What is HER2 « ultralow » breast cancer, and why should you care?
A thread:
A thread:
First, a little history.
The ASCO/CAP guidelines from 2007 defined HER2 IHC 0 as “absence of HER2 staining”.
In truth, it did not make much difference if the tumor was 0 or 1+, since both were considered “negative” for HER2 protein expression.
1/11
ascopubs.org/doi/10.1200/JC…

The ASCO/CAP guidelines from 2007 defined HER2 IHC 0 as “absence of HER2 staining”.
In truth, it did not make much difference if the tumor was 0 or 1+, since both were considered “negative” for HER2 protein expression.
1/11
ascopubs.org/doi/10.1200/JC…

The 2013 update changed the definition of HER2 IHC 0 to:
-no staining
OR
-incomplete, faint/barely perceptible staining in ≤10% of cells
This change still had no clinical impact, since only 3+ and 2+/ampl cases were candidate for anti-HER2 therapy
2/11
ascopubs.org/doi/10.1200/JC…

-no staining
OR
-incomplete, faint/barely perceptible staining in ≤10% of cells
This change still had no clinical impact, since only 3+ and 2+/ampl cases were candidate for anti-HER2 therapy
2/11
ascopubs.org/doi/10.1200/JC…

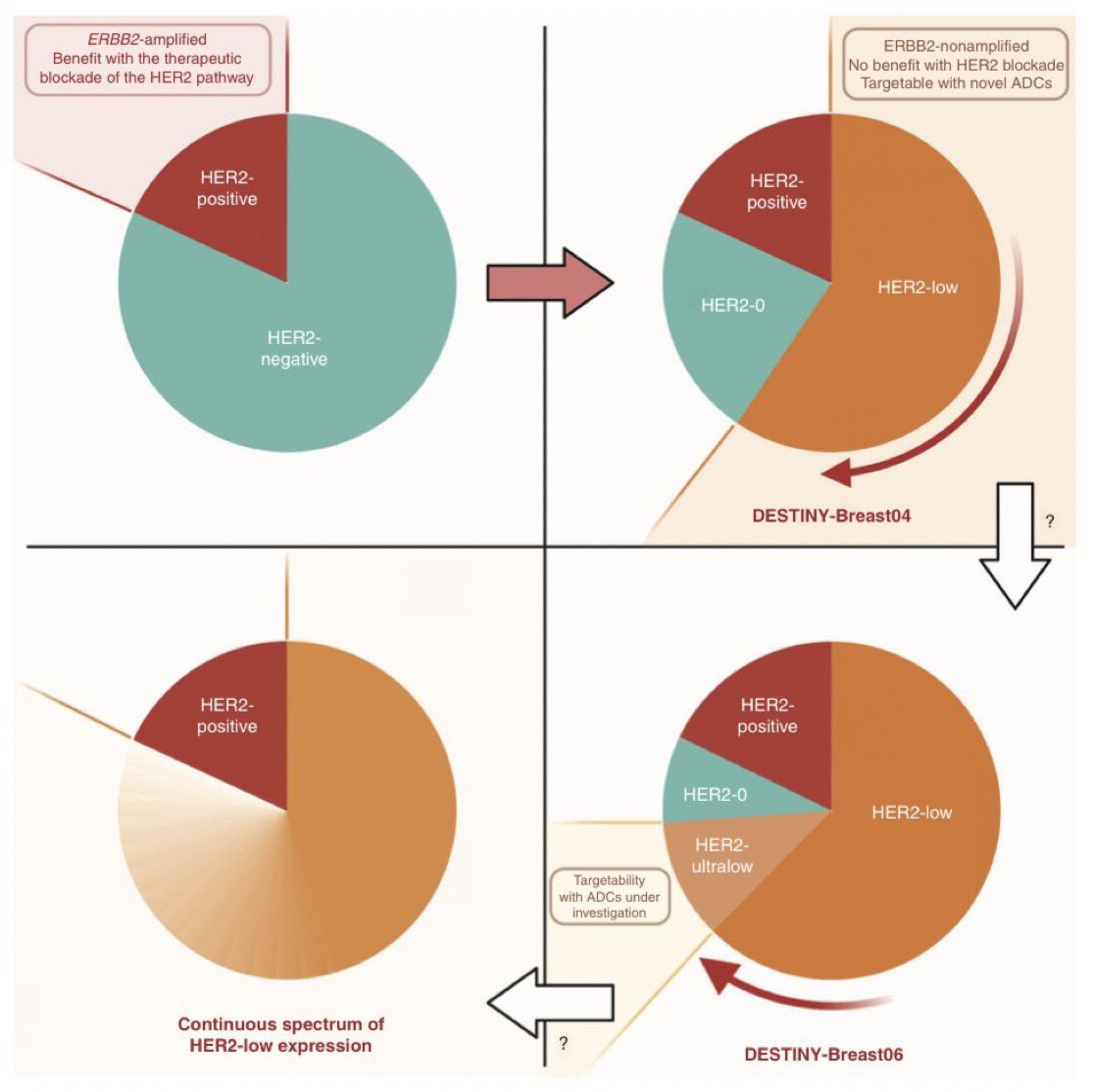
Everything changed between 2018-2022
T-DXd showed to prolong OS in #HER2low mBC, becoming a new actionable entity
What about HER2 ultralow? Still no trace: all pts with HER2 0 mBC were excluded from DB04, irrespective of absence or weak expression
3/11
ascopubs.org/doi/10.1200/JC…

