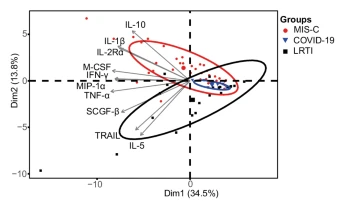
Delighted to share work on immune pathophysiology of MIS-C (PIMSTS) compared to respiratory COVID and other viral infection in children, published today in @NatureComms done @Cambridge_Uni with @GreatOrmondSt @Bham_Childrens @cuh_picu and @unibirminghamlink.springer.com/article/10.103…
High dimensional mass cytometry revealed upregulation of T cell activation, immune checkpoint and co-stimulation receptor markers in TCR Vβ21.3+ T cells in MIS-C, with reduced immune responsiveness in vitro.
And in single cell RNA sequencing of peripheral blood mononuclear cells we demonstrated increased proportions of low-density neutrophils, immature B cells and decreased CD16+ NK, cytotoxic CD8+ T cells in acute MIS-C samples with upregulation of IL-18 in natural killer cells.
There has been a question of where inflammasome activation drives MIS-C but our data suggest while IL-18 signalling links activated innate & adaptive immune cells, and facilitates superantigen-mediated T and NK cell stimulation, it is independent of NLRP3 inflammasome activation
This extensive and novel work is thanks to a large team of scientist, clinical and research nurse colleagues working through the pandemic as well as the support of our patients and their families.
Key people who supported this- @ZhenguangZhang @ClareBryant13 @DrGrahamTaylor @teichlab @pus27 @katebrown220 @BarneyUoB @pic_pram and the @DIAMONDS_2020 team who collaborated with us on this work.
@ZhenguangZhang @ClareBryant13 @DrGrahamTaylor @teichlab @pus27 @katebrown220 @BarneyUoB @pic_pram @DIAMONDS_2020 And most of all to @actionmedres @ACTcharity @CUH_NHS BRC and @gates_cambridge who supported @drjclark as well as the kind support of @jetpackjeth and Prof Michael Levin for facilitating the shared collection of samples for our own and their larger ongoing studies in DIAMONDS
• • •
Missing some Tweet in this thread? You can try to
force a refresh