1/8
I am referred occasional otherwise healthy patients for evaluation of lymphopenia (aka lymphocytopenia).
What to do?
I am referred occasional otherwise healthy patients for evaluation of lymphopenia (aka lymphocytopenia).
What to do?
2/8
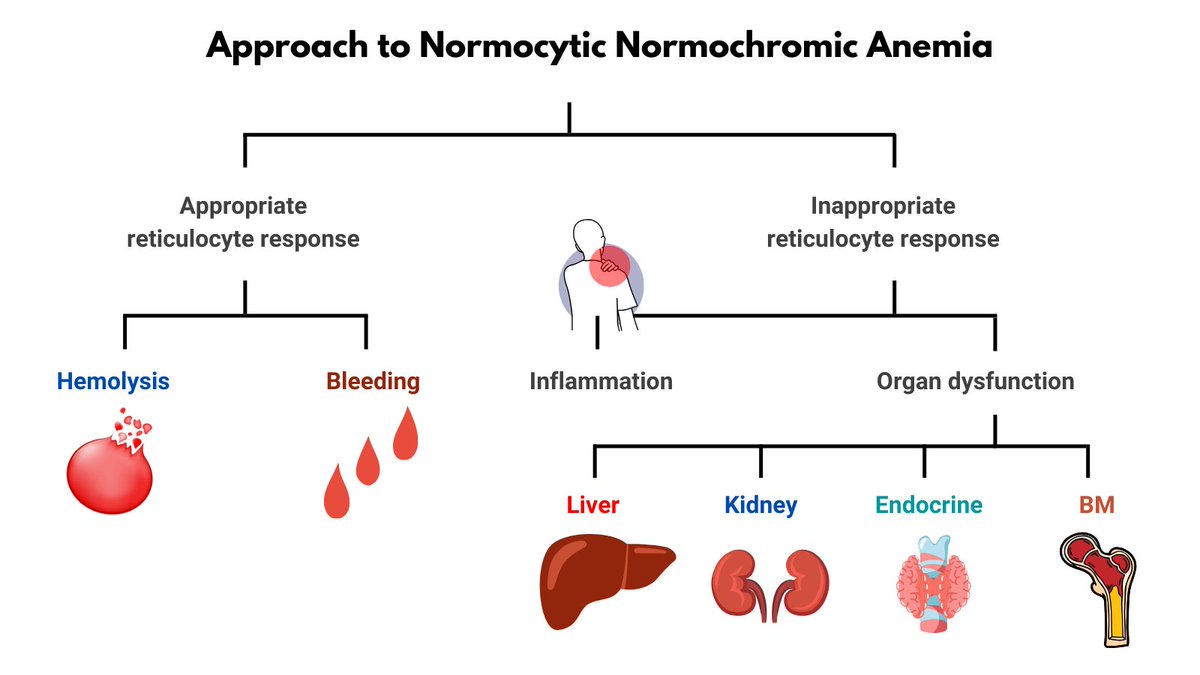
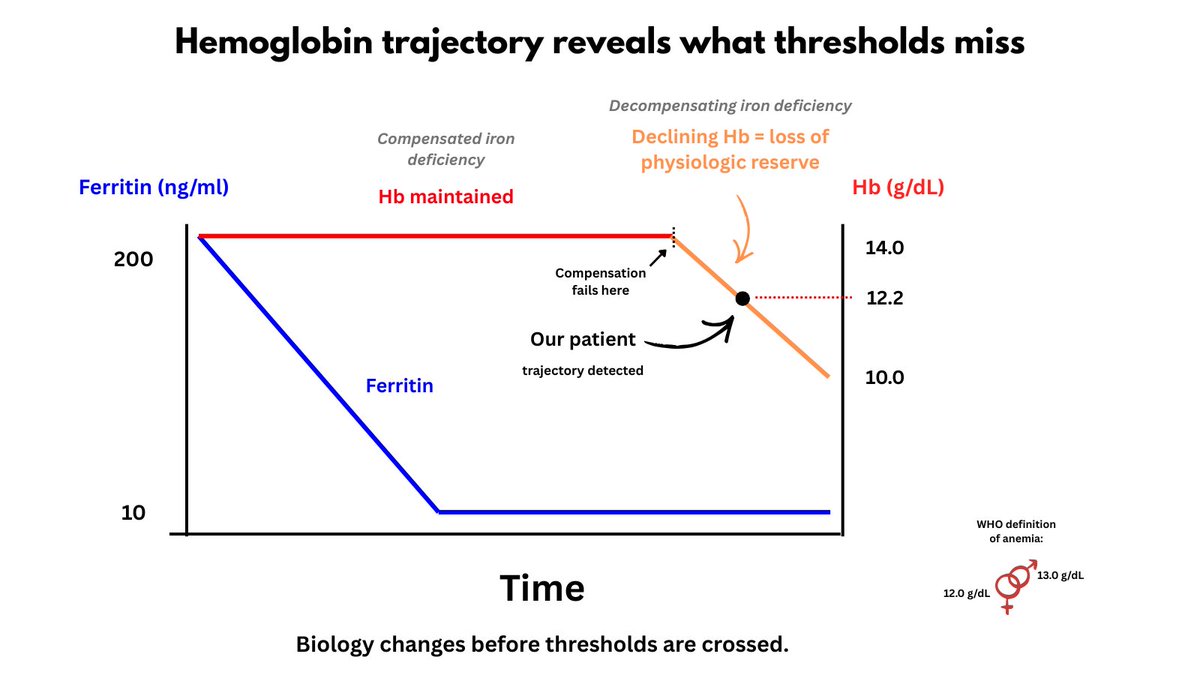
1. Let's begin with definitions - lymphopenia is usually defined as an absolute lymphocyte count ALC <1000 cells/microL for adults
1. Let's begin with definitions - lymphopenia is usually defined as an absolute lymphocyte count ALC <1000 cells/microL for adults