T-DXd showed to prolong OS in #HER2low mBC, becoming a new actionable entity
What about HER2 ultralow? Still no trace: all pts with HER2 0 mBC were excluded from DB04, irrespective of absence or weak expression
3/11
ascopubs.org/doi/10.1200/JC…
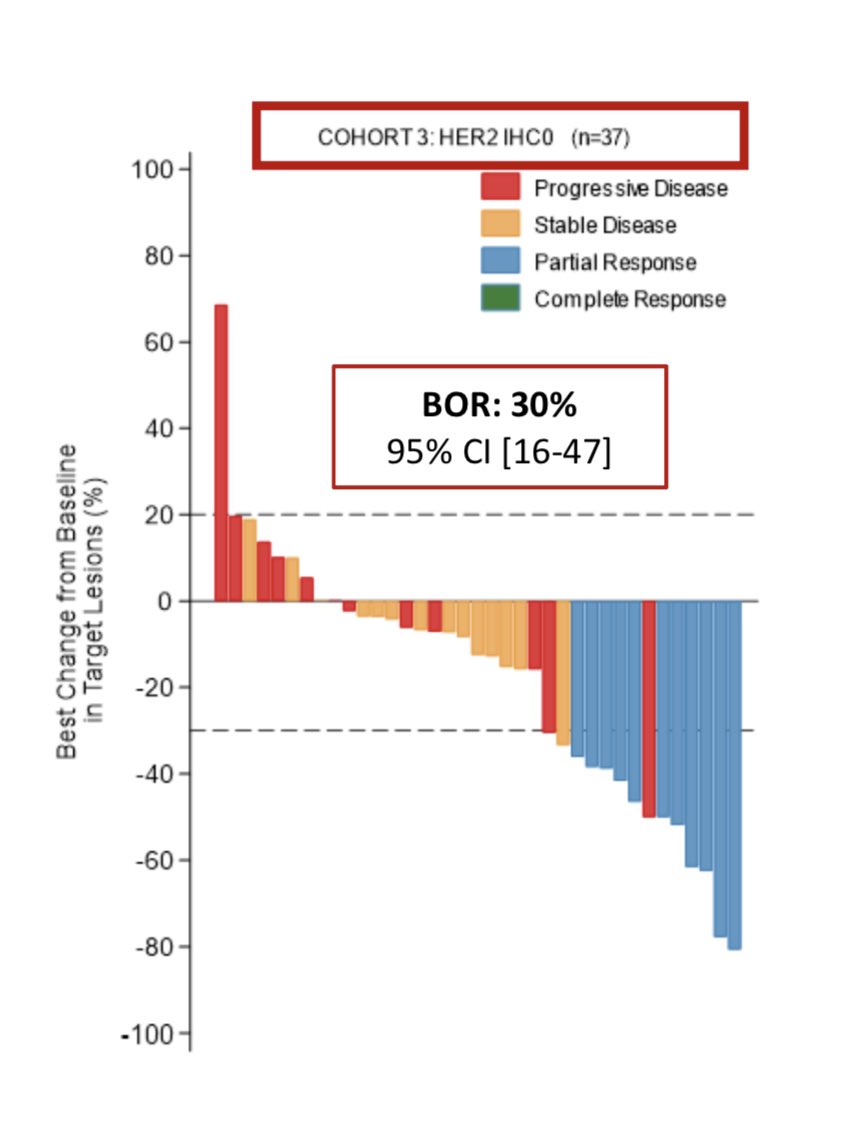
Not for long, though: a preclinical study had shown that T-DXd required only minimal HER2 expression to be active, providing the rationale to test it in some of the patients with HER2-0 disease
➡️those with incomplete, faint staining in <10% of cells
➡️aka, HER2-ultralow!
4/11
➡️those with incomplete, faint staining in <10% of cells
➡️aka, HER2-ultralow!
4/11

