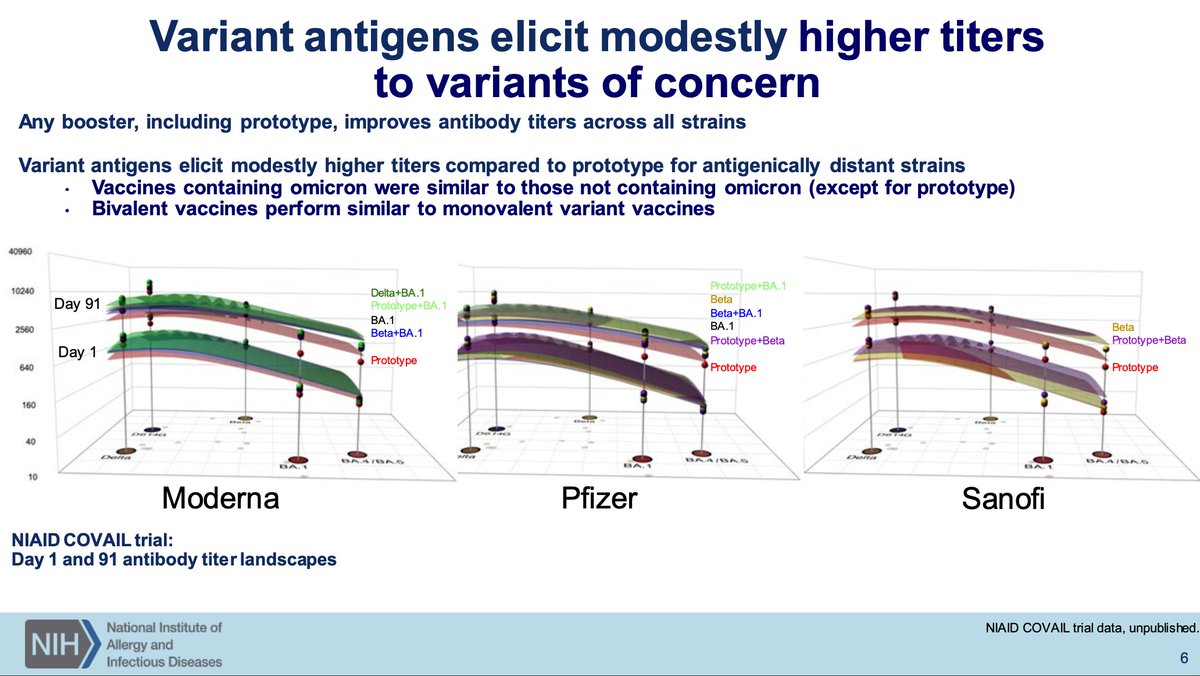
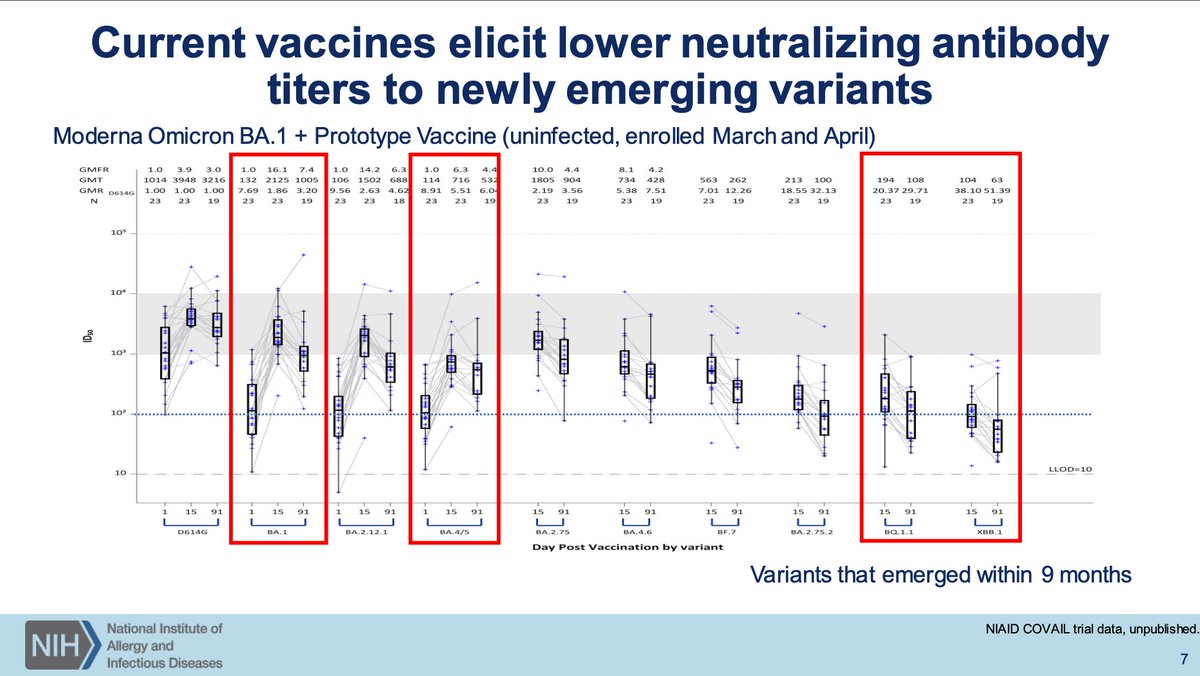
The updated vaccines do improve neutralization to divergent variants but not massively so relative to the monovalent. Increases are also seen with another monovalent dose. Beta seems to be quite nice as an antigen, even with Sanofi.
Serum neutralizing antibody titer has emerged as a correlate of protection from symptomatic SARS-CoV-2 infection with higher titers associating with superior protection. I didn't notice initially but Novavax has a weirdly high number of hypo/nonresponders. Manufacturing problem? 
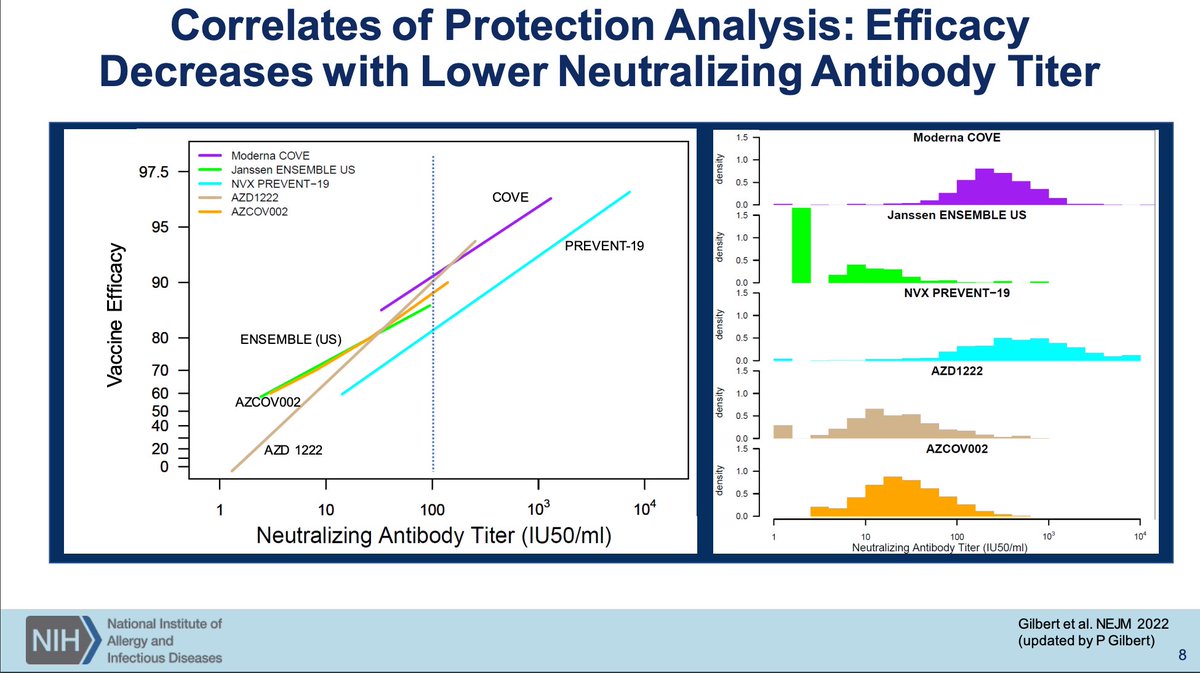
Would also be really nice to have these data for Pfizer and tbh kind of weird we don't. What gives?
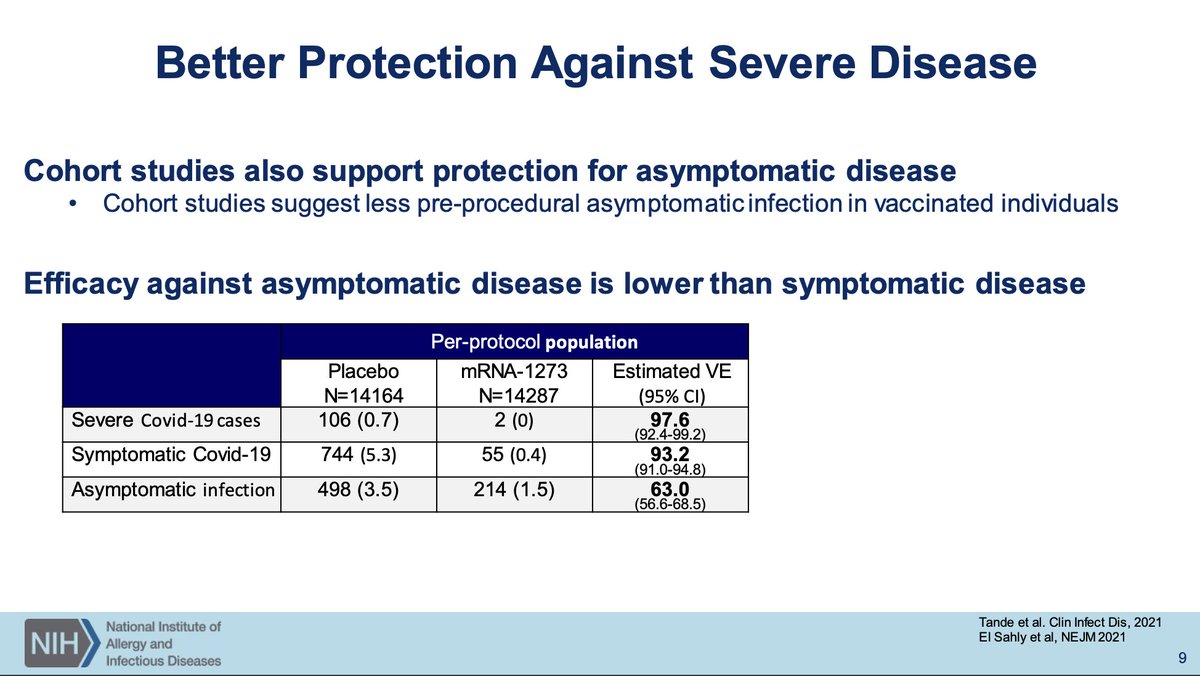
Anywho... effectiveness of vaccines will vary with the specific outcomes as Moderna data shows. Broadly, the worst the disease, the easier it is to prevent, which is good news. Transmission reduction after infection has occurred is... hard. 




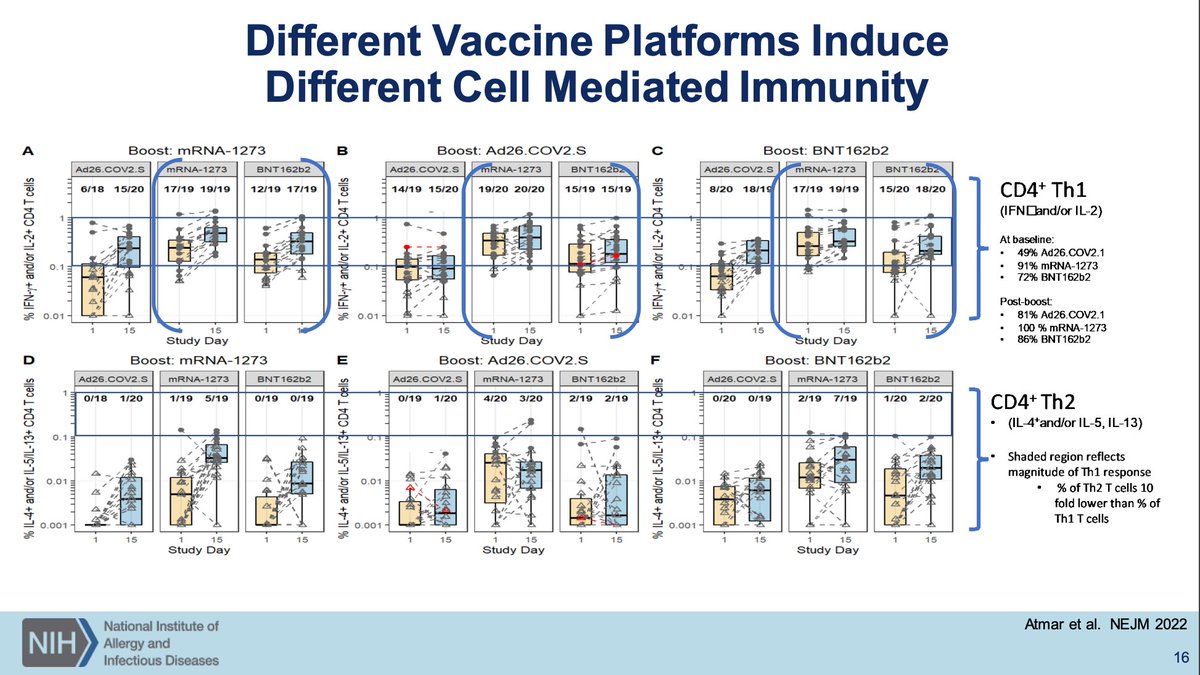
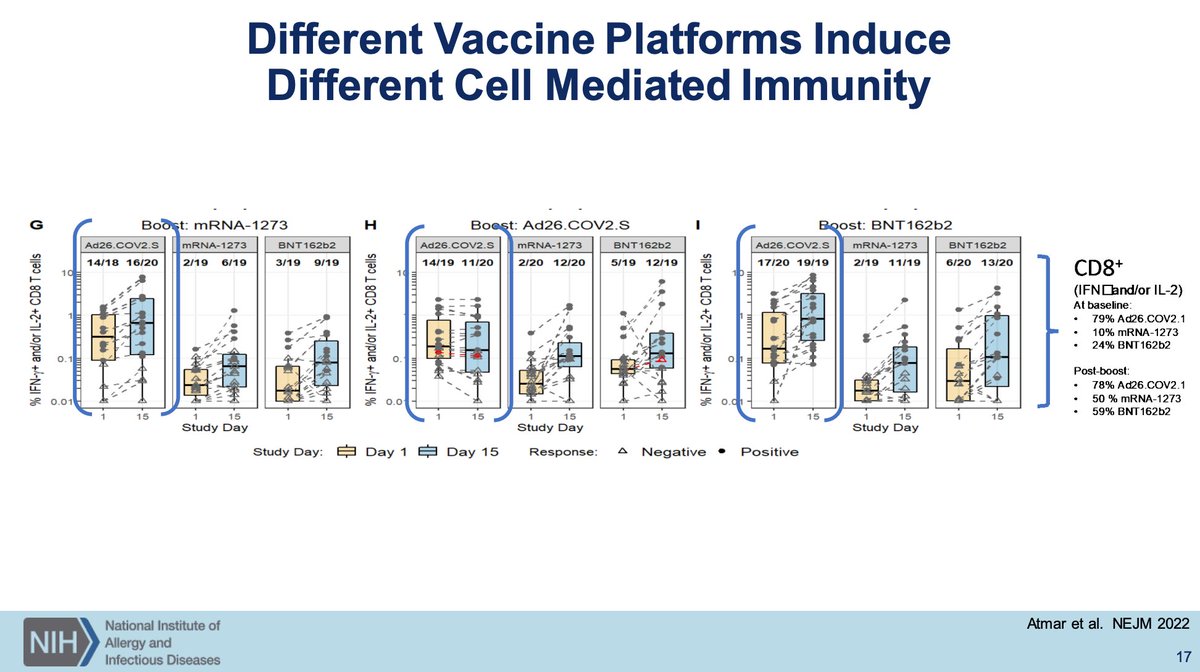
Would love for someone on VRBPAC to ask about mRNA vaccine emphasis on memory B cell vs plasma cell fate but I'm not holding my breath on that one.
Anywho, the goal of a next gen vaccine would be to offer more durable protection against mild disease and be more resistant to emergent subvariants. Easier said than done. 

OWS had strong preclinical basis for which vaccine types to use based on decades of data emphasizing antibodies' importance in minimizing disease severity but we don't have a great idea of what it is for next-gen vaccines. 

Also, neutralizing antibodies cannot explain all of vaccine effectiveness and we urgently need to know what other factors [that we can measure] contribute to protection. The contribution of neutralizing antibodies will vary across vaccine types. 



Nasal IgA titers (and IgG but IgA more so) is strongly predictive of risk of Omicron breakthrough infection. You will not elicit nasal IgA without mucosal administration of a vaccine. Questions exist about what to measure for mucosal vaccine immunogenicity. 

