FDA Director General Eric Domingo says they have approved the application of a hospital for the compassionate special use permit of ivermectin
Domingo: Meron na kaming na-grant dahil ang ivermectin ay isang investigational product at alam naman natin na may mga clinical trials ongoing dito para gamitin against COVID-19...
Domingo: ...so ang pagkakaalam ko may isang ospital sa na nag-apply ng compassionate special permit for the use of ivermectin at ito ay na-grant ngayong araw na ito.
Domingo: Ito lang naman po ang laging sinasabi ng FDA, hindi kami kontra sa ivermectin, kailangan lang po na marehistro ang produkto at dumaan lamang po sa tamang proseso ng pagsiguro ng quality ng gamot na makakarating sa tao.
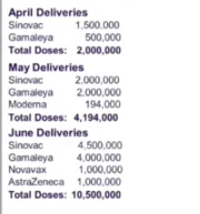
FDA recommends stopping the administration of remaining on-hand Astrazeneca vaccines to individuals aged 60 and below following reports that it can cause rare blood clots
Domingo: We asked DOH, kung meron pang natitirang Astrazeneca vaccine, siguro wag muna nating gamitin sa mga people below 60 years old until we get clearer evidence and guidance from WHO at sa ating mga expert.
Domingo: Actually wala na po tayong Astrazeneca at this time, dahil talagang naubos na natin ito at pailan-ilan na lang daw, at ang susunod na ineexpect ay within one month pa. So that will give us time to study the evidence and to see kung magkakaroon tayo ng panibagong guidance
• • •
Missing some Tweet in this thread? You can try to
force a refresh