My top 7 #ASCO24 abstracts to watch for #mmsm
Will have more details on those once presented/published- those are initial thoughts that may evolve according to more data
There are more interesting data will highlight during the meeting
@ASCO
🧵
Will have more details on those once presented/published- those are initial thoughts that may evolve according to more data
There are more interesting data will highlight during the meeting
@ASCO
🧵
#mmsm #ASCO24
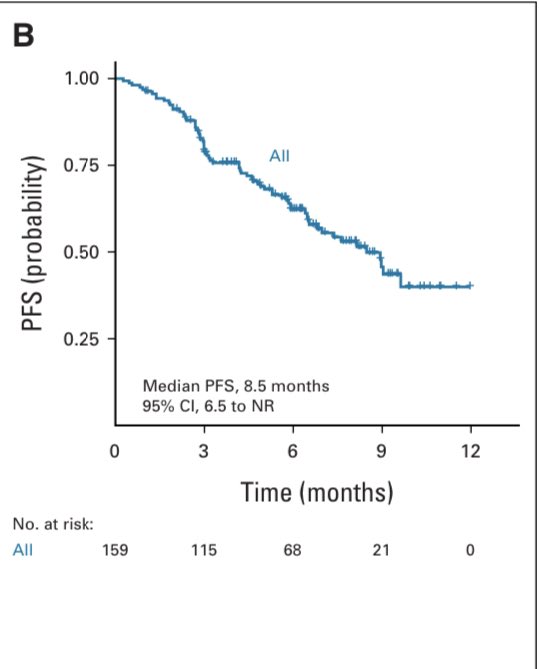
1️⃣ IMROZ: Isa-VRd vs VRd i n transplant-ineligible NDMM pts
➡️
✅ follow-up: 5 years
✅Median PFS Isa-VRd(NR, estimated 7.5 years!!) vs VRd(4.5 years)
🛑 Grade 5 TEAE: 2 times higher with Isa-VRd (11% vs 5.5%) meetings.asco.org/abstracts-pres…

1️⃣ IMROZ: Isa-VRd vs VRd i n transplant-ineligible NDMM pts
➡️
✅ follow-up: 5 years
✅Median PFS Isa-VRd(NR, estimated 7.5 years!!) vs VRd(4.5 years)
🛑 Grade 5 TEAE: 2 times higher with Isa-VRd (11% vs 5.5%) meetings.asco.org/abstracts-pres…

#mmsm #ASCO24
Thoughts on IMROZ
Very important data
✅median PFS: 7.5 yrs(estimated)
✅Grade IV AEs: higher though compare to:
MAIA: Dara-Rd: median F/U <5yrs (shorter median follow up),death related to AEs (10%) in Dara-Rd👇
Thoughts on IMROZ
Very important data
✅median PFS: 7.5 yrs(estimated)
✅Grade IV AEs: higher though compare to:
MAIA: Dara-Rd: median F/U <5yrs (shorter median follow up),death related to AEs (10%) in Dara-Rd👇