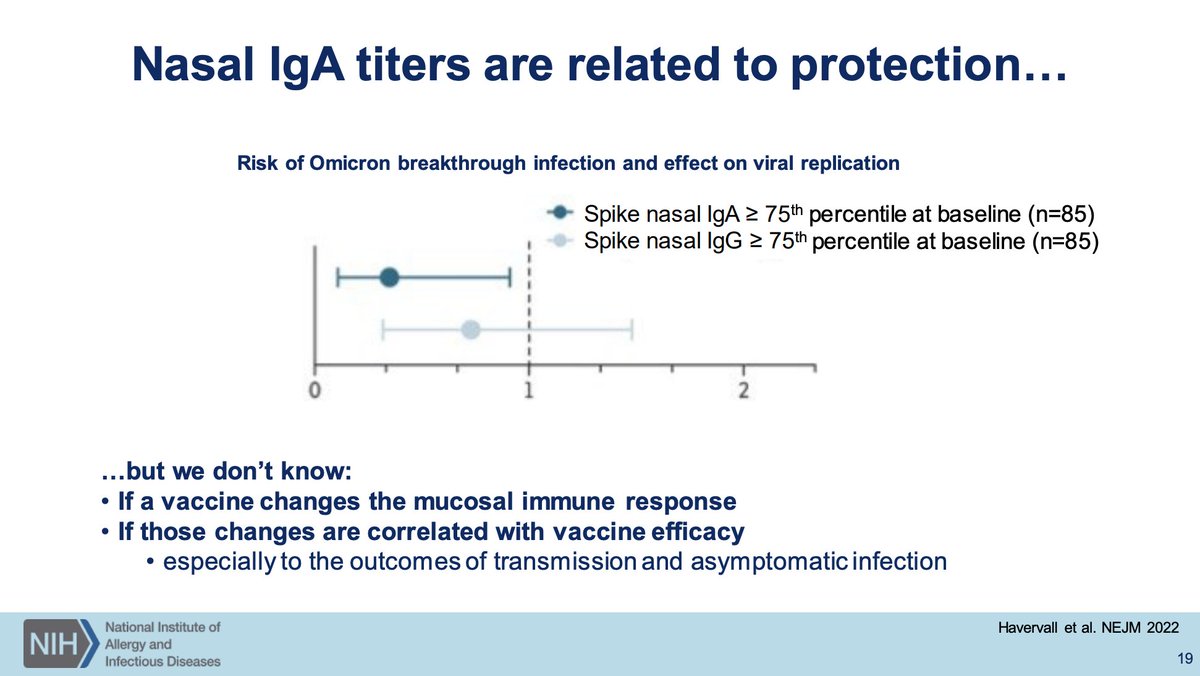
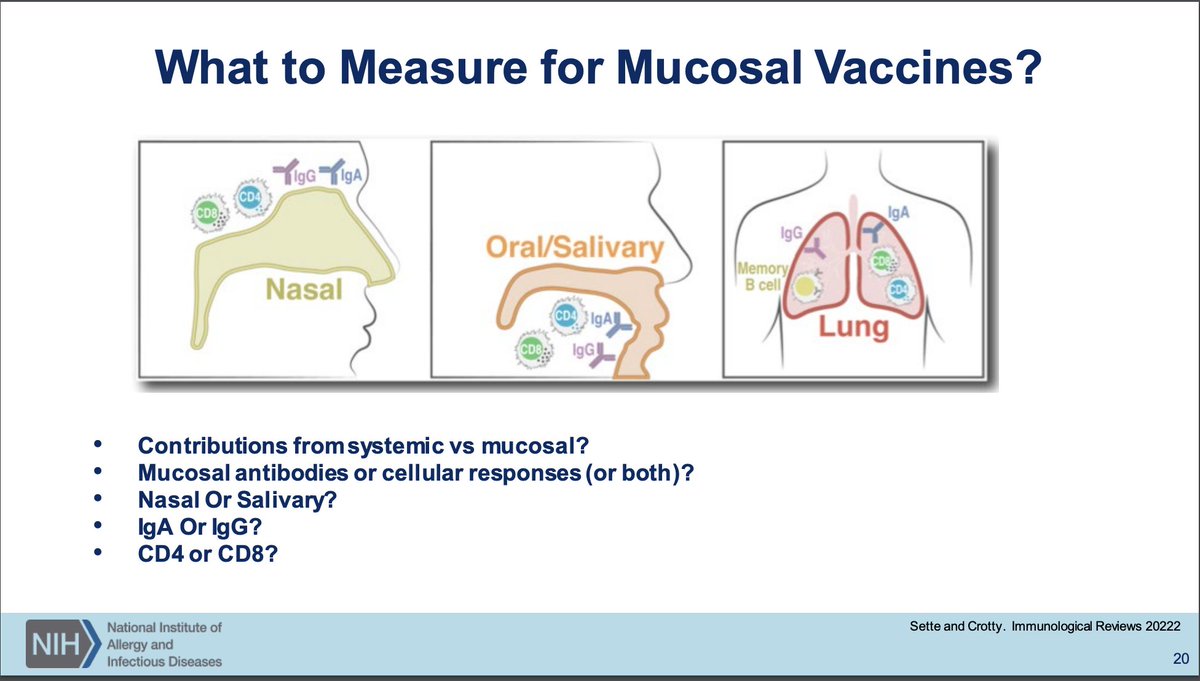
(seriously, why is this a question? Send people for BALs, nasal epithelial fluid washing, serum samples, and lymph node biopsies to longitudinally profile antibody responses by class, subclass, glycoform, CD4 and CD8 T cell response including polyfunctionality, etc.)
Gans- did CoVAIL look at BA.4/5 spike bivalents or only BA.1? We often have to make decisions on the fly of all of the data.
Beigel- No, BA.1; Agree that it would be helpful.
Beigel- No, BA.1; Agree that it would be helpful.
Levy: Deeper approaches to antibody profiling e.g. systems serology and innate immunity would help. How do the correlates of protection change with the subset of the population? Use systems biology to cast unbiased, broad nets, but it is costly. Hope FDA will continue to reflect
Rubin- if you induce immunity via a completely different mechanism, do you expect correlates of immunity will be the same.
Beigel- No. We can measure things but we don't know what they mean until we start doing immune correlates analysis.
Beigel- No. We can measure things but we don't know what they mean until we start doing immune correlates analysis.
Hildreth- Convergent evolution in the spike RBD is obviously occurring. How can we factor this in?
Beigel- we are working on that but we don't know yet where we are going and knowing where we are.
Beigel- we are working on that but we don't know yet where we are going and knowing where we are.
It is now break time.
Some of that was weird. There are literally neutralizing antibody calculators that predict where escape mutations will occur from and managed to successfully predict escape mutations in BA.5 subvariants before they emerged:
Some of that was weird. There are literally neutralizing antibody calculators that predict where escape mutations will occur from and managed to successfully predict escape mutations in BA.5 subvariants before they emerged:
Also oopsies, I read the chart wrong; JJJ has a weird amount of non/hyporesponders, not Novavax:
https://twitter.com/ENirenberg/status/1618641599390691329?s=20&t=v1qGP3dLvBJh-UCJDJSY3w
Now we continue with Moderna's presentation on their bivalent boosters.
Here is the upshot of the Moderna bivalents (also maybe it's just me but this presenter sounds like she is really trying to sell Moderna's vaccine which like... I guess but... anyway). 

Moderna has a lot of clinical trial data on their bivalent vaccines, and Moderna has been good about following the variants and updating their vaccines accordingly (at least until recentishly where they seem to have given up with BQ.1 lol) 


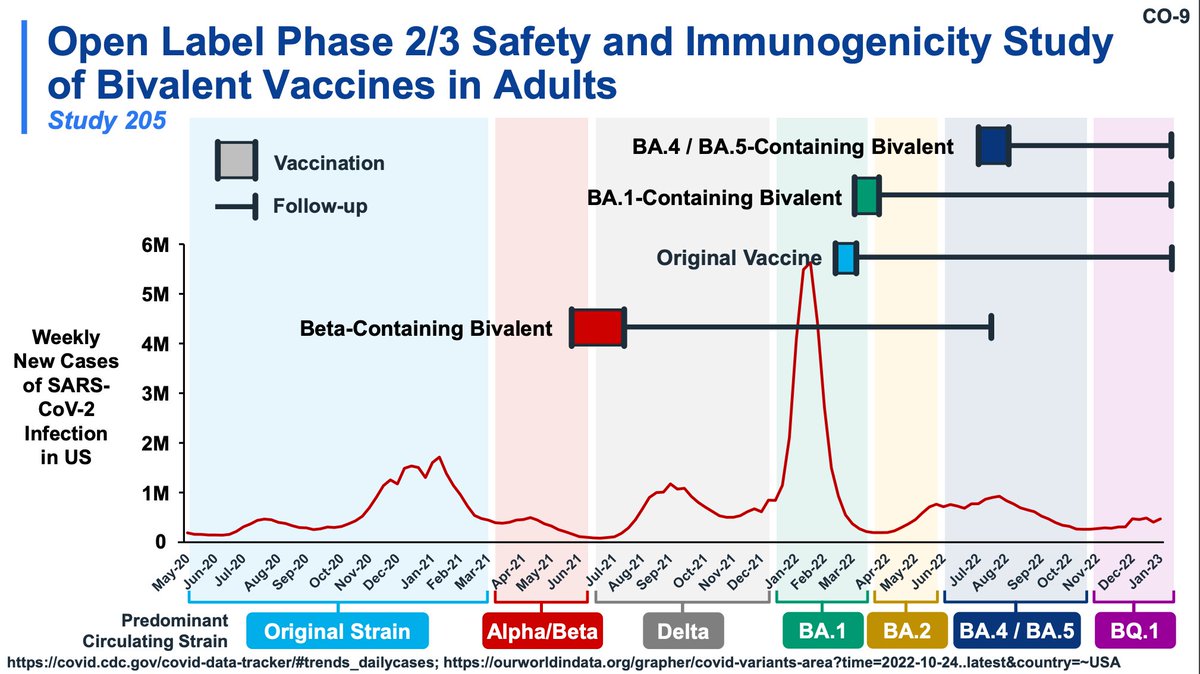
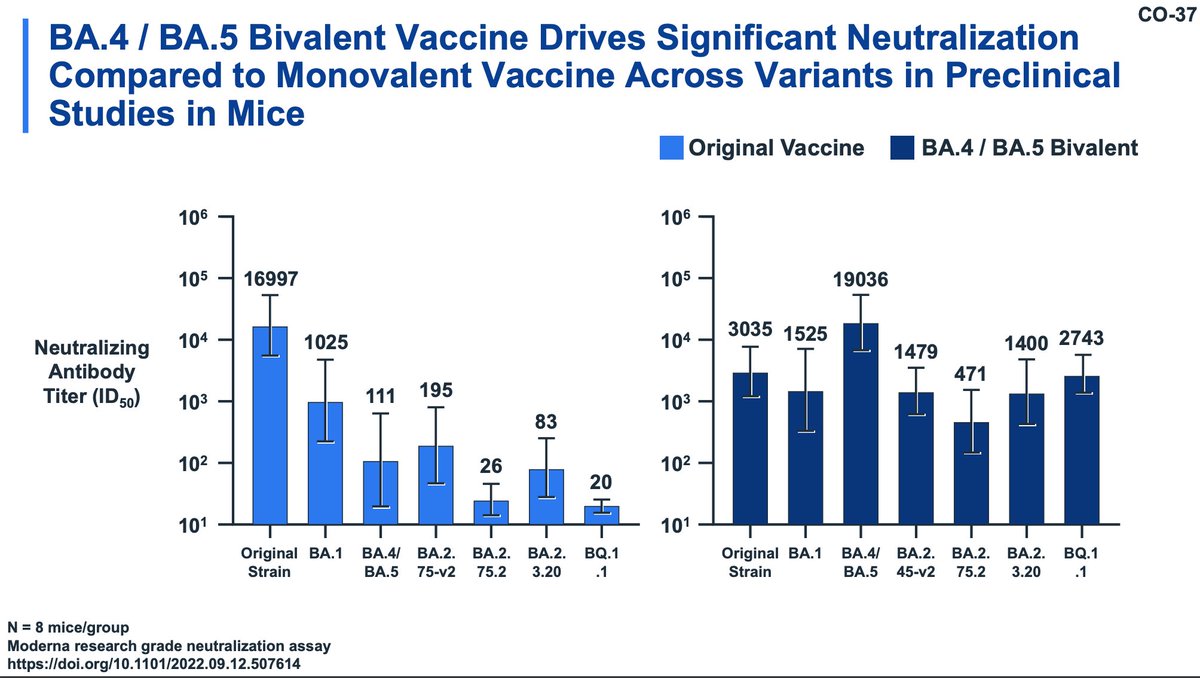
Most data today from Moderna will focus on the BA.4/5 bivalent whereas the meeting in June focused on their BA.1 booster. Note: the control group is not from the same time period as the BA.4/5 vaccinees. Nonetheless, superiority of BA.4/5 is obvious. 

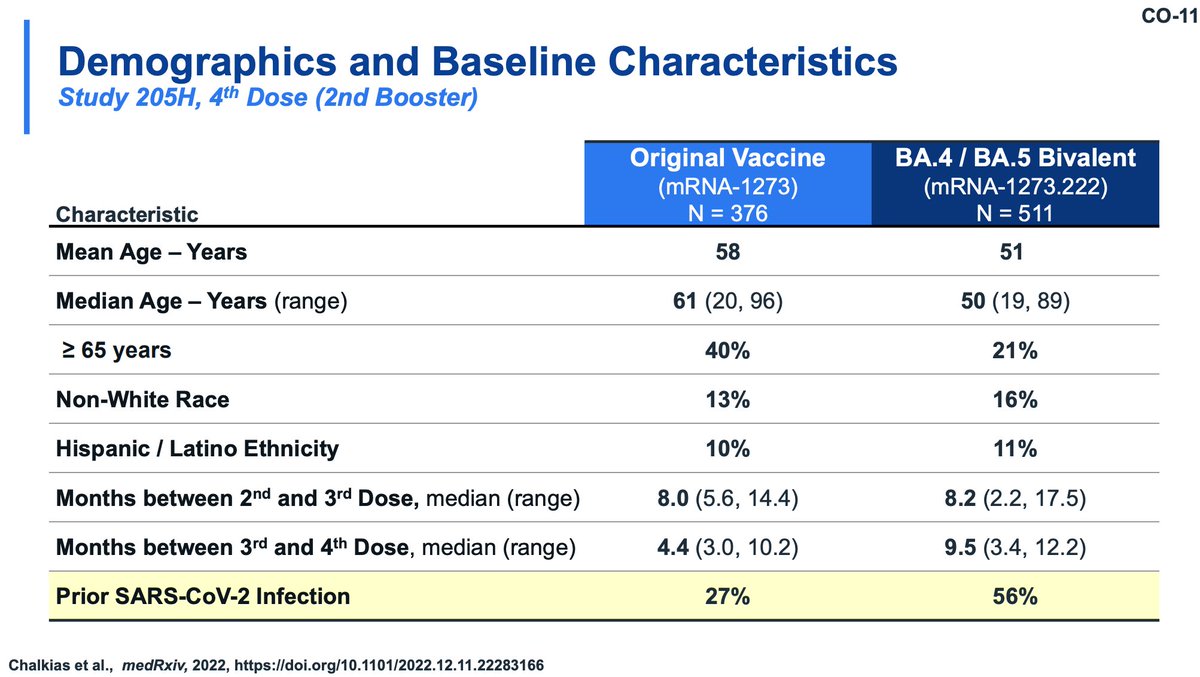

The older adults fortunately also had similar antibody responses to the BA.4/5 boosters compared with younger participants. Neutralizing titers on BQ.1.1, XBB.1, XBB.1.5 are shown 

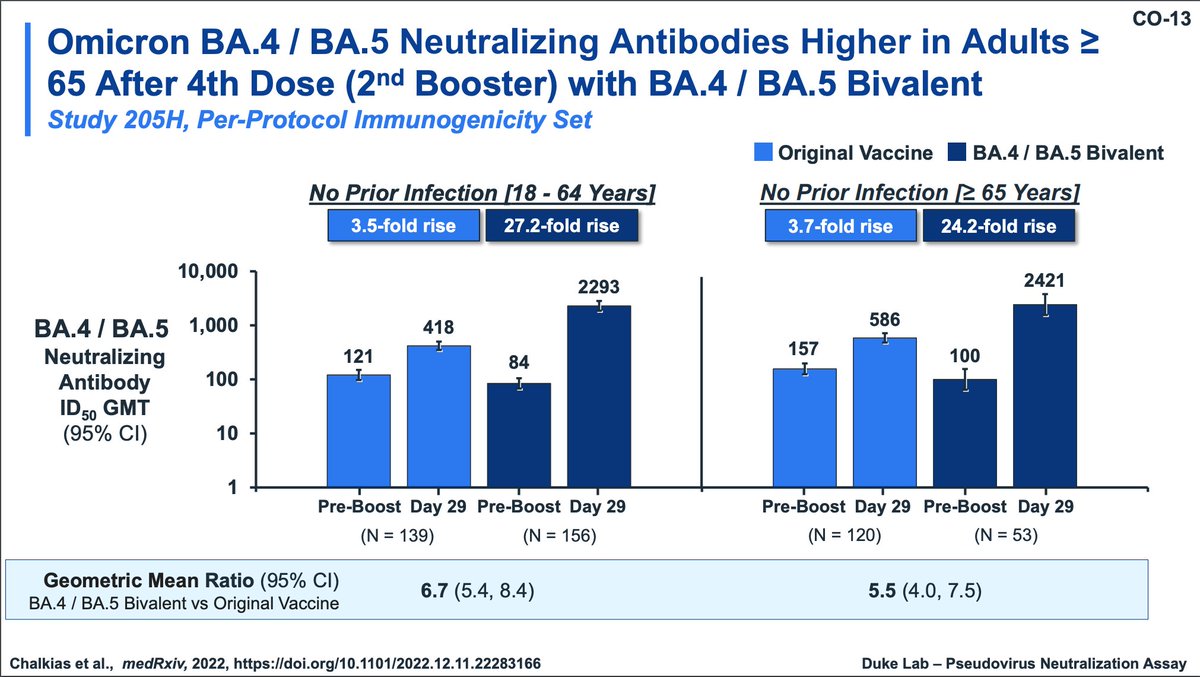
Moderna also did a study of a BA.1 monovalent vaccine but the sample size was smaller. The BA.1 bivalent will be the focus of the data. The study was designed to evaluate immunogenicity and safety, not clinical efficacy. 