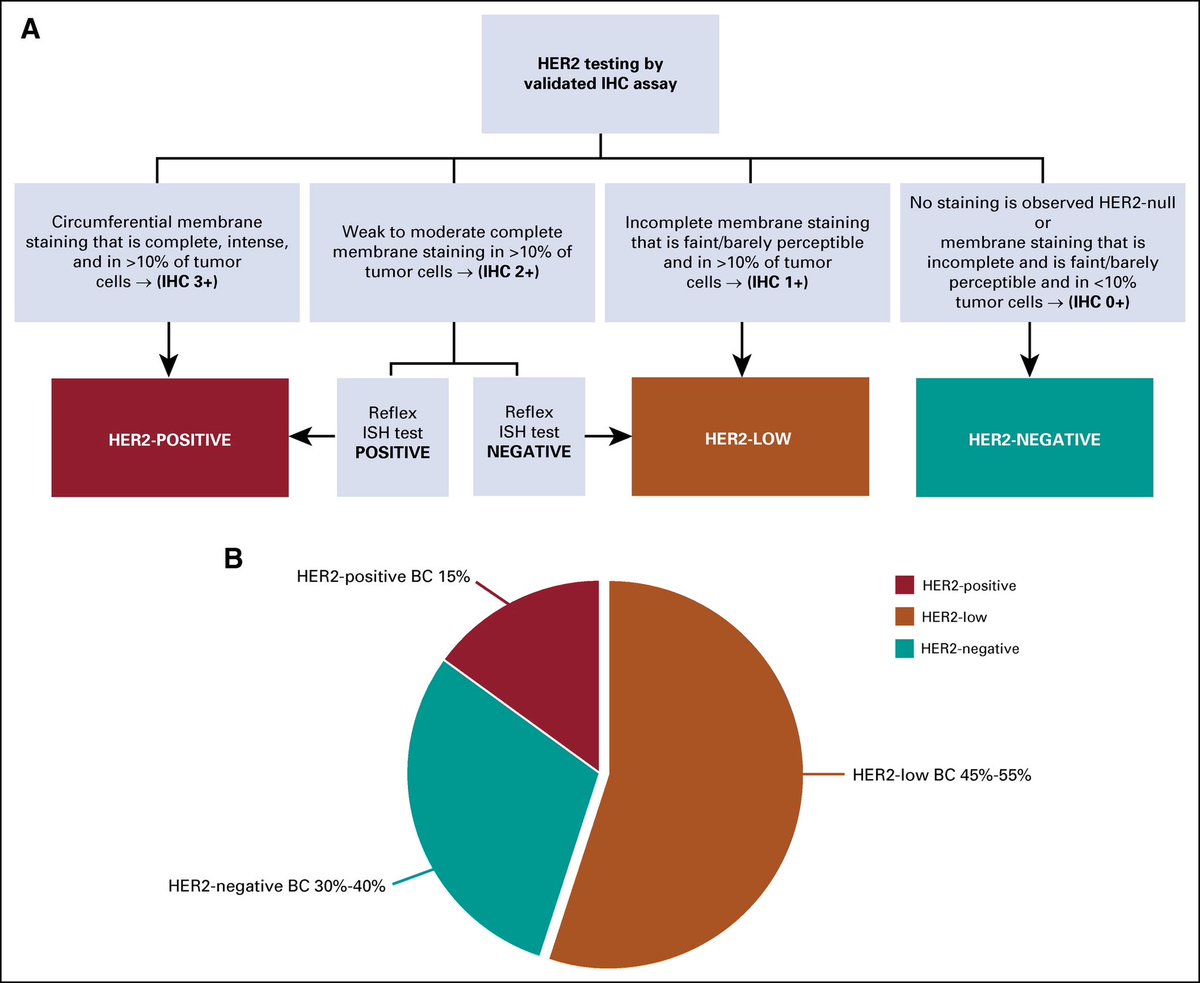
But what is the incidence of HER2-ultralow tumors?
There are only few studies answering this question. The available data suggest that more than half of traditional HER2 IHC 0 tumors can be considered HER2-ultralow
Thus, ≃20% of patients with mBC
5/11
ascopubs.org/doi/10.1200/JC…