#mmsm #ASCO24
Thoughts on IMROZ
🛑baseline characteristics: will be important
For MAIA trial:
- 43% of pts >=75 yrs 👇
- Median PFS:NR was not reached (95% CI 54·8–NR) - at least more than 4.5 years
We will need the complete data
Thoughts on IMROZ
🛑baseline characteristics: will be important
For MAIA trial:
- 43% of pts >=75 yrs 👇
- Median PFS:NR was not reached (95% CI 54·8–NR) - at least more than 4.5 years
We will need the complete data
#mmsm #ASCO24
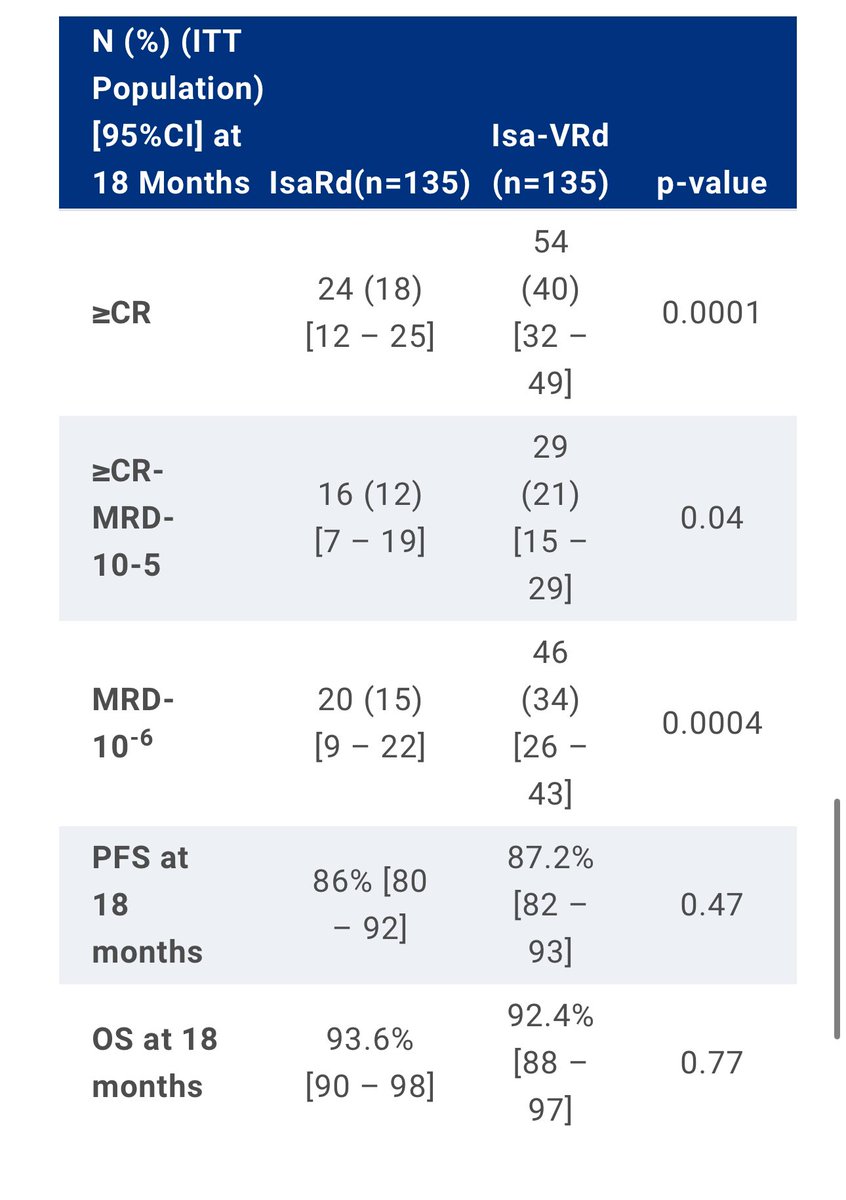
2️⃣BENEFIT/IFM2020-05
The added benefit of weekly Velcade is not (yet) reflected by improved PFS/OS
➡️
✅ median age: 73 yrs, 1 out of 3 pts >75 yrs
✅ median follow-up: 21 months
✅Deeper responses👇
🛑1 of 3 pts with grade ≥2 neuro AEs meetings.asco.org/abstracts-pres…

2️⃣BENEFIT/IFM2020-05
The added benefit of weekly Velcade is not (yet) reflected by improved PFS/OS
➡️
✅ median age: 73 yrs, 1 out of 3 pts >75 yrs
✅ median follow-up: 21 months
✅Deeper responses👇
🛑1 of 3 pts with grade ≥2 neuro AEs meetings.asco.org/abstracts-pres…
#mmsm #ASCO24
Thoughts on BENEFIT
✅We may not need a quad (usual V dosing) in most transplant in-eligible patients
✅Anti-CD38 +Rd is really a great option for most patients
✅Will be looking for high risk groups and older pts to see if quad helps this subset of patients
Thoughts on BENEFIT
✅We may not need a quad (usual V dosing) in most transplant in-eligible patients
✅Anti-CD38 +Rd is really a great option for most patients
✅Will be looking for high risk groups and older pts to see if quad helps this subset of patients
#mmsm #ASCO24
3️⃣DREAM-7: BVd vs DVd
➡️
✅ Median PFS: BVd(3 yrs) vs. DVd (1 yr)
✅ In high-risk cytogenetic subgroup, PFS was 33 mo with BVd vs 11 mo with DVd
🛑 Ocular AEs more frequent with BVd vs DVd (79% vs 29%)
DREAM-8: interesting (N/A-LBA n=8️⃣)meetings.asco.org/abstracts-pres…
3️⃣DREAM-7: BVd vs DVd
➡️
✅ Median PFS: BVd(3 yrs) vs. DVd (1 yr)
✅ In high-risk cytogenetic subgroup, PFS was 33 mo with BVd vs 11 mo with DVd
🛑 Ocular AEs more frequent with BVd vs DVd (79% vs 29%)
DREAM-8: interesting (N/A-LBA n=8️⃣)meetings.asco.org/abstracts-pres…
#mmsm #ASCO24
Thoughts on DREAM-7
✅Addition of belantamab mafodotin to PI (V) showed better PFS in randomized trial (anti-CD38 based)
✅benefit maintained in high risk disease
🛑AEs (mainly ocular) &censoring will be imp to explore- this requires full data (hopefully soon)
Thoughts on DREAM-7
✅Addition of belantamab mafodotin to PI (V) showed better PFS in randomized trial (anti-CD38 based)
✅benefit maintained in high risk disease
🛑AEs (mainly ocular) &censoring will be imp to explore- this requires full data (hopefully soon)
#mmsm #ASCO24
4️⃣ CARTITUDE-4 subgroup analysis
➡️
Difficult to treat pts(functionally high risk)
✅Better PFS with Cilta cel vs SC
✅1-yr PFS for Cilta cel: 77% vs 49%
✅Better responses 👇 meetings.asco.org/abstracts-pres…