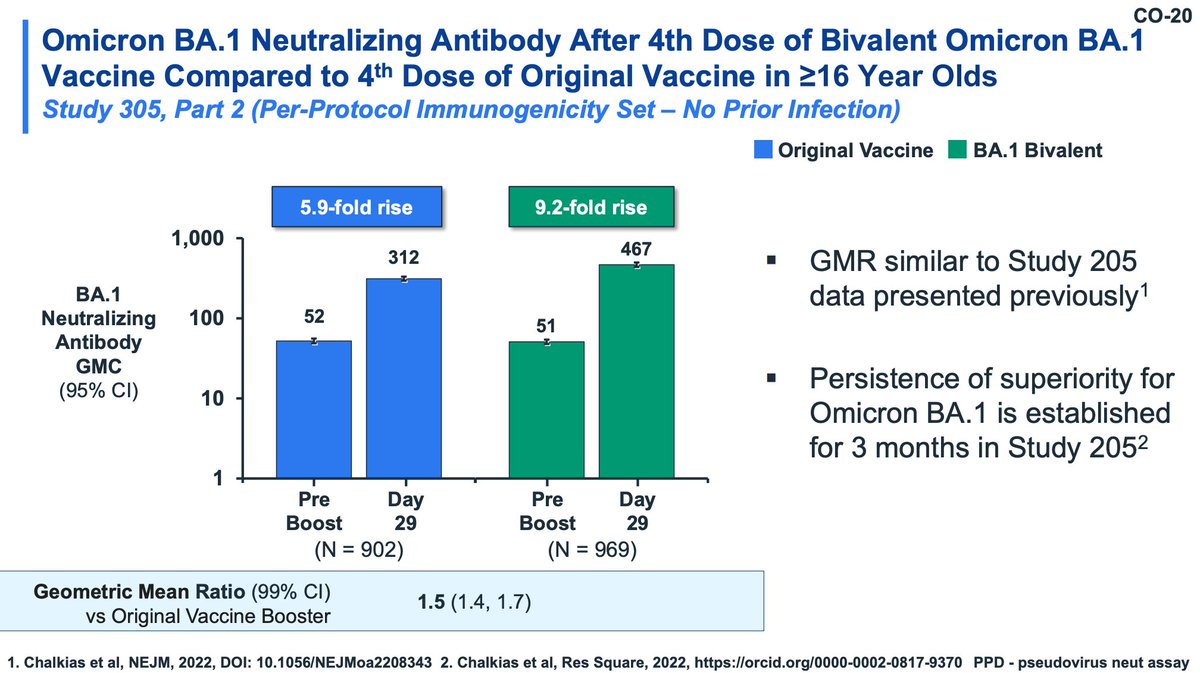
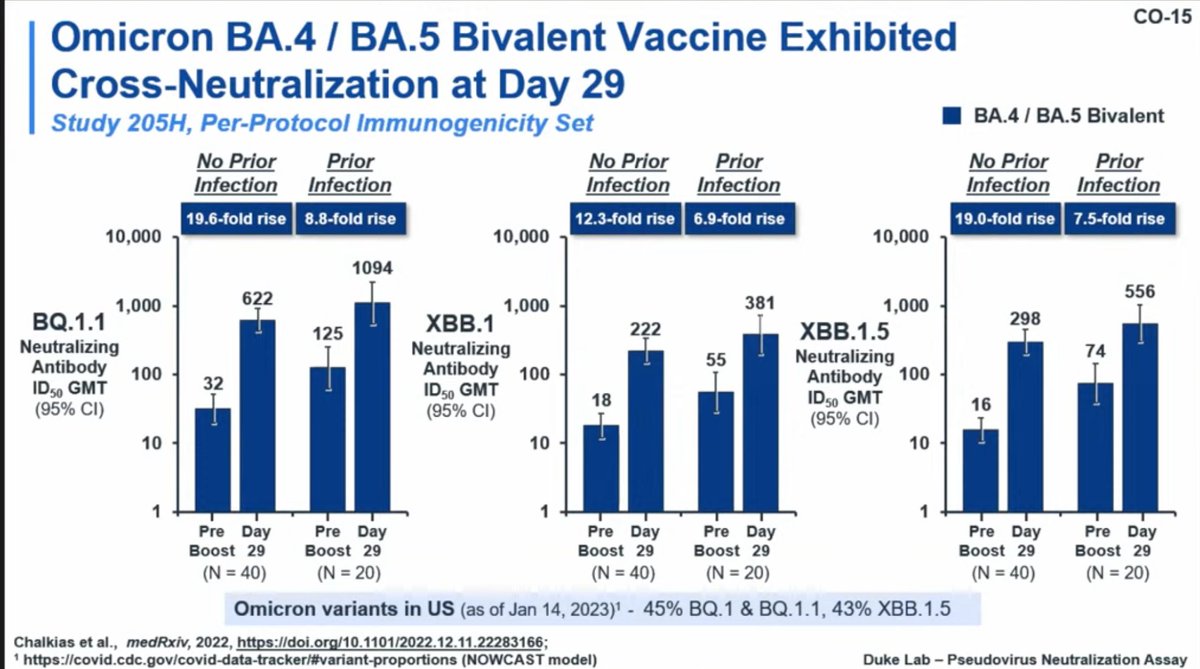
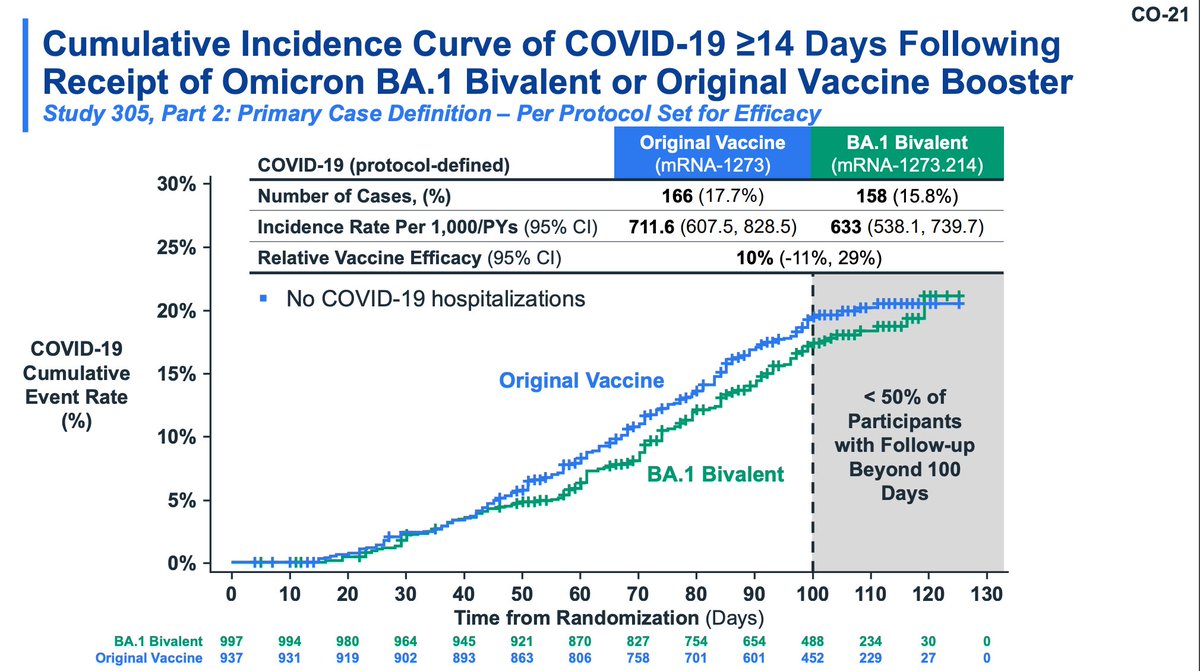
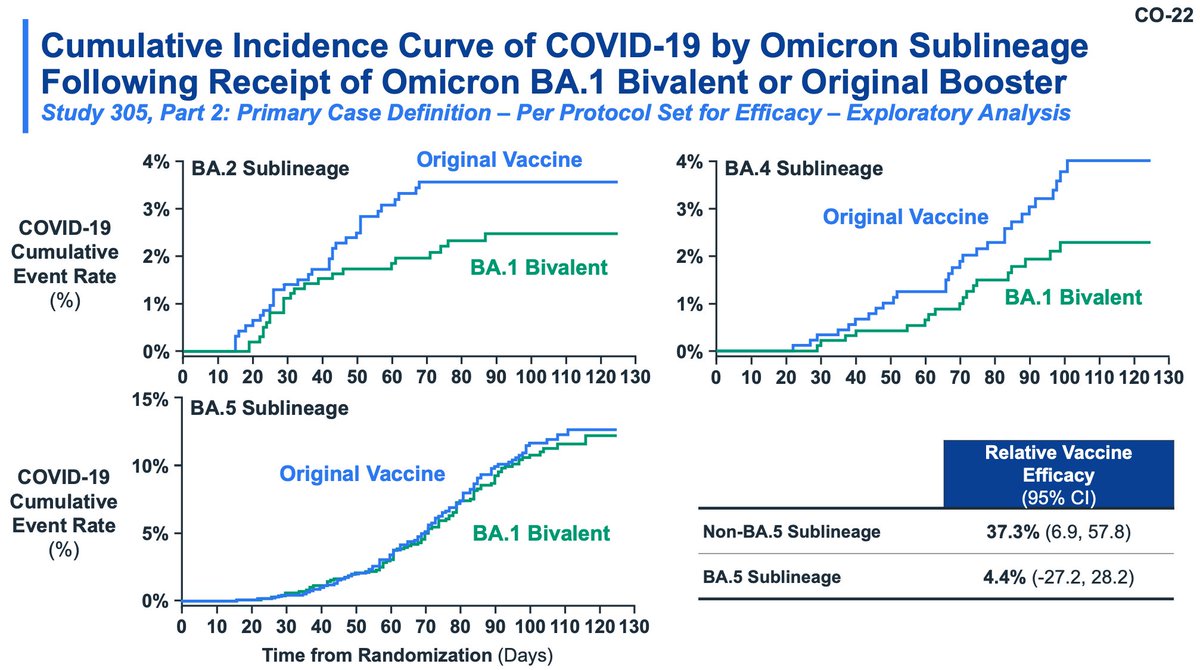
The incidence of COVID-19 is seen to be lower in the BA.1 booster group (slightly) by about day 40, consistent with the time it takes to elicit new antibody responses. The BA.5 strains had similar ability to evade the BA.1 booster. 

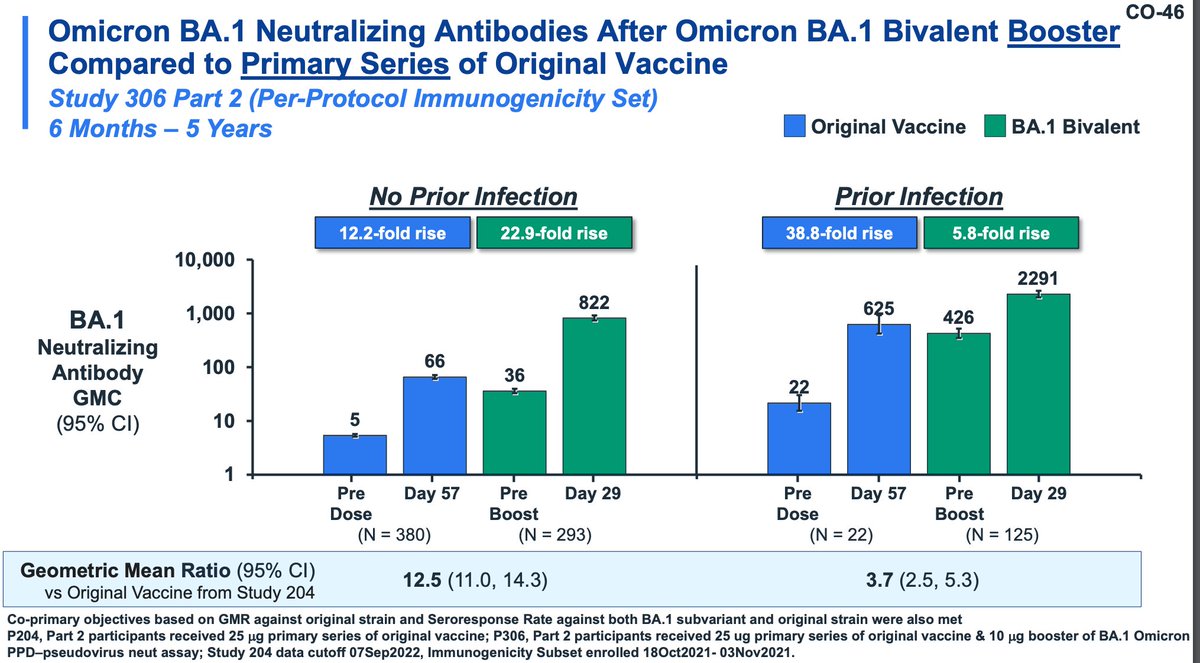
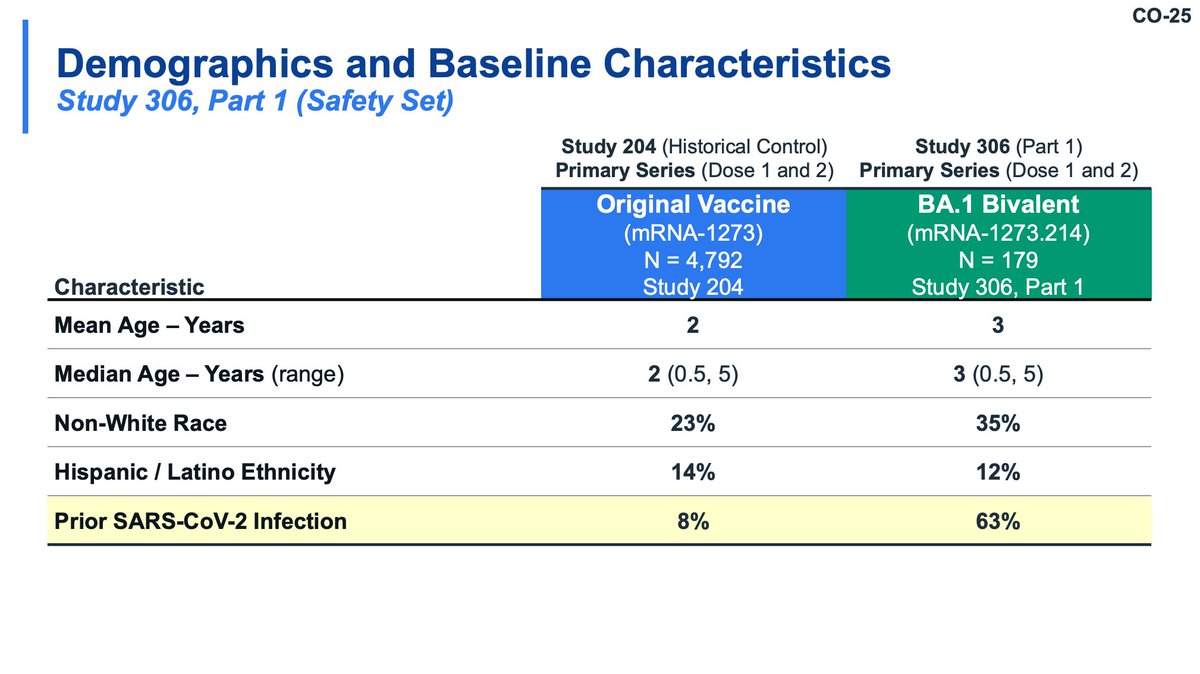
Moderna also evaluated a primary series of bivalent in young children (likely the most relevant group IMO). This study used historical controls from Study 204 (don't love that). Safety was similar to the original vaccine; seropositivity was much higher in bivalent recipients. 




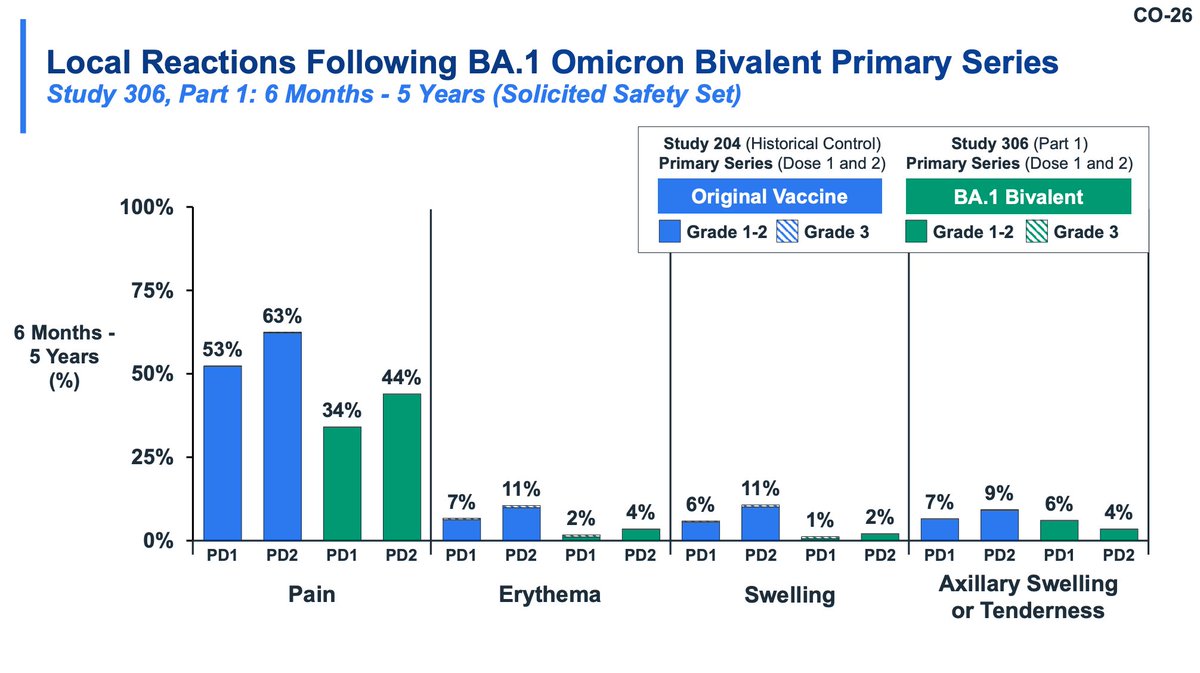
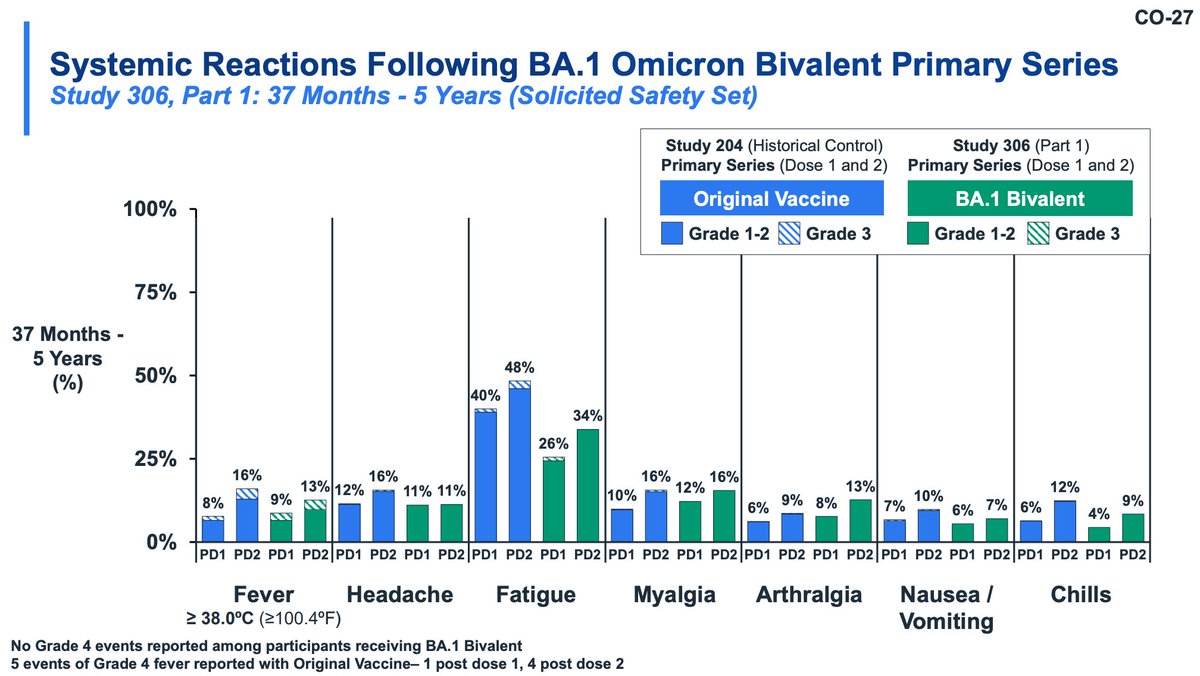
There were no grade 4 adverse events observed (nice!). Immunogenicity included all participants (including seropositive). BA.1 responses are DRASTICALLY higher with BA.1 bivalent primary series compared with original vaccine and titer to ancestral is similar. 
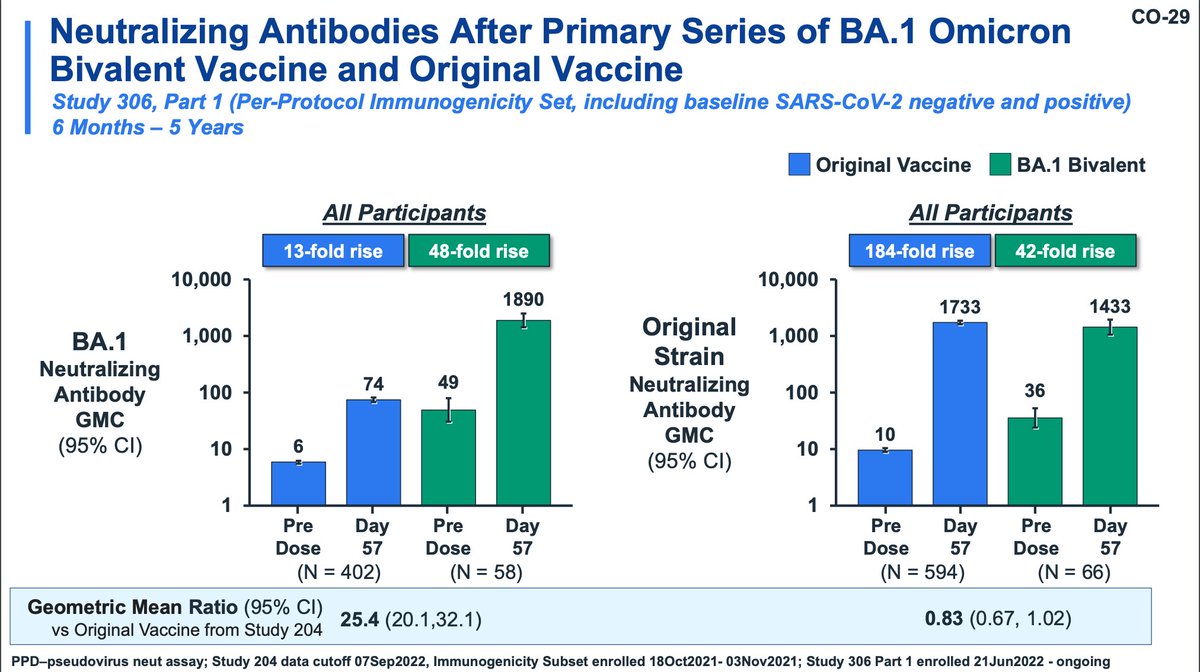
Moderna reports that restricting the responses to seronegative individuals showed similar trends.
(they did not show these results though, so they lose points for that)
Moderna remains very effective against emergent variants severe outcomes (note that this is in the setting of very high population immunity due to widespread infections). Moderna is also reporting that they are working to fill evidence gaps pointed out by regulators. 