There are only few studies answering this question. The available data suggest that more than half of traditional HER2 IHC 0 tumors can be considered HER2-ultralow
Thus, ≃20% of patients with mBC
5/11
ascopubs.org/doi/10.1200/JC…

The study that tested T-DXd in patients with HER2-ultralow breast cancer is DESTINY-Breast06
The trial included 153 patients with HR+ mBC and centrally confirmed HER2-ultralow status, randomized to T-DXd vs. chemotherapy (exploratory analysis)
6/11
The trial included 153 patients with HR+ mBC and centrally confirmed HER2-ultralow status, randomized to T-DXd vs. chemotherapy (exploratory analysis)
6/11

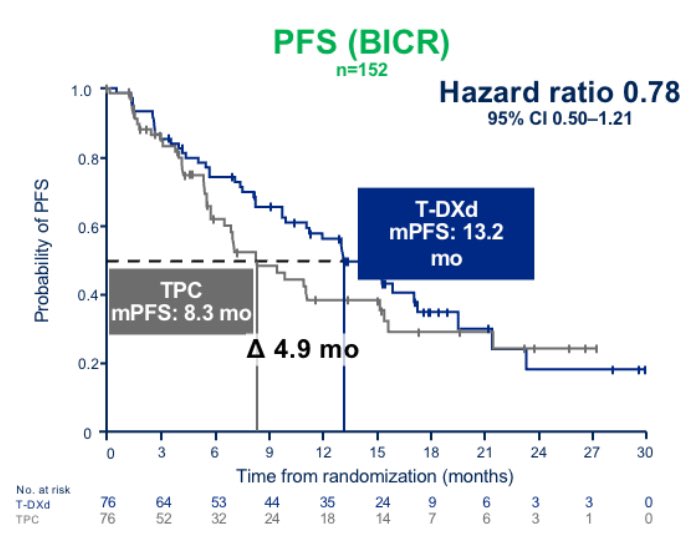
In this population, T-DXd showed a numerical improvement in PFS that appeared extremely consistent with what observed in patients with HER2-low disease
>1 year PFS with T-DXd in patients traditionally considered HER2-0 was quite remarkable to see, and unexpected for many
7/11
>1 year PFS with T-DXd in patients traditionally considered HER2-0 was quite remarkable to see, and unexpected for many
7/11
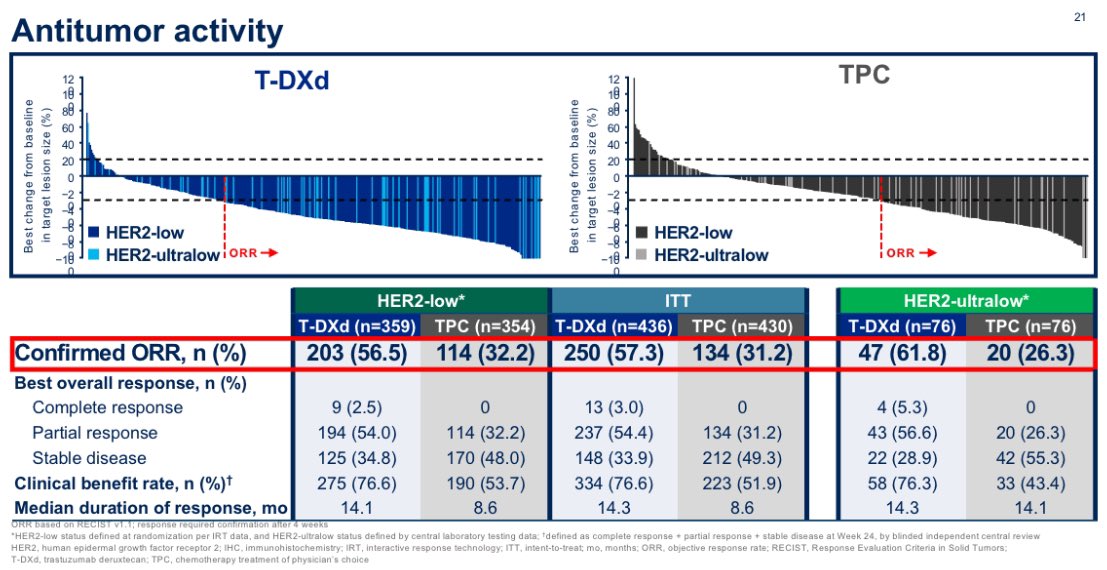
Even more strikingly, T-DXd showed higher response rate in patients with HER2-ultralow (ORR 61%) than HER2-low disease (56%) ‼️ further confirming a clear benefit in this population.
8/11
8/11
Based on these data, HER2-ultralow can now also be considered actionable in breast oncology, at least for HR+ mBC
