Persistent Viral Reservoirs in Post-COVID Patients: Current Evidence and Clinical Implications
🚨Viral persistence isn't fringe theory anymore!
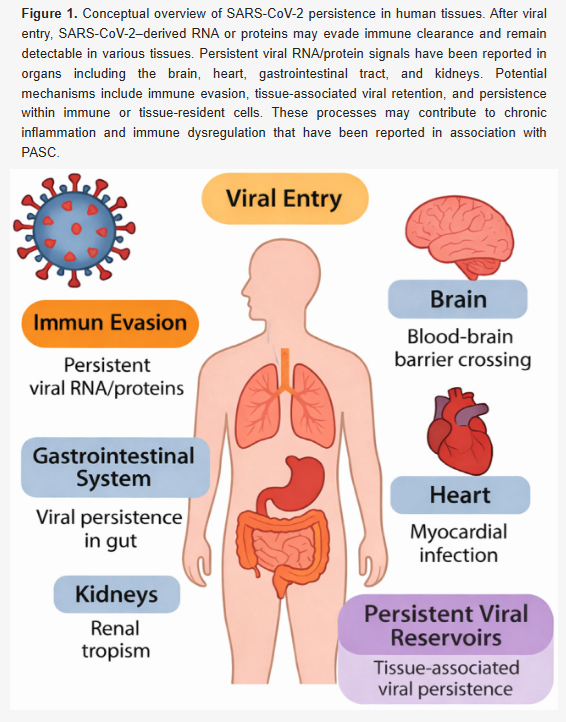
➡️This INTERESTING Korean review article examines the hypothesis that SARS-CoV-2 persists in human tissues beyond acute infection, contributing to long COVID (PASC). Authors synthesize evidence in detail from studies showing viral RNA and proteins (especially spike) detectable in organs like lungs, heart, brain, gut, and kidneys, as well as in immune cells (e.g, monocytes, macrophages) and body fluids (stool, saliva, urine), sometimes up to 15 months post-infection.
➡️Their short Long COVID Overview:
- Long COVID (PASC) features persistent or recurring symptoms (fatigue, brain fog, dyspnea, myalgia, cognitive issues) ≥4–12 weeks post-acute infection, lasting up to at least 24 months.
- ~17% of cases show no recovery and ~18% remain partially symptomatic at 24 months.
- Pathophysiology is heterogeneous and incompletely understood
- Viral persistence is one leading hypothesis,
➡️ Found evidence of SARS-CoV-2 persistence:
- Viral RNA and/or proteins (especially spike) detected in multiple organs/tissues post-acute phase: lungs (alveolar macrophages), heart (myocardial cells), brain (neural tissues), gut (intestinal epithelium), kidneys.
- Persistence in immune cells: monocytes, macrophages, dendritic cells, T cells (via phagocytosis, restricted infection, or antigen retention).
- Detection in body fluids: prolonged RNA in stool, saliva, urine (weeks to months post-onset).
- Duration: signals observed weeks to months (some studies up to 15+ months)
- Infectious virus rarely isolated post-acute phase.
- Methods: RT-PCR, immunohistochemistry, in situ hybridization, electron microscopy but rarely confirms replication-competent virus,
➡️ Mechanisms of viral persistence and immune effects:
- No classical latency (unlike herpesviruses). It involves incomplete clearance, abortive/restricted infection, antigen retention in long-lived cells (e.g, macrophages, microglia).
- Immune evasion: interferon signalling inhibition, suppressed antigen presentation.
- Persistent signals activate innate pathways (TLR3/7/8, RIG-I/MDA5), triggering NF-κB/IRF cascades → sustained cytokine production (IL-6, TNF-α), chronic inflammation, endothelial dysfunction.
- Spike protein persistence may drive autoantibody formation, molecular mimicry, epitope spreading, and autoimmune-like responses,
➡️ Clinical implications and symptom links:
- Persistent viral elements plausibly sustain multi-system inflammation, contributing to fatigue, brain fog, dyspnea, myalgia, cognitive dysfunction, cardiovascular/GI/renal issues.
- Brain: neuroinflammation linked to headaches, mood/cognitive changes.
- Gut/heart/kidney: associated with GI symptoms, myocardial inflammation, renal dysfunction.
- Vascular: endothelial spike presence → microvascular abnormalities, chest pain, tachycardia.
- Causal link to symptoms remains associative, not definitively proven,
➡️Risk factors:
- Increased risk with age, sex, comorbidities (diabetes, obesity, immunosuppression), high acute viral load, lack of vaccination.
- Immune features: impaired interferon responses, pre-existing autoreactivity, high ACE2/TMPRSS2 expression in tissues,
➡️Possible therapeutic approaches:
- No approved disease modifying treatments, current care is symptomatic.
- Investigational: extended antivirals (e.g., nirmatrelvir/ritonavir/Paxlovid up to 25 days in trials like RECOVER-VITAL), monoclonal antibodies.
- Immunomodulatory: corticosteroids, JAK inhibitors, IL-6/TNF biologics to target inflammation.
- Emerging: mesenchymal stem cells (MSC) for repair and anti-inflammation (ongoing trials, e.g, NCT04992247).
- Emphasis on personalized, multi-modal strategies based on symptom profiles,
➡️Conclusions and limitations:
- Strong evidence of persistent RNA/protein signals in tissues/cells, linked to inflammation and long COVID heterogeneity.
- Distinguishing non-replicating remnants from active infection remains challenging, causation unproven.
- Future needs: biomarkers, longitudinal studies, advanced assays (single-cell transcriptomics, negative-strand RNA), targeted trials.
‼️Persistent SARS-CoV-2 RNA and proteins are clearly detectable long after acute infection and plausibly drive chronic inflammation underlying long COVID, yet they do not prove ongoing replication or definitive causation, leaving effective eradication therapies out of reach and patients without validated cures.