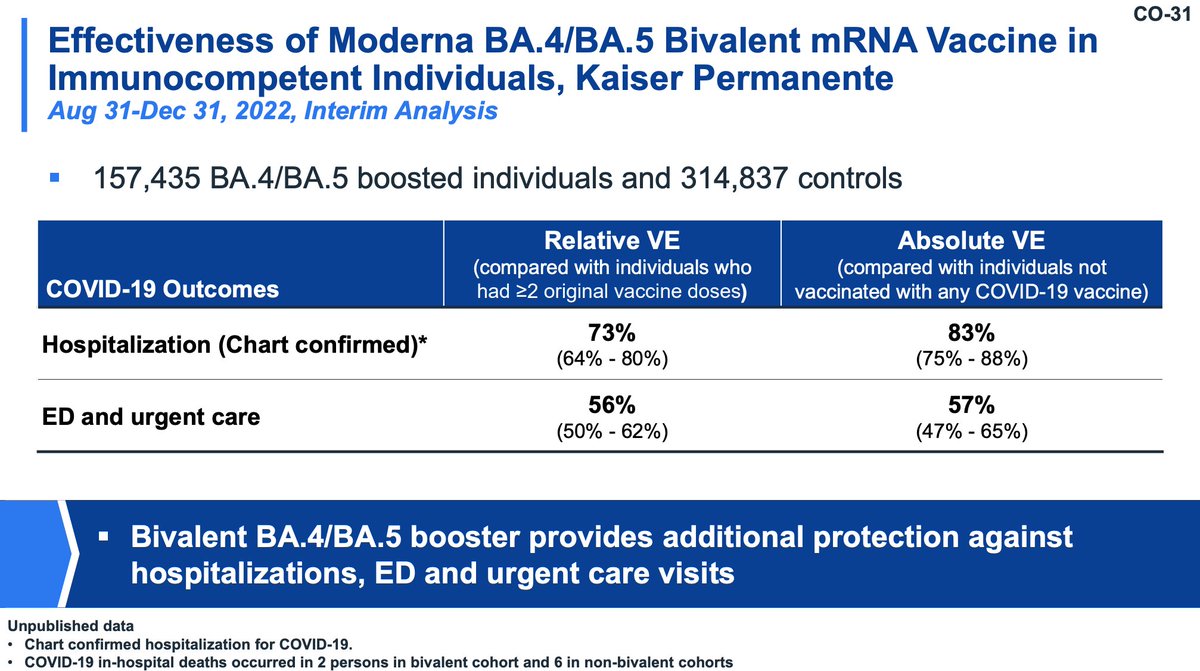

Moderna is also examining preclinical data (animal studies) for updated mRNA vaccines, currently focusing on BQ and XBB, to have expedited delivery of novel vaccines should they be requested. 



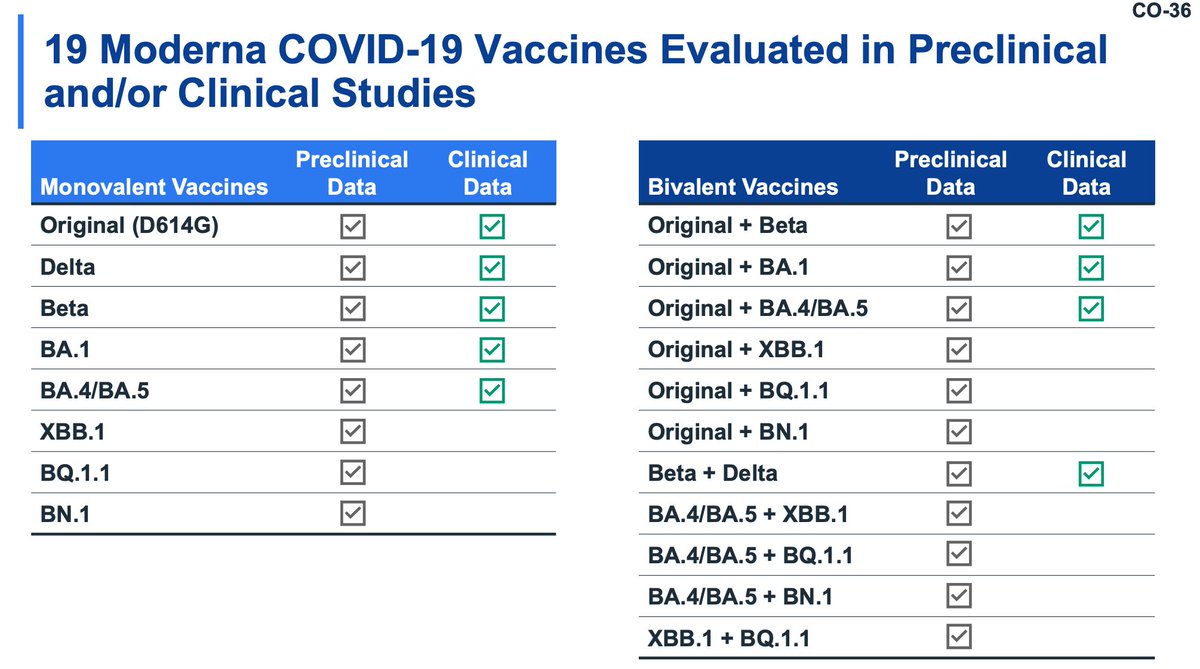
As an aside, an updated bivalent primary series for young children is a complete no-brainer. Hell make it trivalent with a SARS-1 spike.
BA.1 vaccine is favored for non-BA.5 but not for BA.5. The updated vaccines do seem superior (modulo variant divergence) to monovalent ancestral vaccines. 



Perlman- if BA.4 and BA.5 have the same spike, why was there such a disparity in the effectiveness?
Das- BA.5 wave was later, and variant showed higher fitness
(note from me: interestingly, BA.4 has superior subversion of innate immunity to BA.5 but BA.5 took over)
Das- BA.5 wave was later, and variant showed higher fitness
(note from me: interestingly, BA.4 has superior subversion of innate immunity to BA.5 but BA.5 took over)
Levy- the bivalent seemed to have slightly less reactogenicity- why? And will they do nested T cell assays?
Das- We do see slightly reduced reactogenicity- not [statistically significant] but we see it. We aren't sure why but it is consistent. Re CoP, Moderna...
Das- We do see slightly reduced reactogenicity- not [statistically significant] but we see it. We aren't sure why but it is consistent. Re CoP, Moderna...
did the CoVE study to help establish correlates of protection and working to establish variant-specific correlates of protection. We are limited by the samples taken in the studies. Neutralizing antibodies account for most CoP work. Will continue to look at T cell responses.
Offit- UK study managed to contemporaneously compare BA.1 booster to monovalent which was a meaningful strength. There was also a monovalent BA.1- what is the data there. Do we still need ancestral mRNA?
Das- the slide is shown here. GMTs and GMFRs are similar but...
Das- the slide is shown here. GMTs and GMFRs are similar but...
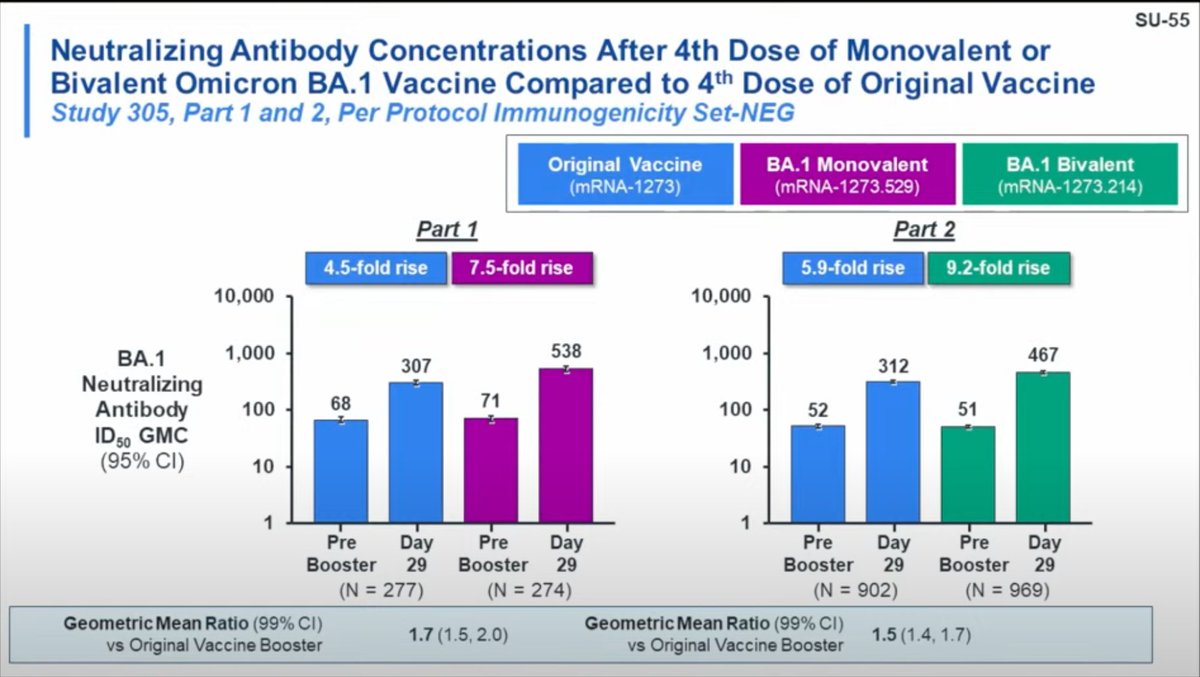
some slight benefit is seen with the bivalent.
Moderna has shown this result before and it's honestly weird because Pfizer doesn't have the same phenomenon.
Moderna has shown this result before and it's honestly weird because Pfizer doesn't have the same phenomenon.
Pfizer emphasizes that preclinical data have reliably predicted the responses in humans; bivalent vaccines may confer broader neutralizing response across VOC vs. monovalent per Pfizer
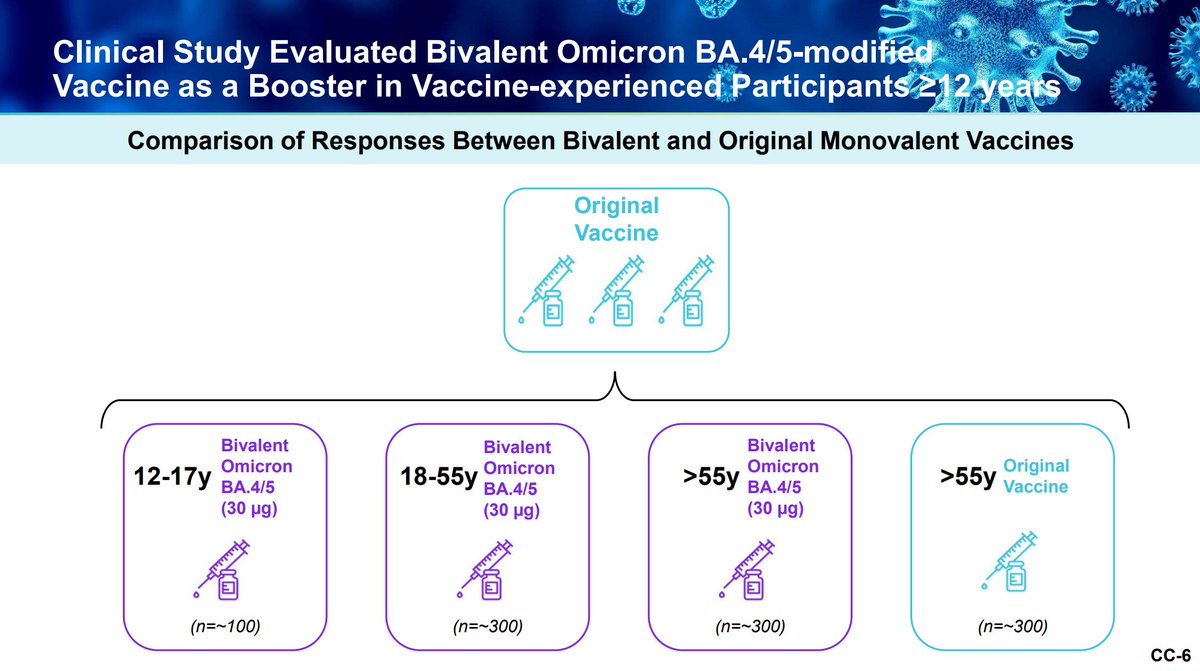
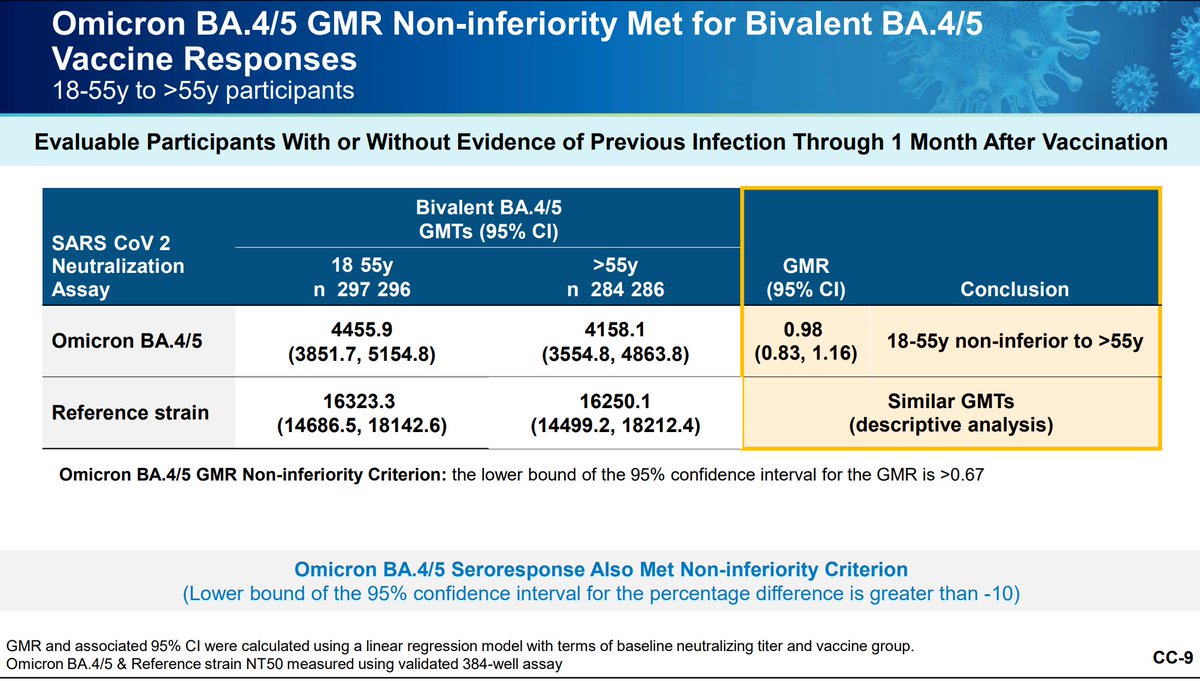
There is a lower incidence of prior SARS-CoV-2 infections in the > 55 year old bivalents.
All groups had similar neutralizing titers pre-boost and they had similar seroresponse rates regardless of age or infection history. 