3/8
2. Epidemiology - lymphopenia has been documented in 1.5-3% of CBCs from both community and hospitalized patients.
2. Epidemiology - lymphopenia has been documented in 1.5-3% of CBCs from both community and hospitalized patients.

4/8
3. Causes/associated conditions - lymphopenia may be found incidentally in someone without obvious underlying cause (most common) or may be associated with a number of diseases/conditions.
3. Causes/associated conditions - lymphopenia may be found incidentally in someone without obvious underlying cause (most common) or may be associated with a number of diseases/conditions.

5/8
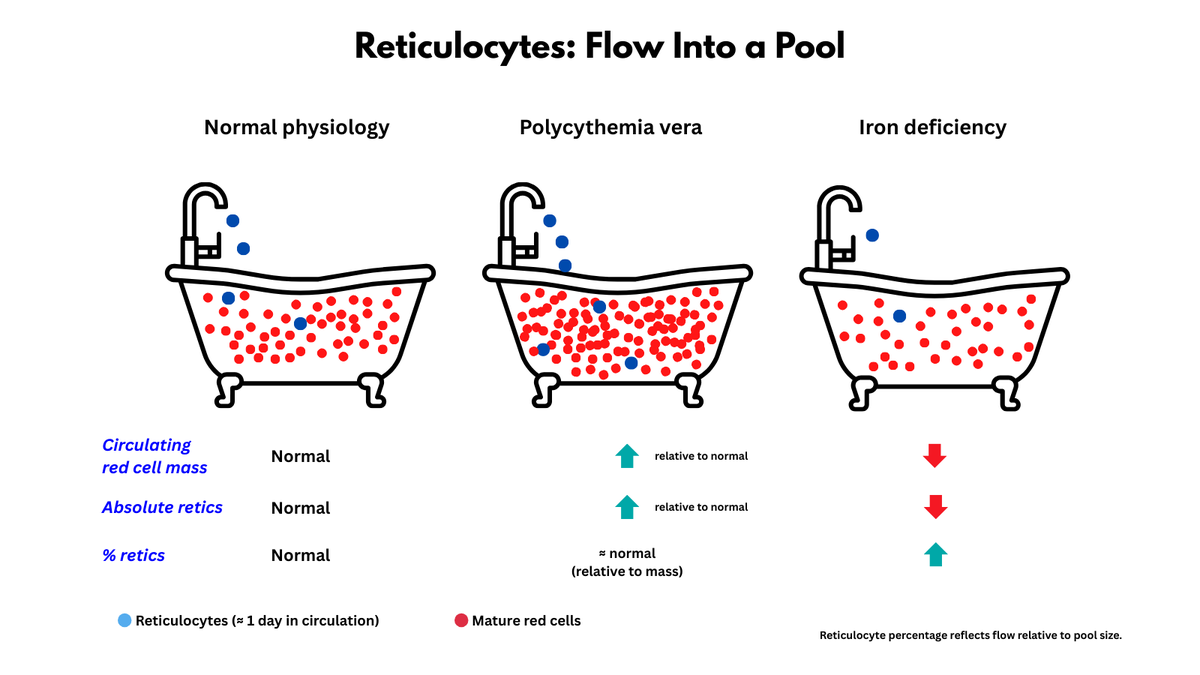
4. Mechanisms - reduced production, increased destruction, increased apoptosis, redistribution of lymphocytes between blood and various lymphoid tissues.
4. Mechanisms - reduced production, increased destruction, increased apoptosis, redistribution of lymphocytes between blood and various lymphoid tissues.

6/8
5. Clinical implications - no evidence (outside of large population studies that do not take into account underlying cause) that lymphopenia associated with increased risk of infection.
5. Clinical implications - no evidence (outside of large population studies that do not take into account underlying cause) that lymphopenia associated with increased risk of infection.

7/8
6. History and physical - focus on symptoms/signs associated with underlying cause/associated conditions(s).
6. History and physical - focus on symptoms/signs associated with underlying cause/associated conditions(s).

8/8
7. Lab tests - generally not needed in an otherwise healthy individual.
8. Treatment - directed towards the underlying cause.
7. Lab tests - generally not needed in an otherwise healthy individual.
8. Treatment - directed towards the underlying cause.

• • •
Missing some Tweet in this thread? You can try to
force a refresh