‼️Reinfections receive essentially zero attention in this review, underscoring that viral persistence theories for long COVID remain overwhelmingly ffocused around unresolved remnants from the first infection, with any potential role of repeat infections left virtually unexplored here!
‼️So, in a field still lacking consensus on long COVID's root causes, this review critically elevates viral persistence from speculative hypothesis to a biologically plausible, and potentially treatable, core driver, yet its ultimate proof and therapeutic translation remain frustratingly elusive, leaving millions without targeted relief.
#AvoidSars2 #AvoidReinfections
mdpi.com/2673-8112/6/3/…
🚨Viral persistence isn't fringe theory anymore!
➡️This INTERESTING Korean review article examines the hypothesis that SARS-CoV-2 persists in human tissues beyond acute infection, contributing to long COVID (PASC). Authors synthesize evidence in detail from studies showing viral RNA and proteins (especially spike) detectable in organs like lungs, heart, brain, gut, and kidneys, as well as in immune cells (e.g, monocytes, macrophages) and body fluids (stool, saliva, urine), sometimes up to 15 months post-infection.
➡️Their short Long COVID Overview:
- Long COVID (PASC) features persistent or recurring symptoms (fatigue, brain fog, dyspnea, myalgia, cognitive issues) ≥4–12 weeks post-acute infection, lasting up to at least 24 months.
- ~17% of cases show no recovery and ~18% remain partially symptomatic at 24 months.
- Pathophysiology is heterogeneous and incompletely understood
- Viral persistence is one leading hypothesis,
➡️ Found evidence of SARS-CoV-2 persistence:
- Viral RNA and/or proteins (especially spike) detected in multiple organs/tissues post-acute phase: lungs (alveolar macrophages), heart (myocardial cells), brain (neural tissues), gut (intestinal epithelium), kidneys.
- Persistence in immune cells: monocytes, macrophages, dendritic cells, T cells (via phagocytosis, restricted infection, or antigen retention).
- Detection in body fluids: prolonged RNA in stool, saliva, urine (weeks to months post-onset).
- Duration: signals observed weeks to months (some studies up to 15+ months)
- Infectious virus rarely isolated post-acute phase.
- Methods: RT-PCR, immunohistochemistry, in situ hybridization, electron microscopy but rarely confirms replication-competent virus,
➡️ Mechanisms of viral persistence and immune effects:
- No classical latency (unlike herpesviruses). It involves incomplete clearance, abortive/restricted infection, antigen retention in long-lived cells (e.g, macrophages, microglia).
- Immune evasion: interferon signalling inhibition, suppressed antigen presentation.
- Persistent signals activate innate pathways (TLR3/7/8, RIG-I/MDA5), triggering NF-κB/IRF cascades → sustained cytokine production (IL-6, TNF-α), chronic inflammation, endothelial dysfunction.
- Spike protein persistence may drive autoantibody formation, molecular mimicry, epitope spreading, and autoimmune-like responses,
➡️ Clinical implications and symptom links:
- Persistent viral elements plausibly sustain multi-system inflammation, contributing to fatigue, brain fog, dyspnea, myalgia, cognitive dysfunction, cardiovascular/GI/renal issues.
- Brain: neuroinflammation linked to headaches, mood/cognitive changes.
- Gut/heart/kidney: associated with GI symptoms, myocardial inflammation, renal dysfunction.
- Vascular: endothelial spike presence → microvascular abnormalities, chest pain, tachycardia.
- Causal link to symptoms remains associative, not definitively proven,
➡️Risk factors:
- Increased risk with age, sex, comorbidities (diabetes, obesity, immunosuppression), high acute viral load, lack of vaccination.
- Immune features: impaired interferon responses, pre-existing autoreactivity, high ACE2/TMPRSS2 expression in tissues,
➡️Possible therapeutic approaches:
- No approved disease modifying treatments, current care is symptomatic.
- Investigational: extended antivirals (e.g., nirmatrelvir/ritonavir/Paxlovid up to 25 days in trials like RECOVER-VITAL), monoclonal antibodies.
- Immunomodulatory: corticosteroids, JAK inhibitors, IL-6/TNF biologics to target inflammation.
- Emerging: mesenchymal stem cells (MSC) for repair and anti-inflammation (ongoing trials, e.g, NCT04992247).
- Emphasis on personalized, multi-modal strategies based on symptom profiles,
➡️Conclusions and limitations:
- Strong evidence of persistent RNA/protein signals in tissues/cells, linked to inflammation and long COVID heterogeneity.
- Distinguishing non-replicating remnants from active infection remains challenging, causation unproven.
- Future needs: biomarkers, longitudinal studies, advanced assays (single-cell transcriptomics, negative-strand RNA), targeted trials.
‼️Persistent SARS-CoV-2 RNA and proteins are clearly detectable long after acute infection and plausibly drive chronic inflammation underlying long COVID, yet they do not prove ongoing replication or definitive causation, leaving effective eradication therapies out of reach and patients without validated cures.
‼️Reinfections receive essentially zero attention in this review, underscoring that viral persistence theories for long COVID remain overwhelmingly ffocused around unresolved remnants from the first infection, with any potential role of repeat infections left virtually unexplored here!
‼️So, in a field still lacking consensus on long COVID's root causes, this review critically elevates viral persistence from speculative hypothesis to a biologically plausible, and potentially treatable, core driver, yet its ultimate proof and therapeutic translation remain frustratingly elusive, leaving millions without targeted relief.
#AvoidSars2 #AvoidReinfections
mdpi.com/2673-8112/6/3/…
• • •
Missing some Tweet in this thread? You can try to
force a refresh