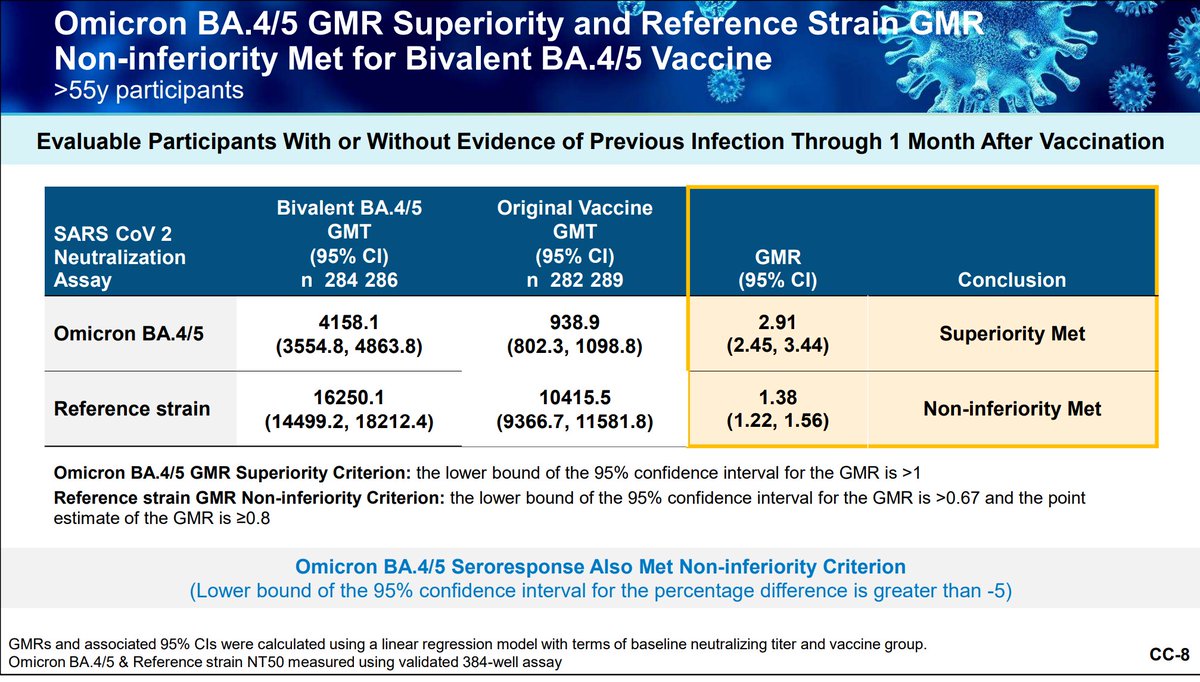
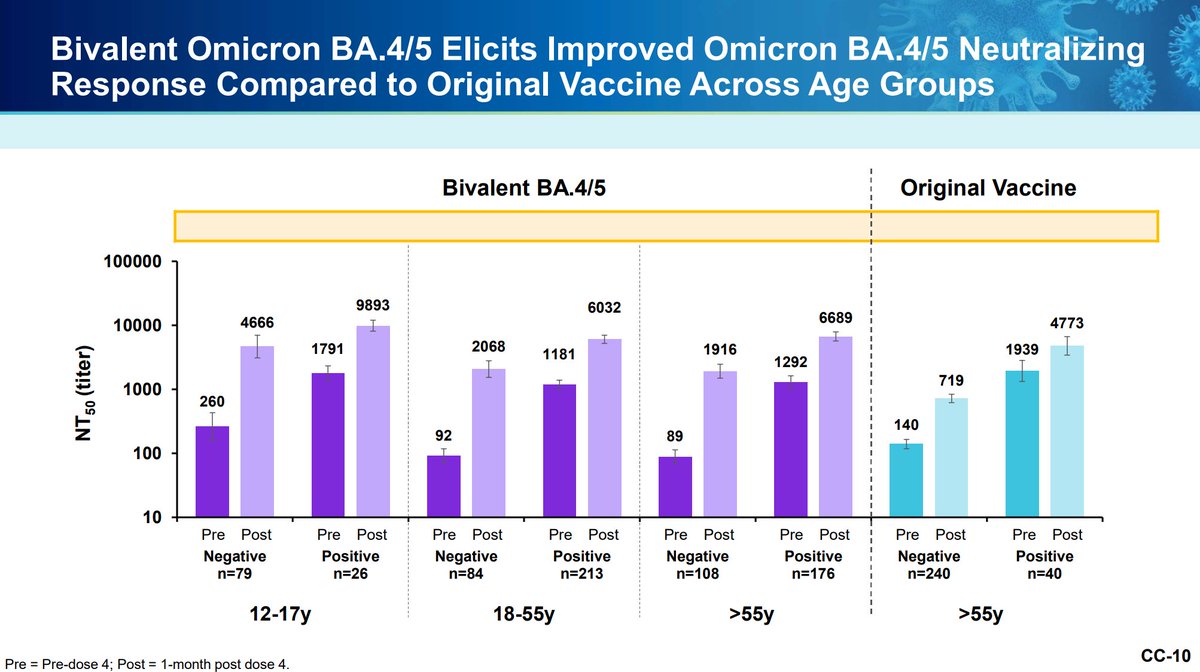
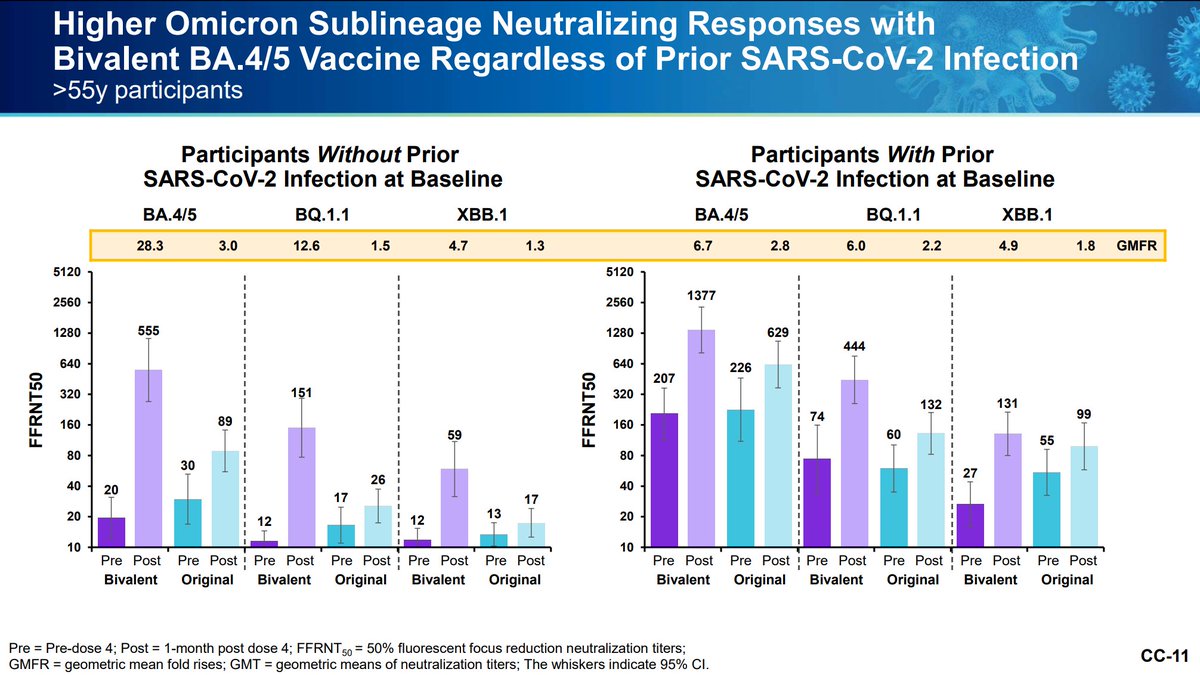
Pfizer is also working on studying its bivalent vaccine as a primary series in those younger than 12. The bivalent booster appears superior with similar reactogenicity. 




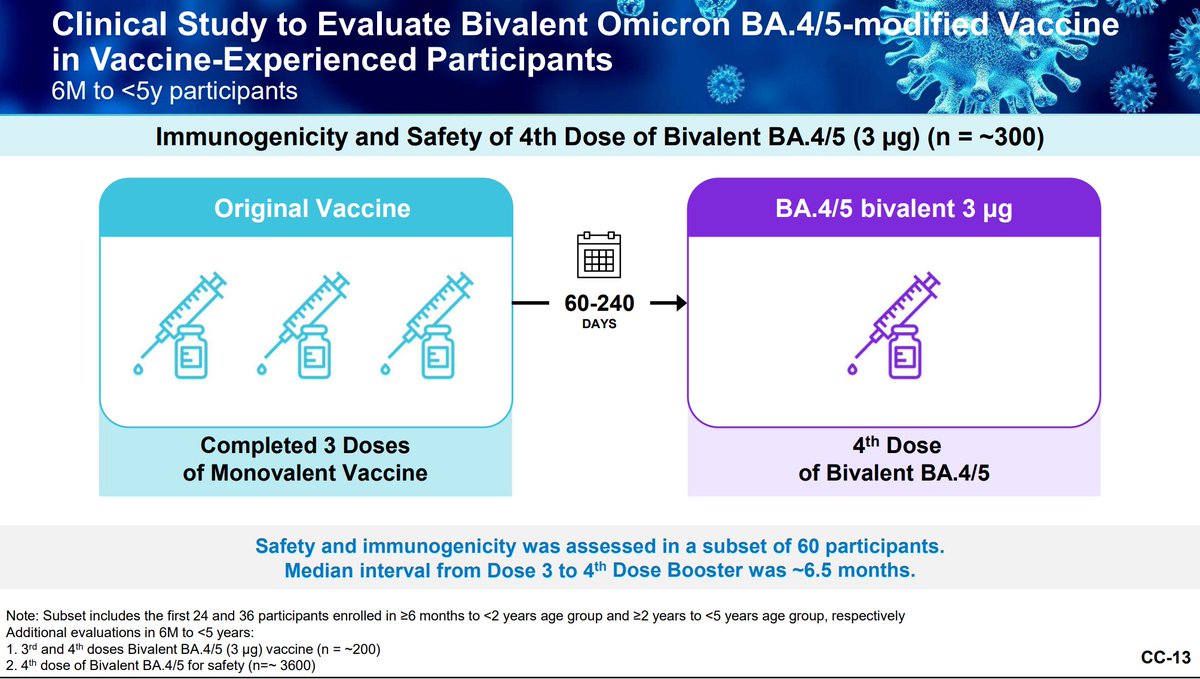
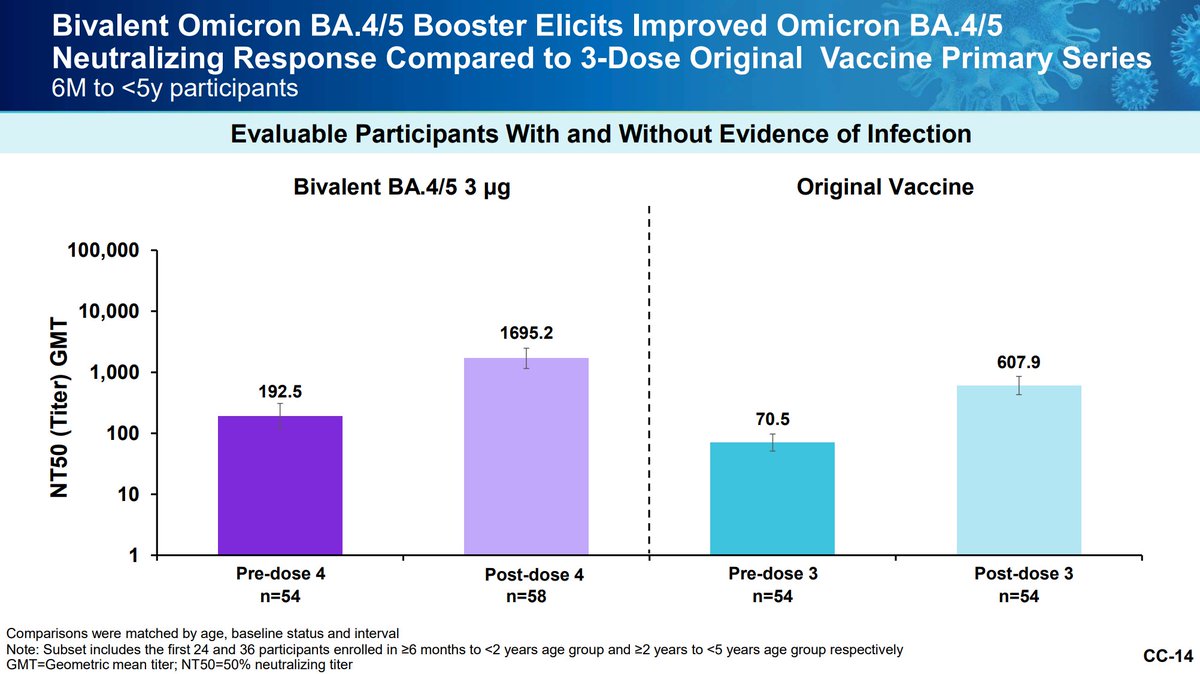
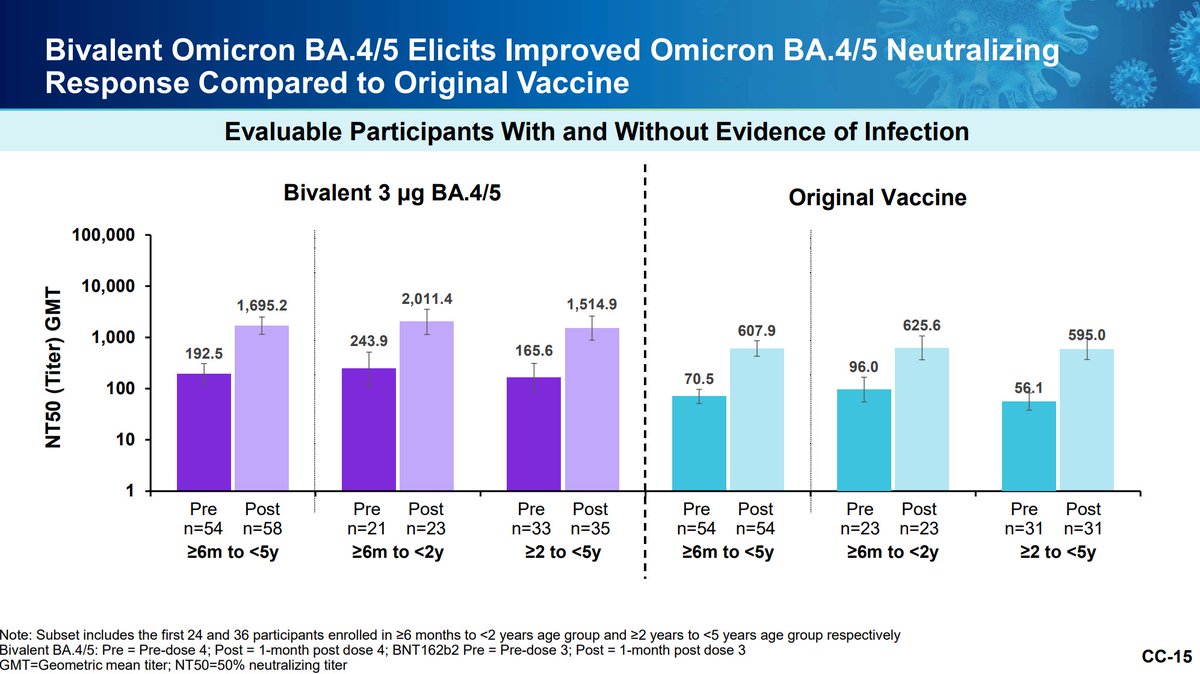
Pfizer wishes to minimize potential for waning immunity and maximize protection from further antigenic drift. Ongoing evidence collection with antigenic distance analysis is critical, as well as imprinting questions. A library of variant specific vaccines could...
be created at risk and deployed as dictated by the population. The influenza model should be amenable per Pfizer to this approach with no clinical studies to support licensure. Off-cycle updates could also be done if necessary (i.e. out-of-season surge). 

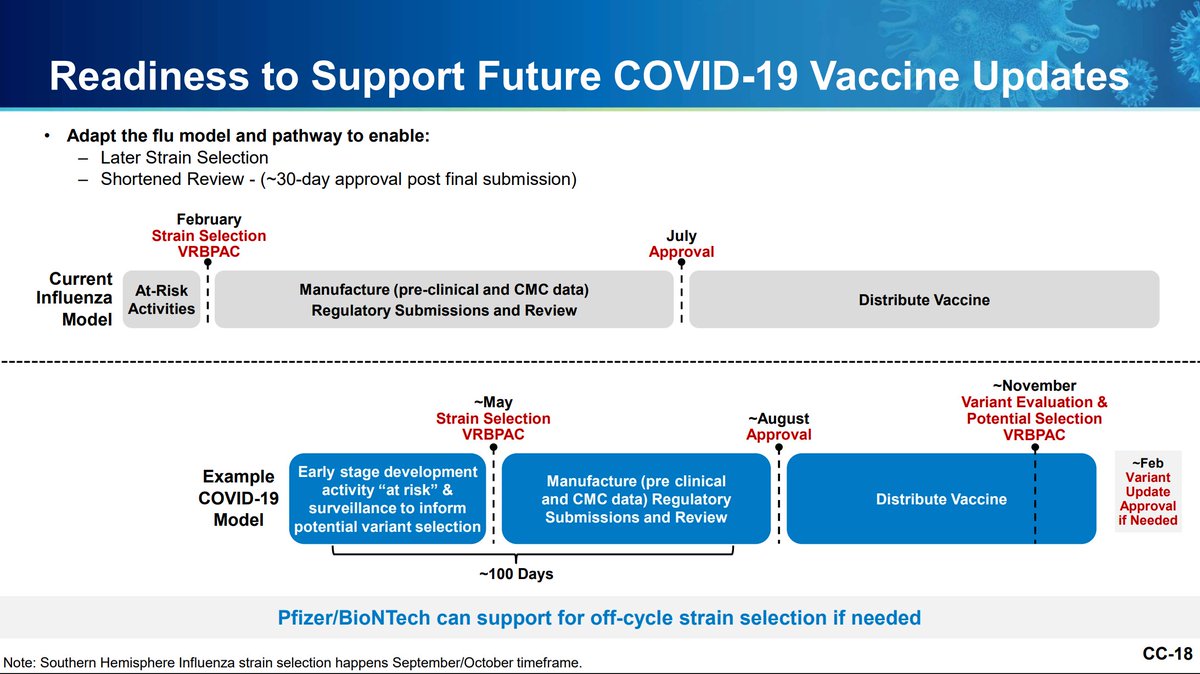

Pfizer urges a defined strain selection process and regulatory pathway for vaccine updates to optimize protection against COVID-19.
Sawyer- Bivalent booster study only had 20 children under 2; how many children will you have in future studies?
Pfizer- that was based on a limited immunogenicity subset; primary series phase 1 study will have 90 participants in the 2-5 and under 2 each. For the booster, 300.
Pfizer- that was based on a limited immunogenicity subset; primary series phase 1 study will have 90 participants in the 2-5 and under 2 each. For the booster, 300.
Gans- we don't actually see evidence of imprinting (she means original antigenic sin) and I don't actually see evidence of this in the data (THANK YOU SOMEONE FINALLY SAID IT). When we're considering new vaccines, are dosing considerations undertaken again i.e. 3 doses for kids?
Pfizer- we do know from the totality of the data, when you have an omicron component in the vaccine you get improved responses against Omi and its sublineages. We know the history of influenza where there is more extensive imprinting (this point is complicated but I don't...
completely agree with her comments). 2 vs 3 doses will be examined and it's a moving target as the variant landscape changes. We also want to understand whether the vaccines perform the same way including 3 mcg dose vs higher doses.
Gellin- question for production- Pfizer said 100 days to go from update to clinic. If we talk about flu model 2,3,4-valent, does that affect production timeline?
Pfizer- timeline would be similar regardless of component number
Pfizer- timeline would be similar regardless of component number
McInnes- where are the data on protection from disease? What about platforms beyond mRNA?
Perlman- Novavax will discuss protein vaccines
Pfizer- Unaware of head to head efficacy/effectiveness for monovalent vs bivalent. We have RCT for monovalent Omi vs bivalent. Both...
Perlman- Novavax will discuss protein vaccines
Pfizer- Unaware of head to head efficacy/effectiveness for monovalent vs bivalent. We have RCT for monovalent Omi vs bivalent. Both...
superior to original booster, and the monovalent had a trend towards higher responses. We are unsure whether that is meaningful.
It is Novavax's turn. They report broad and long-lived immune responses irrespective of the variant of the spike protein used. Escape is similar for XBB and BQ for mRNA vaccines.
Novavax is a trimeric spike protein presented as a nanoparticle with a saponin-based Matrix M adjuvant. Novavax had high efficacy in clinical trials (pre-Omicron; unusually low 2-dose VE to beta). 


These are the titers with boosting to spike: note, these are NOT neutralizing titers. They are any spike binding titers. 