4️⃣ CARTITUDE-4 subgroup analysis
➡️
Difficult to treat pts(functionally high risk)
✅Better PFS with Cilta cel vs SC
✅1-yr PFS for Cilta cel: 77% vs 49%
✅Better responses 👇 meetings.asco.org/abstracts-pres…

#mmsm #ASCO24
Thoughts on CARTITUDE-4 subgroup analysis
🛑This subgroup of pts remain with sub-optimal outcomes: Meaning 1 out of 4 pts who got Cilta cel earlier with functionally high risk progressed in <= 1 year
✅ Access to Cilta Cel earlier for this group is imp
Thoughts on CARTITUDE-4 subgroup analysis
🛑This subgroup of pts remain with sub-optimal outcomes: Meaning 1 out of 4 pts who got Cilta cel earlier with functionally high risk progressed in <= 1 year
✅ Access to Cilta Cel earlier for this group is imp
#mmsm #ASCO24
5️⃣ PERSEUS MRD data
➡️
✅ Rates of sustained MRD neg for ≥12 mo were higher for D-VRd vs VRd
- 10-5: 2 out of 3 compared to 1 out of 3
- 10-6: 1 out of 2 compared to 1 out of 5 meetings.asco.org/abstracts-pres…

5️⃣ PERSEUS MRD data
➡️
✅ Rates of sustained MRD neg for ≥12 mo were higher for D-VRd vs VRd
- 10-5: 2 out of 3 compared to 1 out of 3
- 10-6: 1 out of 2 compared to 1 out of 5 meetings.asco.org/abstracts-pres…
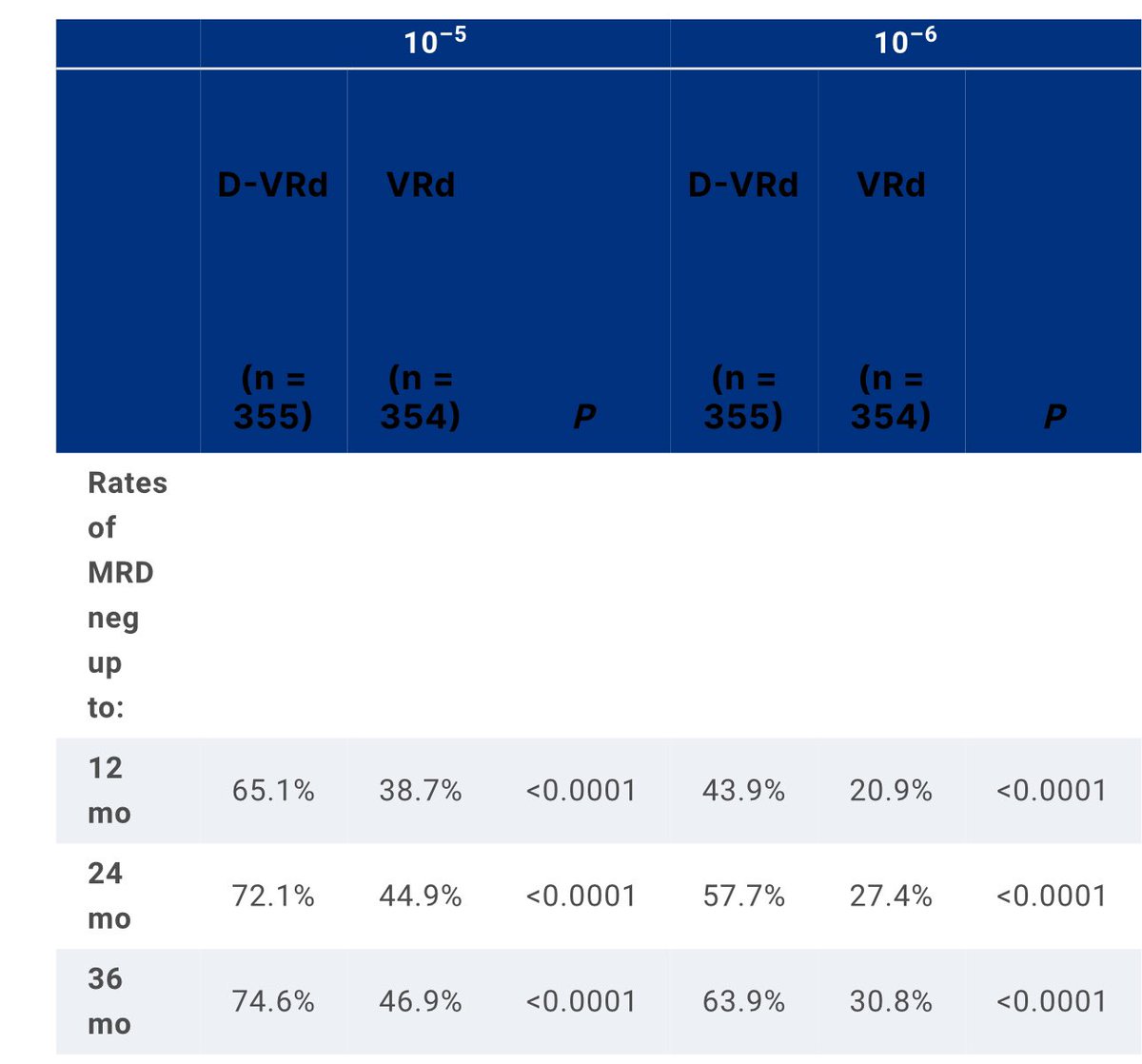
#mmsm #ASCO24
thoughts on PERSEUS MRD data
✅ Addition of anti-CD38 to induction/maintenance regimen showed improvement with long term sustained disease control
🛑We need to follow patients for >10-15 years in such important studies to better know the long term outcomes
thoughts on PERSEUS MRD data
✅ Addition of anti-CD38 to induction/maintenance regimen showed improvement with long term sustained disease control
🛑We need to follow patients for >10-15 years in such important studies to better know the long term outcomes
#mmsm #ASCO24
6️⃣MRD2STOP
➡️
✅ treatment discontinuation study
✅Median follow up after discontinuation: 2.5 years
