
Distracted by the other EMA thing, I'm only just catching up with the 218-page EMA assessment report for the J&J vaccine ema.europa.eu/en/documents/a…
For background, my summary from the 62-page FDA report is here absolutelymaybe.plos.org/2021/03/16/com…
Tweeting about what's new or different...1/n
For background, my summary from the 62-page FDA report is here absolutelymaybe.plos.org/2021/03/16/com…
Tweeting about what's new or different...1/n

...As usual, in-depth look at quality of manufacturing. "No raw materials of human or animal origin are used" in the final production. The cell line used to optimise production of the vaccine vector is PER.C6, which has already been studied for safety...2/n
...Current batches manufactured mostly in Leiden & emerging in Baltimore: they've shown consistency in the European manufacture - EMA wants them to follow-up with data showing the Baltimore production is comparable (this is all standard) ...3/n
...From first scan, looks as though J&J have publications/preprints of all the preclinical animal studies (or close to), but there's quite a bit of detail here. Plus a reproductive toxicity study in rabbits: no cause for concern on reproduction or fertility ...4/n
...If you're interested in their early phase immunogenicity study, there might be some more detail here than there was in the publication - that's here: nejm.org/doi/10.1056/NE… ...5/n
...Something FDA didn't report: there was an increase in SARS-CoV-2 infections at 2 sites in Brazil. Because of concerns about variants, an originally unplanned analysis of immunogenicity was done for people randomly selected from there & other sites in Brazil, SA, USA ...6/n
... All up, that was 188 people in Brazil, 118 in SA, 74 in the US. Results are shown (Table 11): antibody & responder rates were similar across the sites. A higher rate of participants from Brazil had signs of previous SARS-CoV-2 infection ...7/n
...If you're interested in their ongoing immunogenicity studies internationally, there's some detail in here on those. (Perhaps of particular interest to people interested in the 2-dose issue.) ...8/n
... Just an aside here - the EMA is consolidating their position as the producer of the best evaluations of these vaccines with this report - it's very impressive (& I'm not halfway through yet)...9/n
...Where was I? Oh yeah - the 2 doses question. Although people had Ad26 (the viral vector) antibodies, "it did not suggest an apparent negative impact of antiAd26 vector immunity induced by the first vaccine dose on the post-dose 2...responses"...10/n
...Onto the phase 3 ENSEMBLE trial: this is the same data as in the FDA report. They have a bit on statistics, which FDA didn't (probably covered in the protocol:jnj.com/coronavirus/en… ) ...11/n
... After 28 days, they conclude, efficacy wasn't so different between SA, Latin America, USA. Onset was later for SA ⬇️ "It might be hypothesised that Ab and/or T cell responses of higher magnitude are needed for
protection against the SA variant."...12/n
protection against the SA variant."...12/n
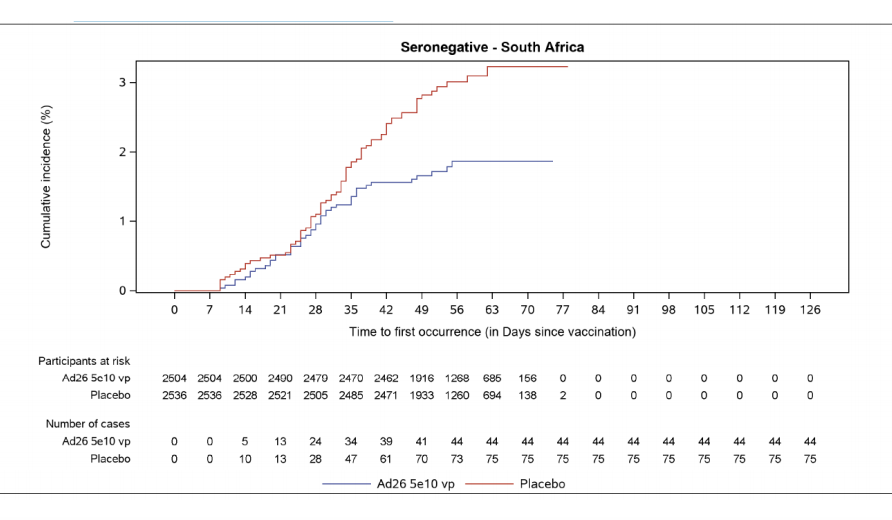
...The majority of Covid-19 classified severe was based on self-measured oxygen saturation measures ...13/n
...About 2% of people had 1 or more solicited adverse events rated severe, mostly judged related to the vaccine - but only about half the solicited adverse events overall were judged related. (My explainer on the language here & what will follow): absolutelymaybe.plos.org/2021/02/21/a-r…) ...14/n
...The judgments on serious adverse events (SAEs) seem the same as for the FDA (an exception will follow). (Of note given recent events: none of the thromboembolic events described here are like the ones under investigation in Europe for another vax at the moment) ...15/n
...Exception is not related to this specific vaccine, but to a previous vaccine: there had been some people who had incidental HIV infections in a trial of a previous vaccine. For those who knew this & were wondering: there were no incidental HIV infections in this trial ...16/n
...No safety concerns were noted for the 601 people living with HIV who participated in this trial, but it's too small a group to draw conclusions ...17/n
...They looked at safety data on pregnancy for the few (4) in this trial, but also for previous J&J vaccine: "did not reveal any safety concern related to Ad26-based vaccine exposure during pregnancy"...18/n
...Because so few people had mild Covid-19 in this trial, EMA concludes efficacy against Covid-19 ...19/19
And now we go to breaking news on the Oxford-AstraZeneca vaccine from the US...
And now we go to breaking news on the Oxford-AstraZeneca vaccine from the US...

• • •
Missing some Tweet in this thread? You can try to
force a refresh