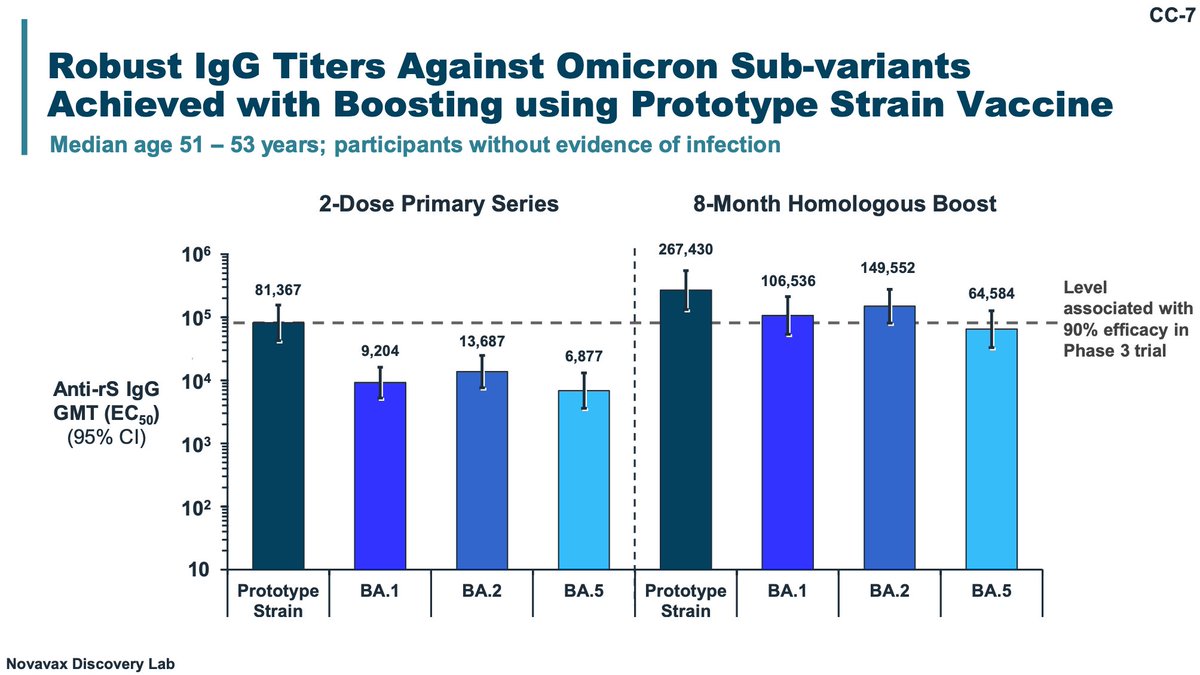
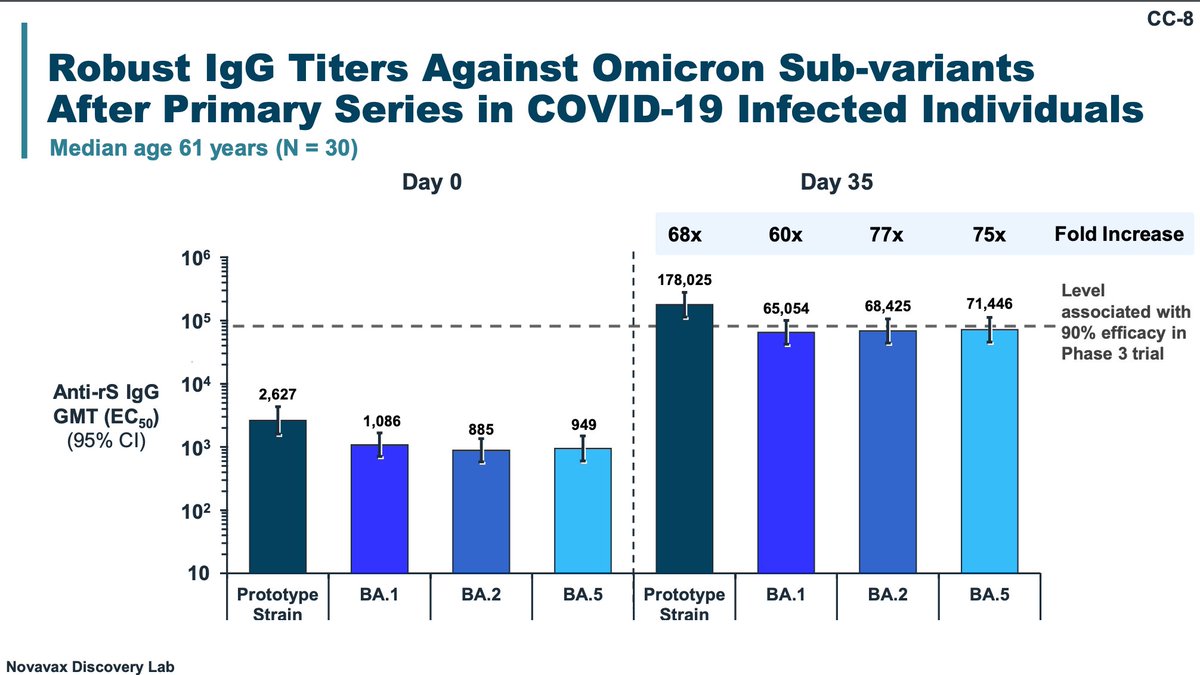
These are neutralizing titers and antigenic cartography with the boosters (unsure why mRNA manufacturers didn't show antigenic cartography plots- you would see the same trends). The efficacy values for CoP presented are dubious because different variant kinetics. 


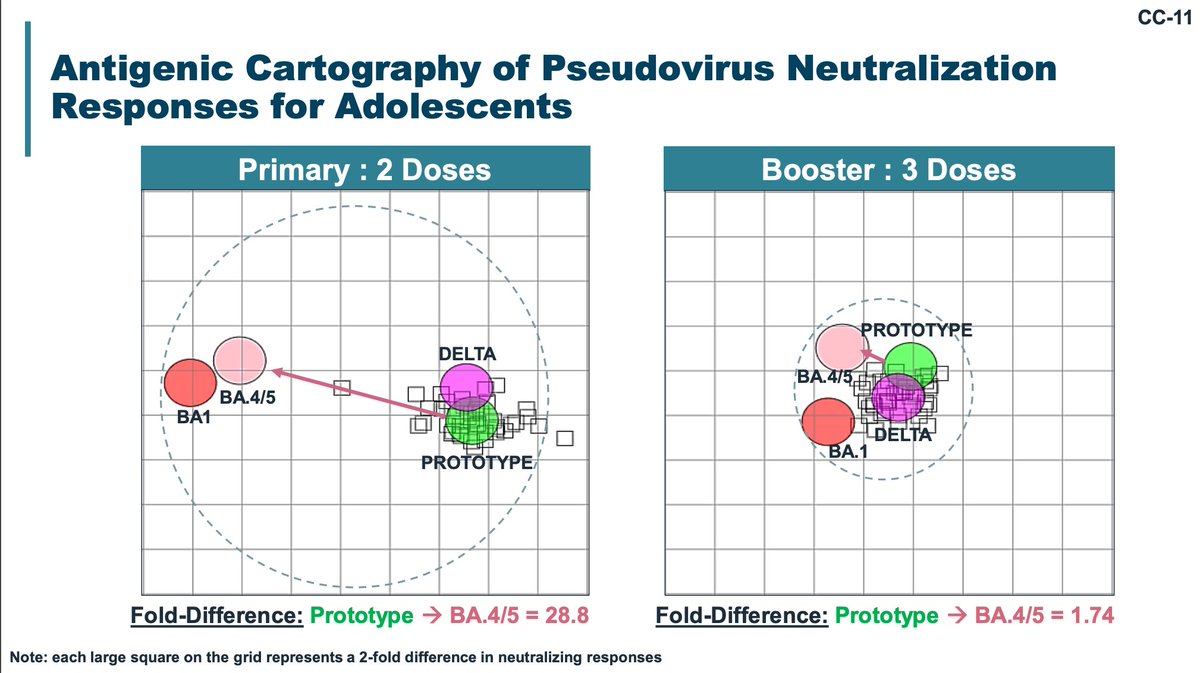
Novavax performed a study of a bivalent and BA.1 spike vaccine in Australia; similar incidences of prior infection irrespective of group and composition is similar. All boosters were broadly similar in their immunogenicity in their convenience immunogenicity sample. 




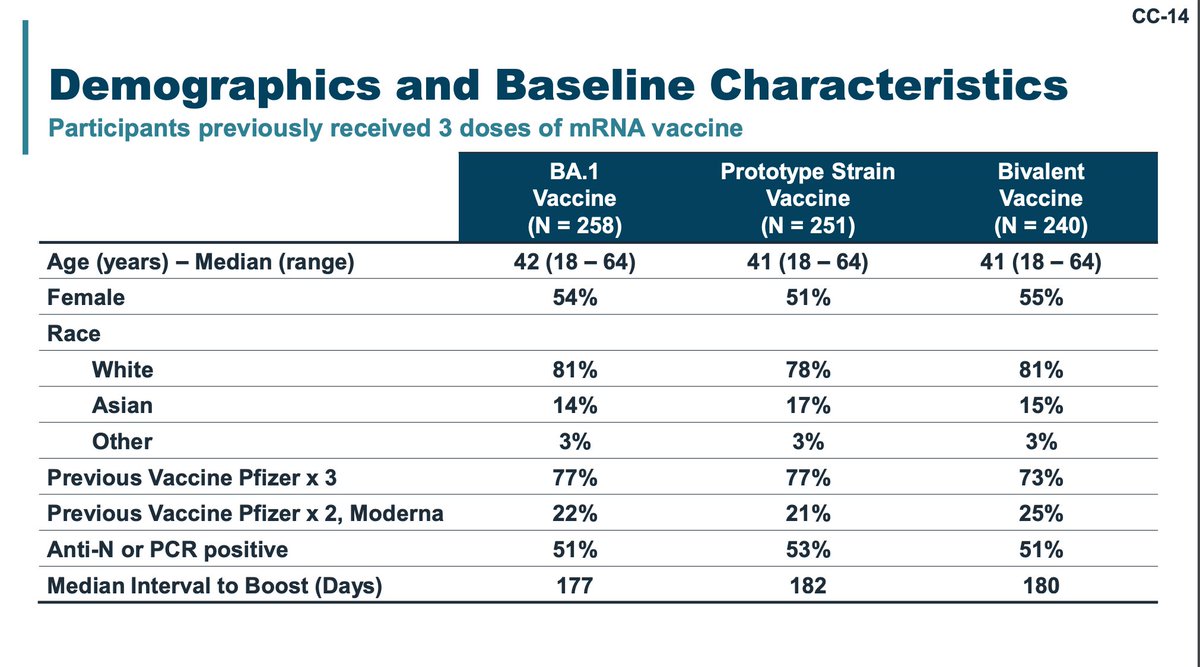
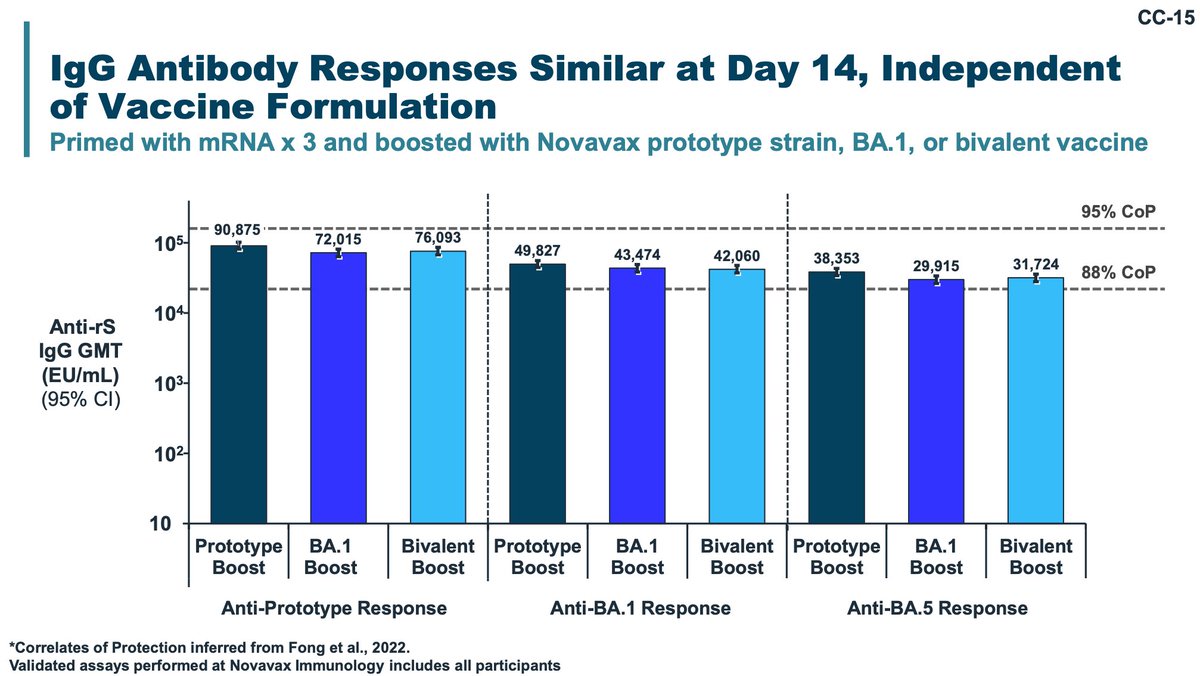
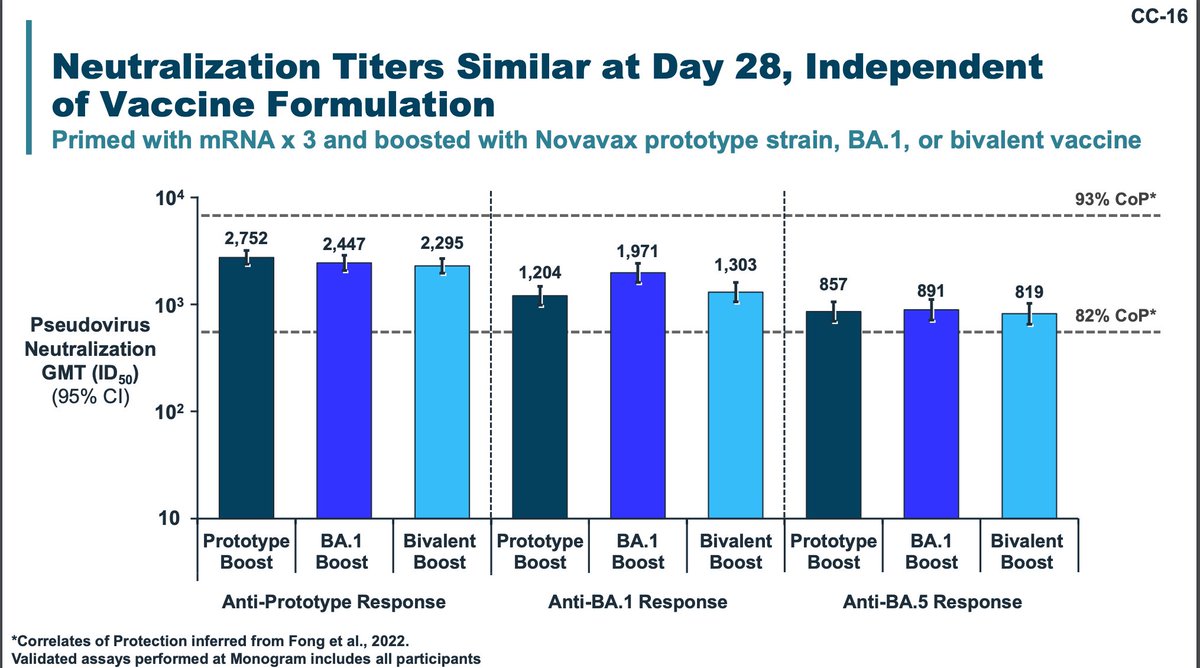
Novavax also performed a lot-to-lot consistency study examining booster responses in different vaccine priming regimens. The demographics are poorly matched. Again, correlates of protection values here are dubious. 