✅ ~10% of pts progressed after discontinuation (90% didn’t)
✅ 3-year PFS was 85%meetings.asco.org/abstracts-pres…
6️⃣MRD2STOP
➡️
✅ treatment discontinuation study
✅Median follow up after discontinuation: 2.5 years
✅ ~10% of pts progressed after discontinuation (90% didn’t)
✅ 3-year PFS was 85%meetings.asco.org/abstracts-pres…
#mmsm #ASCO24
Thoughts about MRD2STOP
🛑Many pts didn’t qualify to stop tt (need of MRD -ve), some of those may do well with discontinuation
🛑Data on high risk will be imp to see.
🛑Longer follow up and more pts needed
Thoughts about MRD2STOP
🛑Many pts didn’t qualify to stop tt (need of MRD -ve), some of those may do well with discontinuation
🛑Data on high risk will be imp to see.
🛑Longer follow up and more pts needed
#mmsm #ASCO24
7️⃣ T cell subsets as predictors of response to BCMA BsAbs
➡️
✅79 pts treated with BCMA BsAb
✅ Increased proportion of CD4 cells within the ALC is significantly associated with better response to BCMA targeting bsAb (absolute no. Not impmeetings.asco.org/abstracts-pres…
7️⃣ T cell subsets as predictors of response to BCMA BsAbs
➡️
✅79 pts treated with BCMA BsAb
✅ Increased proportion of CD4 cells within the ALC is significantly associated with better response to BCMA targeting bsAb (absolute no. Not impmeetings.asco.org/abstracts-pres…
#mmsm #ASCO24
Will be looking forward to meet many colleagues, mentees, mentors and share some other important presentations @ASCO
Make sure you stop by Trainee lounge for important trainees/early career sessions @ASCOTECAG @HemOncFellows
End 🧵
Will be looking forward to meet many colleagues, mentees, mentors and share some other important presentations @ASCO
Make sure you stop by Trainee lounge for important trainees/early career sessions @ASCOTECAG @HemOncFellows
End 🧵
• • •
Missing some Tweet in this thread? You can try to
force a refresh