And how do we now call HER2-0 tumors that are NOT ultralow?
The agreement within the first ESMO consensus was to call them “HER2-null”
9/11
annalsofoncology.org/article/S0923-…

And how do we now call HER2-0 tumors that are NOT ultralow?
The agreement within the first ESMO consensus was to call them “HER2-null”
9/11
annalsofoncology.org/article/S0923-…
To produce data with T-DXd in TNBC and in the “HER2-null” setting, a new trial was initiated: #DESTINYBreast15, a phase 3b trial that will enroll 250 patients with HER2-0 or HER2-low mBC
10/11
clinicaltrials.gov/study/NCT05950…
10/11
clinicaltrials.gov/study/NCT05950…
What next?
🎯 New assays!
HER2 IHC categories have never been fine tuned for HER2 ADCs. Quantitative assays in development may instead refine (and expand) our ability to select patients for ADCs
We hope to show some data soon.
Stay tuned.
/end
aacrjournals.org/cancerdiscover…

🎯 New assays!
HER2 IHC categories have never been fine tuned for HER2 ADCs. Quantitative assays in development may instead refine (and expand) our ability to select patients for ADCs
We hope to show some data soon.
Stay tuned.
/end
aacrjournals.org/cancerdiscover…
If you found this thread informative, please share! #bcsm
https://twitter.com/ptarantinomd/status/1810081196023349689
• • •
Missing some Tweet in this thread? You can try to
force a refresh