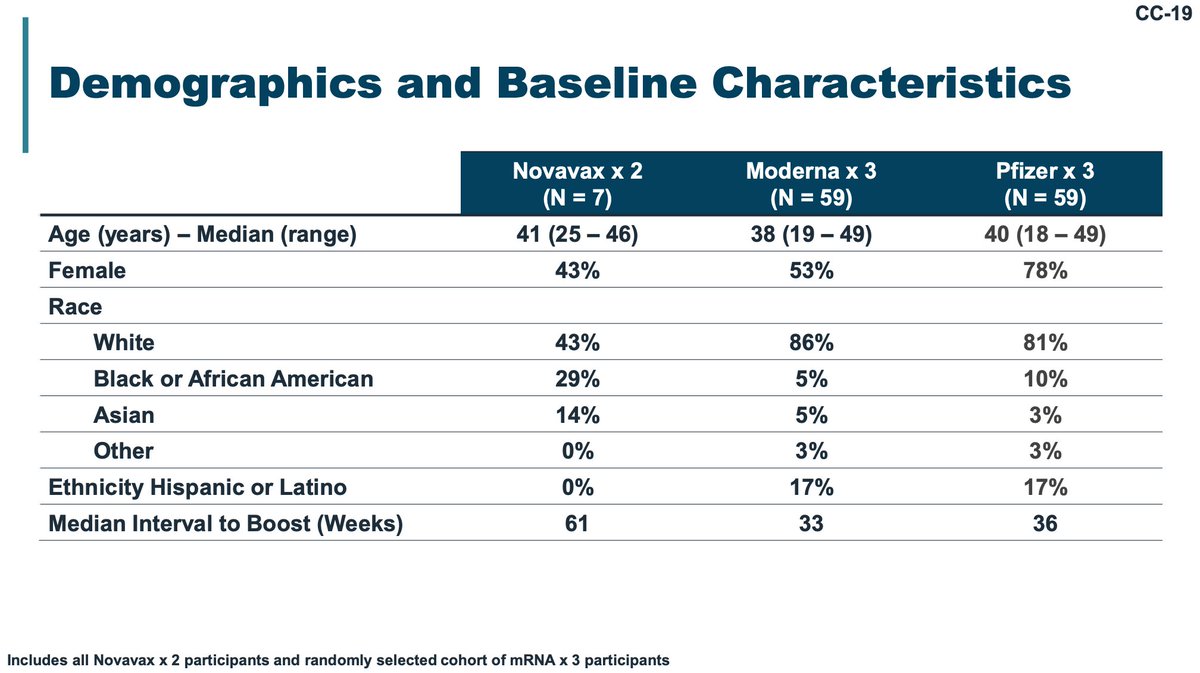
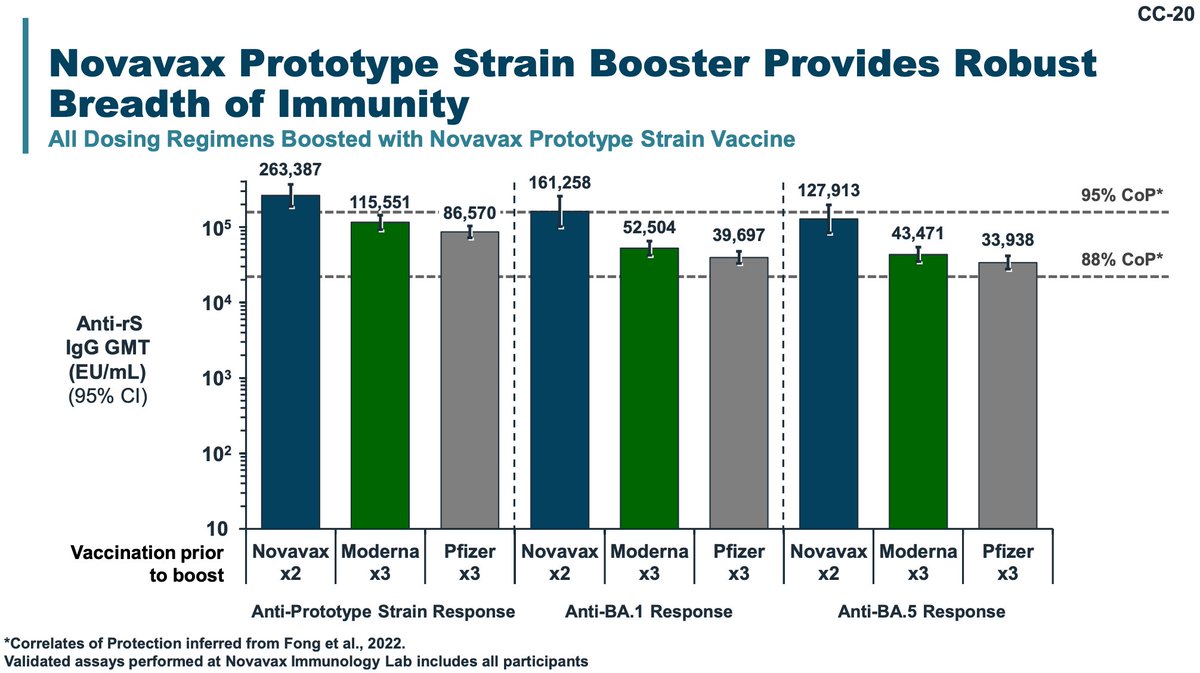
A study of neutralization of Novavax vaccinees and mRNA vaccinees was done- it doesn't show any clear advantage to mRNA vaccines for novel variants. 
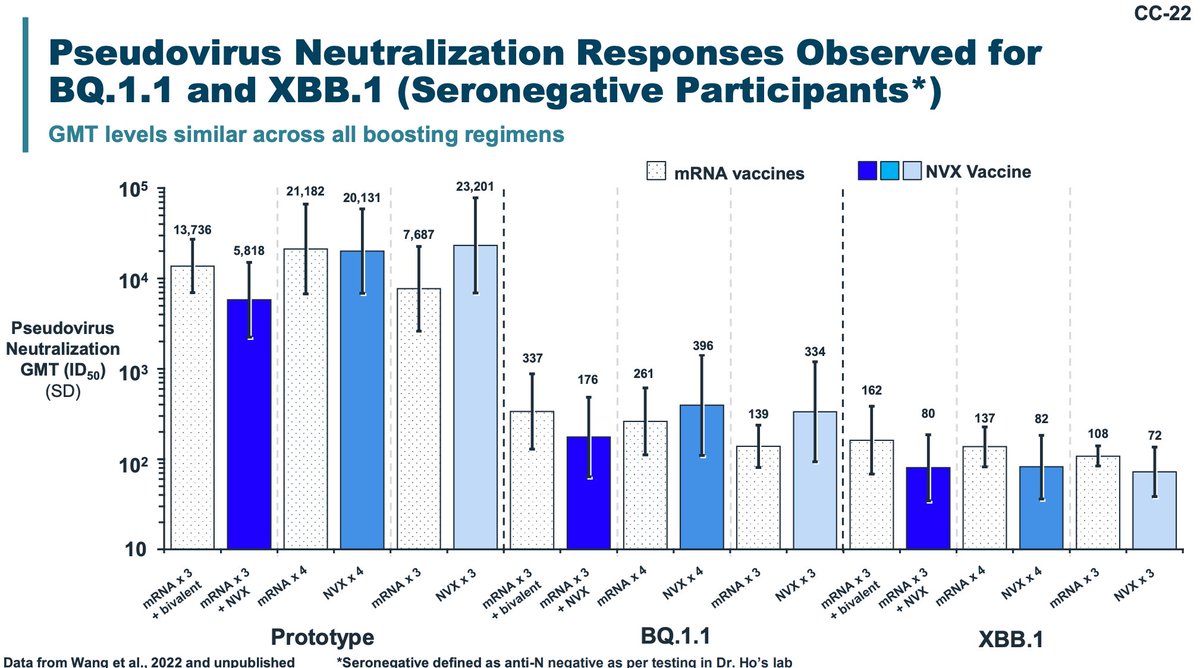
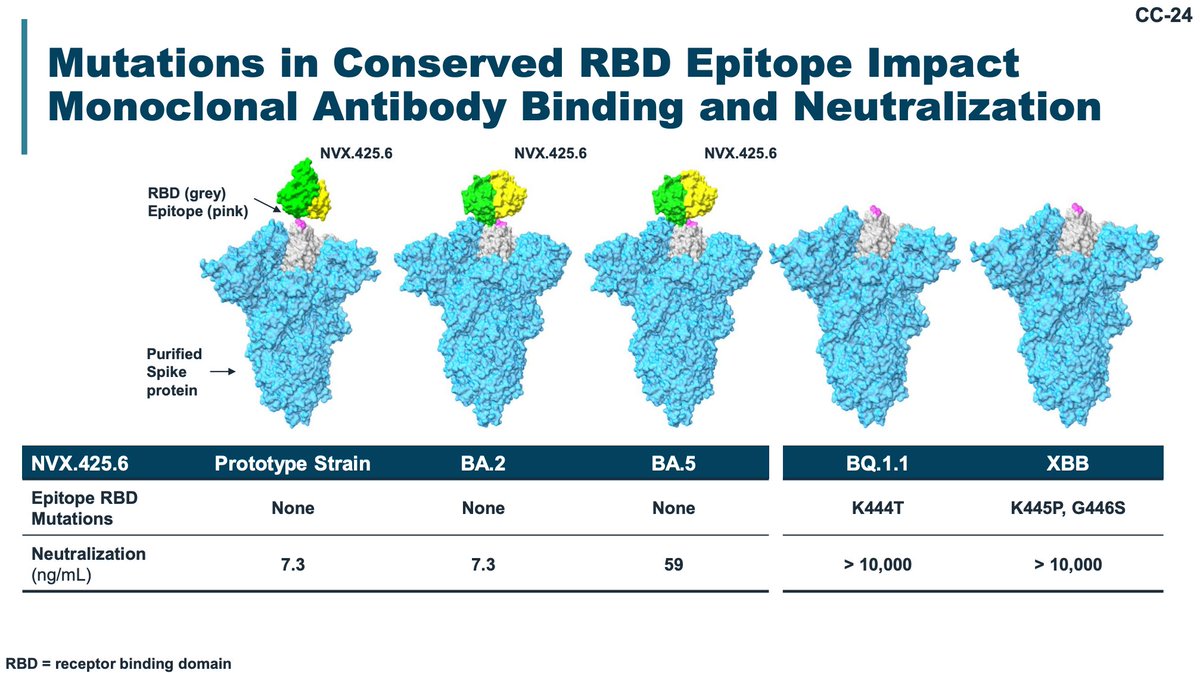
Novavax previous identified an epitope that was very well conserved across spikes and could be targeted by a neutralizing antibody elicited by their vaccine. That epitope is lost in BQ.1.1 and XBB variants. 
Here are Novavax's summary slides. It's a solid vaccine. Could do with way less overt marketing throughout their presentation. 



Novavax reports needing 6 months between strain selection and distribution of the vaccine... not ideal, but that's a fundamental limitation of the technology.
Berger- subanalyses looking specifically at minority populations to ensure that the performance does not vary by race/ethnicity?
Novavax- group sizes are too small for that analysis with current studies but prior studies showed no differences.
Novavax- group sizes are too small for that analysis with current studies but prior studies showed no differences.
Gans- efficacy data was not mentioned. On the immunogenicity question you favor a new vaccine that would be antigenically similar to VOCs but novavax monovalent and bivalent do similarly- confused about this discordance.
Novavax- data supports that the immune responses...
Novavax- data supports that the immune responses...
held up well through BA.5, but BQ and XBB are different. We have seen data today that efficacy/effectiveness are preserved even among current variants. If we move to closer matched strains, we can recapitulate those gains and protect against milder infections and spread (agree)
Structure data clearly shows the loss of the key protective epitopes; monovalent vs bivalent is less important. Efficacy is still maturing- not enough vaccine uptake to reliably measure.
Levy- tell us about the adjuvant
Novavax- matrix M was critical for the breadth and neutralization of responses; was also seen with an influenza vaccine. If we can get a strain selection that lands us on the same antigenic tree, the tech will induce broad response to drifted VOC
Novavax- matrix M was critical for the breadth and neutralization of responses; was also seen with an influenza vaccine. If we can get a strain selection that lands us on the same antigenic tree, the tech will induce broad response to drifted VOC
Gellin- need an end of first quarter decision to allow for 6 month delivery?
Novavax- antigenically matched strains would help, 6 months is broadly necessary. The time period could be shortened but 6 months from sequence to product.
Novavax- antigenically matched strains would help, 6 months is broadly necessary. The time period could be shortened but 6 months from sequence to product.
Chatterjee- does novavax plan to offer any more conserved proteins than spike in their pipeline?
Novavax- they would not really elicit a benefit or antibodies and that is the focus of the vaccine's immunology
Novavax- they would not really elicit a benefit or antibodies and that is the focus of the vaccine's immunology
It is now lunch break time :)
Anyway I will check out questions but the overall theme here is if we try a flu model (and IMO we should), the mRNA vaccines will clearly win. Pfizer's idea of an at-risk pipeline of VOC-adapted vaccines to be deployed just in case is a really good one.
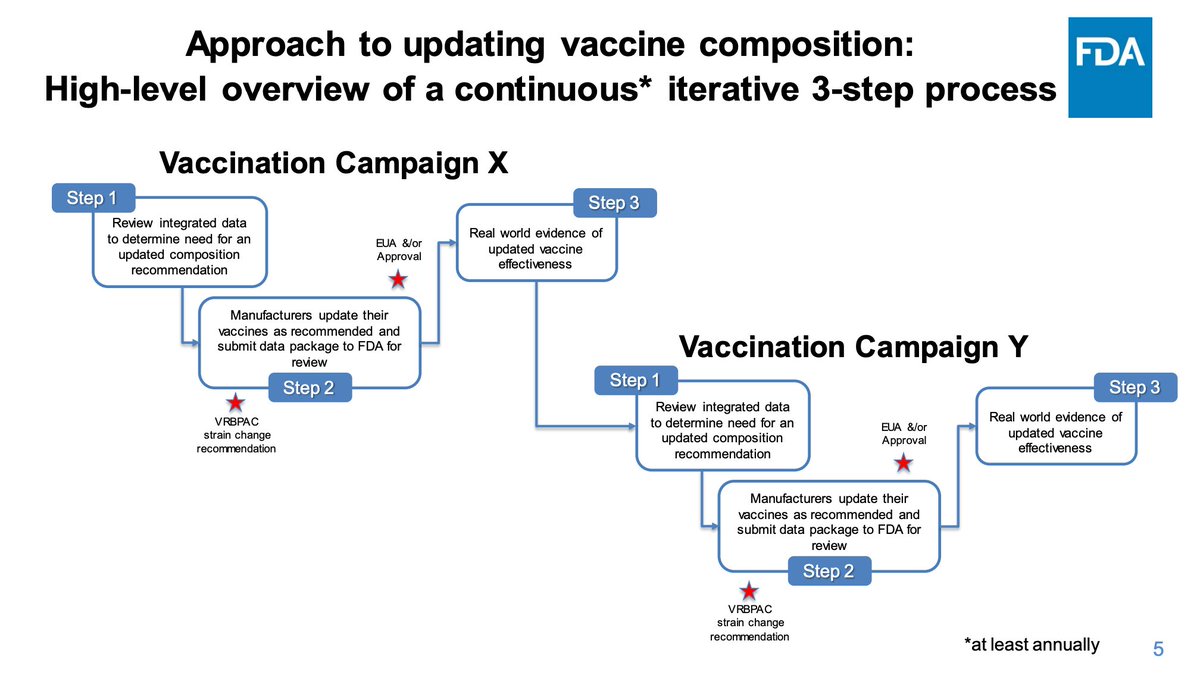
We have now returned to this shared reality to discuss considerations for changes to COVID-19 vaccine strain composition, presented by Dr. Jerry Weir. Here is an overview of where we are now and how we got here 

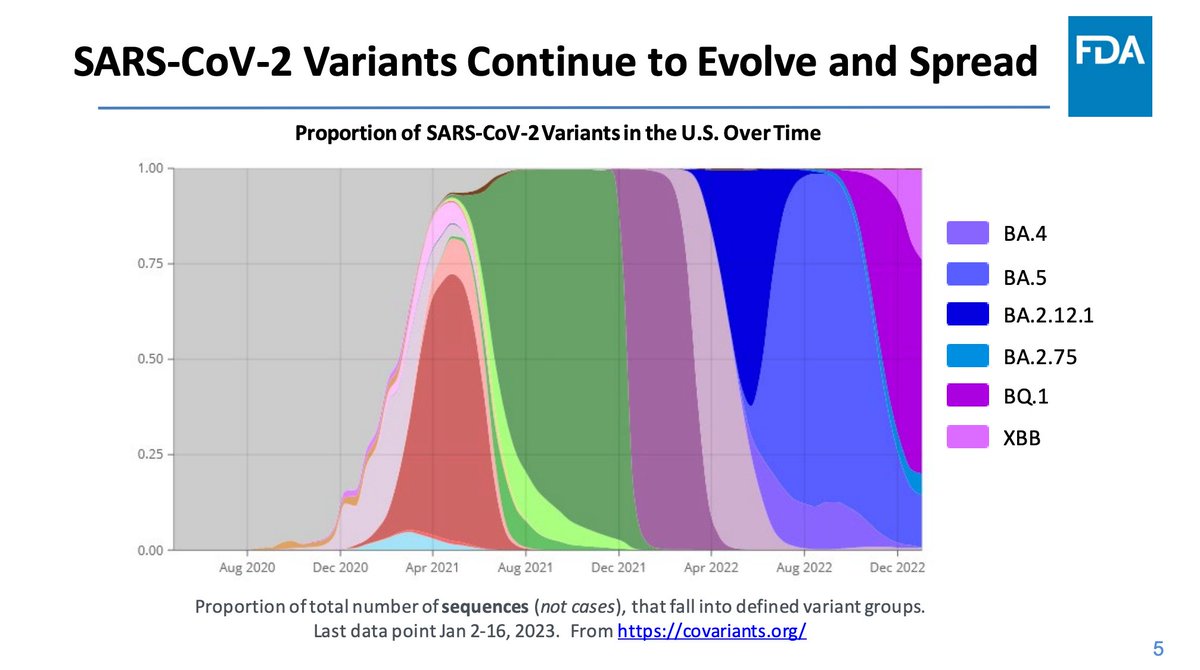
SARS-CoV-2 has continued to evolve. We've seen a lot of these figures from Dr. Scobie's presentation. Omicron behaved similarly to an antigenic shift event rather than an antigenic drift in analogy to influenza. 

The bivalent vaccines elicited superior immune responses to monovalent vaccines. These are prospective, randomized studies submitted to the FDA for review and had enough participants for statistical analysis and the assays used are validated by FDA. 

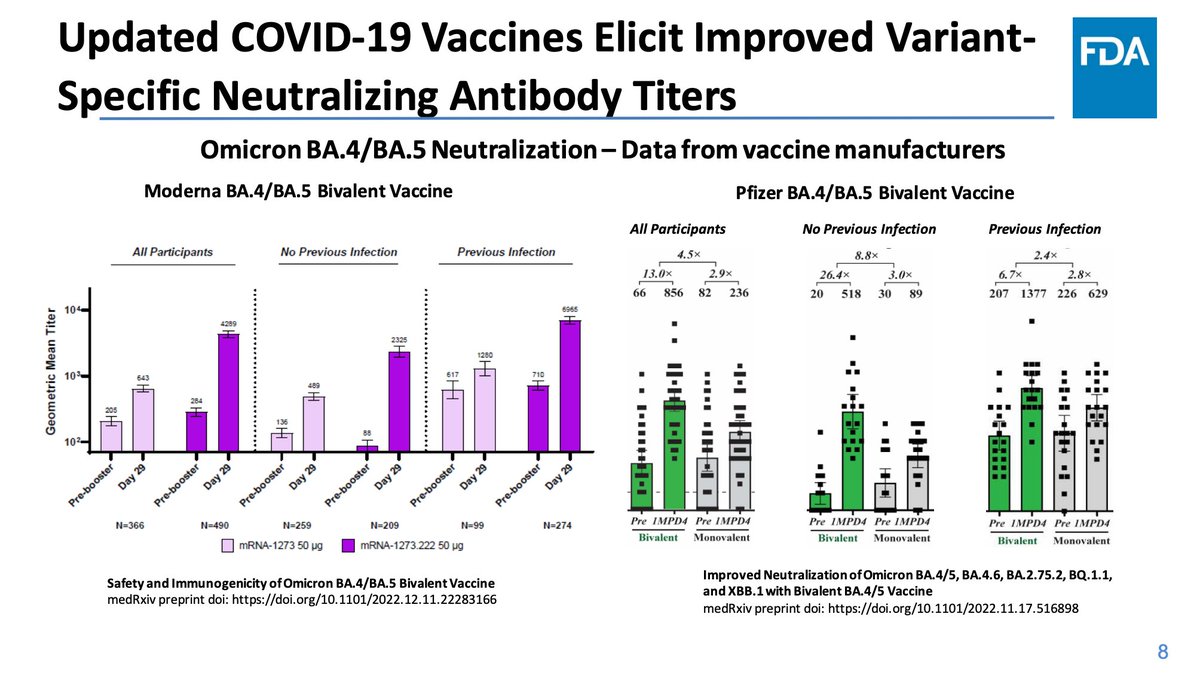

In addition, we have observational data outside of manufacturers also support the value of mRNA bivalent boosters in improving immunogenicity and in enhancing protection. The data are very consistent across the studies. 



Weir notes that the bivalent vaccines are essentially giving half doses of a novel spike, suggesting room for improvement (presumably with monovalent boosters of the drifted spikes).
We have different primary series and booster compositions which is a challenge for administration and can lead to errors. Here are possible ways to simplify this issue. In June, FDA and VRBPAC did not consider a bivalent for the primary series. 



This was because there was very little data to suggest it was applicable for a primary series (ehh...) and some data suggested that Omicron spike was less immunogenic (true, but that's why you have ancestral spike in the bivalent...). It doesn't seem to hold true for later...
omicron subvariants. There was a sense to move conservatively because WHO had not advised a change to the primary series (weak IMO). Today, we know a lot more, including specific trials for these primary series and boosters.
At this point in the pandemic, given widespread pre-existing immunity, a single dose vaccine should suffice for most people as a booster. Population-level review should be able to identify gaps in seroprevalence and increased risk indicating the need for additional doses. 



FDA has recommended a process for strain composition update to COVID vaccines in April 2022, and manufacturers have reached out with questions to clarify. Weir raises concerns that Novavax might not be able to keep up with strain updates. 





Weir says the object is not to chase variants as this seems unrealistic (disagree), but the data indicate we can continue to make improvements to the vaccines (agree). The emergence of a more pathogenic escape virus would trigger an ad hoc VRBPAC meeting as for 2009 flu. 
Offit- surprised to liken omicron to an antigenic shift; increased risk in severe disease is not apparent here and that isn't the case with Omicron (disagree)
Weir- Drift before omicron was a lot smaller than what we had with Omicron
Offit- T cells continue to protect...
Weir- Drift before omicron was a lot smaller than what we had with Omicron
Offit- T cells continue to protect...
Offit cont- question about clinical significance of increased neutralizing antibody titers etc. Do you see us heading to vaccines that do not use the ancestral strain.
Weir- to be discussed at the next VRBPAC on strain composition (are you for real? do it now)...
Weir- to be discussed at the next VRBPAC on strain composition (are you for real? do it now)...
gut feeling is monovalent would have been better than the bivalent (probably).
Levy- what are the goals here re outcomes? Do we want to prevent milder disease too? Is that realistic? What are the correlates of protection by age group and by vaccine? Does FDA plan to analyze CoP by endpoint?
Weir- that will be very difficult given seroprevalence but...
Weir- that will be very difficult given seroprevalence but...
nonetheless it's very important.
Chatterjee- if a manufacturer decided not to update, would FDA withdraw their EUA/BLA?
Weir- cannot give a definitive answer, internal discussion for that needed.
Chatterjee- Encourage FDA to plan for full licensure rather than EUAs going forward
Weir- cannot give a definitive answer, internal discussion for that needed.
Chatterjee- Encourage FDA to plan for full licensure rather than EUAs going forward
There is now a short break
Rubin- what should the public take away regarding the safety of vaccines with respect to ischemic strokes?
Shimabukuro- CDC continues to recommend all eligible individuals receive a vaccine. A statistical signal was found and it is being assessed but evidence is not sufficient..
Shimabukuro- CDC continues to recommend all eligible individuals receive a vaccine. A statistical signal was found and it is being assessed but evidence is not sufficient..
to conclude that there is a real safety issue due to stroke and everyone eligible should receive a bivalent booster. More formal epidemiological investigations are underway.
Forshee- Multiple systems are in place to look for any potential safety signals with vaccines...
Forshee- Multiple systems are in place to look for any potential safety signals with vaccines...
and it is treated as follows: early warning system identifies a signal, rigorous systems used to dissect whether or not it's real. Systems sometimes give signals in one database but not others- this is why we investigate.
Shimabukuro- Systems are designed to be...
Shimabukuro- Systems are designed to be...
sensitive and to rapidly identify potential concerns and address them. The thoroughness in which these findings are being assessed demonstrates the safety system is working (strongly agree). The public and medical community should be confident in our ability to assess safety...
and to communicate in a timely and transparent manner about these matters.
Gans- safety surveillance appears very robust but how are we handling how vaccines may affect other events that are not necessarily of interest or amenable to RCA? Also for Novavax- is there going to be peds data looked at and when will it be available?
For everyone- a bank of preclinical data of vaccines not including ancestral and future variants, etc.
Shimabukuro- outcomes prespecified in RCA, but VAERS accepts all reports and CISA which does detailed consultation at the request of healthcare providers...
Shimabukuro- outcomes prespecified in RCA, but VAERS accepts all reports and CISA which does detailed consultation at the request of healthcare providers...
with respect to reports of people experiencing debilitating illnesses and long-lasting health problems following COVID vaccination and no specific cause for symptoms may be found. Illness is disruptive and stressful under those circumstances and these health problems...
are extremely disruptive and we wish for improvement and will continue to examine them.
Novavax- currently doing pediatric development down to age 2. Plan to escalate down to 6 months after safety cohorts. Preprint available from partners at Serum Institute of India and...
Novavax- currently doing pediatric development down to age 2. Plan to escalate down to 6 months after safety cohorts. Preprint available from partners at Serum Institute of India and...
the vaccine there is authorized down to age 7.
Moderna- Library of variant vaccines accumulates based on risk assessment and epidemiology and can respond to off-cycle selection. Last year produced BA.4/5 in 90 days. Welcome continued conversations about it.
Moderna- Library of variant vaccines accumulates based on risk assessment and epidemiology and can respond to off-cycle selection. Last year produced BA.4/5 in 90 days. Welcome continued conversations about it.
Pergam- follow up on Gans question; is there going to be enough study population to do initial studies on these updated vaccines as primary series? How to approach immunosuppressed populations given their altered immunization schedule needs?
Peter Marks- for immunocompromised people, we have had multiple vaccines and an extra vaccine is part of the primary series; whether that translates into 2 vaccines per year or what it would for an initial vaccination needs a discussion and data. Part is bc...
immunocompromised is a spectrum and it varies widely in its immunization needs.
Berger- data on hospitalization rates and other impacts was helpful; any granularity on length of stay or severity, preserving healthcare system capacity?
Jones- COVID-NET includes that information and it is published. Post-hospitalization requires additional platforms.
Jones- COVID-NET includes that information and it is published. Post-hospitalization requires additional platforms.
Gellin- when in the calendar do we want to have optimal protection i.e. seasonality, and when should vaccine be available/be done, incorporating waning? The CDC data about diversity of variants around the country is revealing- we have seen coordination with WHO but this has...
not been discussed with a global perspective in mind.
Jones- Generally seen peaks during winter months but interseasonal peaks as well. More seasons needed. Agree with FDA approach of fall booster campaigns in anticipation of max protection during winter.
Jones- Generally seen peaks during winter months but interseasonal peaks as well. More seasons needed. Agree with FDA approach of fall booster campaigns in anticipation of max protection during winter.
It would take a very complex model to understand how fall rollout of vaccine would affect Delta, Alpha etc.
Marks- Seasonality is emerging and we see advantages to winter seasonality because we can cover in Fall and ease burden from RSV, flu, COVID tripledemic.
Marks- Seasonality is emerging and we see advantages to winter seasonality because we can cover in Fall and ease burden from RSV, flu, COVID tripledemic.
We would like to work with global partners including WHO and other regulators to coordinate well, it's just a matter that not all regulatory agencies and we need global surveillance of the variants and good coordination with all the partners therein.
Weir- Agree with Marks. WHO does have a group that monitors variants and at least occasionally makes recs. They do not have a set schedule. About 6 months ago they invited FDA to join and FDA enthusiastically accepted. We will participate if we can coordinate.
Unlike flu, global distribution of variants is more variable (see Covariants to see how it differs by geography). Variants don't sweep across the world uniformly like they seem to for flu. First responsibility however is the US.
Nelson- there is a seasonality with a late summer mini-peak in addition to a winter peak. Are these two peaks distinct or are they connected i.e. winter peak targeting to ameliorate summer peak?
Jones- Recency of infection affects your ability to be infected and transmitted
Jones- Recency of infection affects your ability to be infected and transmitted
Nelson- re rare AEs, can we reassure the public and committee that there are mechanisms in place to look at signals across entire population but also specific subpopulations with enhanced risk?
Shimabukuro- VAERS monitors everything and attempt to follow up for serious AEFI...
Shimabukuro- VAERS monitors everything and attempt to follow up for serious AEFI...
and CDC follows up and requests medical records for these situations. Epi studies are needed to address specific events in specific subgroups. In monitoring for serious, rare AEs attempts are made to get as much clinical information as possible.
Forshee- we have experience working with very rare AEs, in flu for instance, GBS is regularly monitored which is very rare and risks below 1 per 100,000 can be detected because of the size of the databases.
Bernstein- a lot of emphasis on neutralizing, but more T cell data would help; is it known what the ideal amount of antigenic content is needed for durable T cell protection by age, especially in young children?
Weir- No not known currently and how it contributes is...
Weir- No not known currently and how it contributes is...
unclear. We aren't sure how to measure it.
Cohn- Immunization in very young children- are there plans to modify schedule to align more with routine vaccination schedule in children or wider spacing to optimize immunogenicity?
Moderna- using 8 week interval in Baby CoVE to align with CDC schedule; select dose and get...
Moderna- using 8 week interval in Baby CoVE to align with CDC schedule; select dose and get...
to placebo part of the study.
Pfizer- evaluating the vaccines in children 6 months to 5 years and also extended intervals and further studying that to see impact on immunity
Pfizer- evaluating the vaccines in children 6 months to 5 years and also extended intervals and further studying that to see impact on immunity
Reingold- simultaneous administration of flu vaccine vs. COVID vaccine is an issue because if people have to come in on separate days it will hurt coverage
Marks- Formal epi studies on coadministration of flu vaccines in large databases will be done. Some companies may be...
Marks- Formal epi studies on coadministration of flu vaccines in large databases will be done. Some companies may be...
studying this. Uptake of boosters was not adequate this current past fall.
Meissner- do we really want to stop asymptomatic infections by SARS-CoV-2? That doesn't seem like a reasonable objective and an asymptomatic infection will boost immunity and enhance protection. That is likely not possible. The vaccine lags behind the virus's evolution.
Beigel- That's more than we can accomplish today. Decreasing overall community transmission and ease burden of disease (YES THANK YOU JESUS WHY IS THIS SO HARD FOR PEOPLE). Theoretical benefits of asymptomatic infection need to be balanced.
Lee- coadministration of flu and COVID, looking at stroke data- is there any reason to think that spreading those out would reduce stroke risk?
Shimabukuro- post hoc analysis; there may be reasons other than vaccination to account for the finding and evidence...
Shimabukuro- post hoc analysis; there may be reasons other than vaccination to account for the finding and evidence...
to date is not sufficient is there to conclude that this signal is real and so discussion of spacing out the flu and covid vaccines are premature.
Forshee- Agrees; more formal epi study forthcoming to explore the question
Forshee- Agrees; more formal epi study forthcoming to explore the question
Kim- What is FDA's position on Novavax and Novavax, what is the view on how the vaccine can be used in the context of today's discussion?
Novavax- approved for primary series and booster; Important tool for boosting in the upcoming season.
Novavax- approved for primary series and booster; Important tool for boosting in the upcoming season.
Novavax cannot chase the strains so wants to emphasize breadth.
Weir- I mean... it's an authorized vaccine so FDA is clearly fine with it (lol what is this question?)
Weir- I mean... it's an authorized vaccine so FDA is clearly fine with it (lol what is this question?)
Sawyer- can each manufacturer discuss whether they will shift to single dose vials or not? Lots of vaccine waste going on in pediatrics.
Moderna- Working to make single dose vials and prefilled syringes to simplify things and can achieve it in time.
Moderna- Working to make single dose vials and prefilled syringes to simplify things and can achieve it in time.
Pfizer- Also will be using single-dose vial and can support that with future vaccine updates.
Novavax- single dose vial is being aimed for
Novavax- single dose vial is being aimed for
McInnes- We think we can have a periodicity of the response, but operationalizing that is really tough for manufacturers to get both flu and covid vaccines. COVID is distinct from flu- how do you build an operationalized periodicity?
Weir- the manufacturers told us how much time they would need. It seemed to work this June in 2 months fine. We have to be flexibility- there isn't a good pattern yet. We will adjust over time.
Chatterjee- a combination influenza/COVID-19 vaccine product being evaluated was discussed in the comment section. Are manufacturers working on this?
Pfizer- work initiated to evaluate combined vaccine and in early stages; robust safety assessments are part of that.
Pfizer- work initiated to evaluate combined vaccine and in early stages; robust safety assessments are part of that.
Moderna- phase 1 has been initiated for a combo covid/flu.
Novavax- earlier this year announced a combo product as well and started a Phase 2. Data should be available midyear.
Novavax- earlier this year announced a combo product as well and started a Phase 2. Data should be available midyear.
Meissner- it is hard to say with precision how frequently boosters should be offered right now. We need to see what happens with disease burden. We may or may not need annual vaccination- it's just awfully early to answer that. Much discussion of CoP- its very hard to...
disentangle humoral immunity from cell mediated immunity (that's why we do passive/adoptive transfer studies in animals). We need more effort from CDC about hospitalizations caused by COVID
Bernstein- we cannot realistically have all things (long covid, mild disease, transmission, etc). Unclear that using a bivalent in the youngest makes sense (what meeting has this person been attending???)
Offit- would support harmonization with BA.5/ancestral booster and think it makes sense to get closer to what is circulating. Many people simply will not make a good immune response- we need antivirals. Any of these vaccines seem to do very well in...
protecting against severe disease. There it does not fit with flu- flu vaccines when you miss it's a BIG miss. That isn't true for COVID-19 (mmm... agree and disagree)
Sawyer- Support harmonization; data on young children seems lacking for bivalent primary series (small numbers). How much data do we need to justify this for children?
Marks- Look at the totality of the data- some data are mutually reinforcing. We agree numbers now are small...
Marks- Look at the totality of the data- some data are mutually reinforcing. We agree numbers now are small...
but hopefully as more data come in we will have a larger dataset. The reassuring thing is the safety profile with the bivalent boosters mirrors very well the monovalent vaccines. We will need more data. Getting towards one vaccine composition will ultimately help.
Berger- Agree with Offit; our job is to protect against severe disease. When you look at 16-fold risk difference in hospitalization in boosted vs. unvaccinated it's clear. Questions about dosage remain unanswered. Harmonization makes sense.
McInnes- have the bivalent boosters added anything to the monovalent? We lack randomized data to show risk reduction in severe disease.
This trial is like impossible lol
This trial is like impossible lol
Gans- Question is really about vaccine harmonization, and there is agreement here in thinking about how we're moving and how we get to the next stage. Having the primary and booster match makes sense. More kids with coinfections- they are seriously sick. Disease in youngest...
can be quite bad with coinfections. Composition is confusing as opposed to harmonization.
Wharton- This is the right move for the program; some data and evidence gaps to be filled in but this is a smart move
Cohn- This is a very good decision from a programmatic and implementation perspective. Very concerning to see such low coverage in 6 months to 5 years. We have more kids aging into this group and needing the vaccines.
Hawkins- Support the discussion; safety and effectiveness are confirmed, myocarditis notwithstanding. US has broadly accepted that COVID is here to stay. Need to fill in knowledge gaps and protocols therein
Gellin- we can't keep doing what we're doing, we need to move on. Despite how good these vaccines are, we need better ones. This is a reasonable approach. We have to remember this isn't influenza.
The vote is unanimously in favor to update all vaccines in circulation in the US to be bivalent. As a reminder, VRBPAC's recommendations are not binding. 
They are now explaining their votes but given they were all unanimous and I am tired so I will only put it in the thread it if it's novel/interesting
Cohn- Note that we also saw data that the monovalent in kids seems to work quite well against symptomatic infection and it will take time to implement bivalent primary series- parents should not delay vaccinating their children for the bivalent
Rubin- everyone not vaccinated and without solid evidence of prior infection should receive 2 doses. We know very little about dosing intervals and the effects on protection. We really want to do those studies- they are very important. We need to decide in advance what data...
needs to be collected to help with that.
Marks- this discussion is not meant to be limited to a 2-dose series.
Marks- this discussion is not meant to be limited to a 2-dose series.
Gans- This discussion will differ for different groups. We need age and comorbidity-specific data. Those who need to be immunologically naive year to year- we need to decide whether 2 doses are enough. Broad immunogenicity studies are needed- T, B, mucosal, efficacy, persistence
Chatterjee- want a lot more data for young children- the trials are too small as they are now to justify certain decisions as we have entered an endemic phase (that's a... spicy assessment)
Wharton- A reset for vaccine recs given the widespread state of immunity does make sense. It has been raised that there is a lot of uncertainty but what are the exceptions? Dosage and number of doses in young children are key. Think about what is needed to protect high risk.
Sawyer- simpler is better; pediatricians are used to adjusting dose numbers based on age. Similar idea for immunocompromised individuals.
Offit- certain things are clear- the virus is here to stay and vulnerable to serious disease. We need to know exactly who those people are from CDC. That needs to be given with immunological data to define both in terms of cellular and humoral effectors, then we can decide...
who gets what and when and what is best.
Pergam- confusion still exists in pediatrics with dose numbers. As we discuss this, we need to keep in mind how we approach this question with other platforms.
Pergam- confusion still exists in pediatrics with dose numbers. As we discuss this, we need to keep in mind how we approach this question with other platforms.
Reingold- whether the vaccines are intended to prevent infection or not; much time spent explaining that the vaccines are not intended to stop infection. Important to prevent rare, serious outcomes by vaccinating everyone (YES)- we need better messaging (HELL YES)
Gans- we really need safety data as well as efficacy and immunogenicity- there are tons of data globally to work with here. We should be collaborating
Marks- FDA is working with over 60 countries to do surveillance and collaborating therein and they did not see a stroke signal which was reassuring
Meissner- interchangeability of the vaccines needs to be addressed in pediatrics and adults; mixing platforms- we have some data there on heterologous series but we may want more
McInnes- we should leave space for approaches beyond mRNA.
Marks- we agree and we hope harmonizing will encourage other sponsors to provide their specific platforms and we always encourage improvements
Marks- we agree and we hope harmonizing will encourage other sponsors to provide their specific platforms and we always encourage improvements
Chatterjee- paying attention to the epidemiology is key; we do not want to chase variants (disagree). Timing should be proposed as well. Everything is a moving target and we have to respond accordingly.
Rubin- we don't yet know how often we want to be boosting; hard to say whether it will be annual. Bivalent is better even if the advantage is small.
Gans- we should consider composition more broadly. The antigenic drift will affect outcomes with our current immunity and this is tightly imbricated with correlates of protection. Look at dosing- we have a chance to reexamine it in the context of immunologically experienced ppl
Pergam- open to the flu annual model where it might work but there was Delta, and then Omicron in the summer. Wonder if initially they should discuss at different time points as we sort through this. We're planning for the future without acknowledging history here.
Should we include the ancestral strain in future vaccines? We need to discuss that.
McInnes- Struck by us kind of leapfrogging over the data we normally had with dose-response to antigen and our ongoing lingering on T cells and mucosal responses which we don't know enough about. We need, from a public health perspective, breadth of protection
I just have so many questions about this comment
Gellin- will we be here again in June? what is the plan?
Marks- another meeting in Mayish to discuss what is circulating and what to put in a fall vaccine- as good a guess as we can make
Marks- another meeting in Mayish to discuss what is circulating and what to put in a fall vaccine- as good a guess as we can make
Marks- agree that this isn't flu, but the model of strain selection has been a key public health advance and we can take the best of that
Gellin- is tis there a companion ACIP meeting to discuss this?
Wharton- ACIP and CDC will work closely with FDA to address this.
Wharton- ACIP and CDC will work closely with FDA to address this.
Nelson- we need dose response curves and we need to make sure COVID vaccines are available throughout the year given that it can appear out of season and that would likely have value
Gans- June may not be enough time given that there are still data for questions that still need to be gathered. We need more time for the data.
Bernstein- the pattern is not necessarily the flu; it may be important to get it over the summer as well.
Perlman- we need to continue sequencing efforts; with 229E this virus was thought to be invariant and yet serological data confirms drift. Keeping ancestral strain in is unclear because the virus might not recycle like it does for flu (which still takes years)
General agreement about updating; we need additional strategies, mixing of mRNA and protein vaccines, immunogenicity data, etc.
Marks- the comments are well taken; a data driven approach going forward is critical and FDA will continue to look forward to working with scientists and clinicians on next gen COVID vaccines
Meeting adjourned
• • •
Missing some Tweet in this thread? You can try to
force a refresh