Excited to share our work by @BenIsraelow et al published today. We asked what are the roles of antibodies vs. T cells in controlling primary infection, reinfection, and vaccine-mediated protection? (1/n)
science.org/doi/10.1126/sc…
science.org/doi/10.1126/sc…
First, we asked if B cells are needed to control primary infection. We used muMT mice (devoid of B cells) transduced with AAV-hACE2. These mice had only a slight delay in viral clearance. Thus B cells are not necessary for controlling primary SARS-CoV-2 infection. (2/n) 

However, in mice that have neither T cells nor B cells (RAG-/-), SARS-CoV-2 persisted with no sign of clearance. Thus, innate immunity is insufficient, and adaptive immunity is required to control primary infection. (3/n) 

These findings have important implications in the persistent SARS-CoV-2 infection we see in immunocompromised patients, and imply that defects in T and B cell immunity predispose people for chronic COVID infection. (4/n)
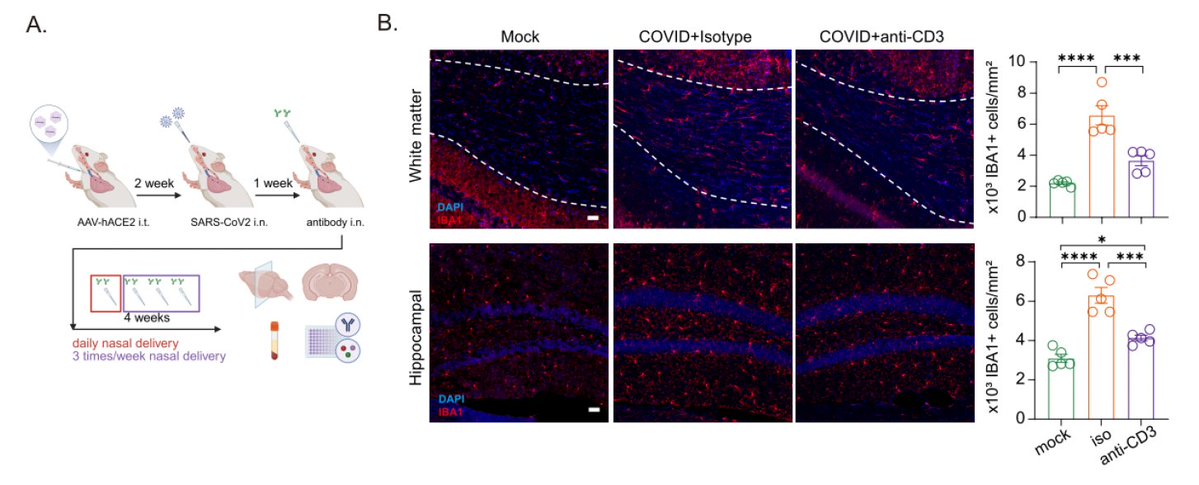
Next, we wanted to know if CD4 vs. CD8 T cells are required for clearance of primary SARS-CoV-2 infection. Depletion of CD4 or CD8 had moderate effects on loss of viral control. However, depletion of both CD4 and CD8 T cells resulted enhanced viral replication. (5/n) 

What is the role of CD4 T cells in primary SARS-CoV-2 clearance? It turns out that the role of CD4 T cells is mainly to support antibody production (panel D), because in the absence of B cells (panel C), CD4 depletion had little impact on viral control. (6/n) 

Are T cells or Ab sufficient to control primary infection with SARS-CoV-2? @BenIsraelow collected either sera (Ab) or T cells from infected mice at 14 days, and transfer to RAG-/- host, which were challenged with virus. Turns out that Ab > T cells in controlling virus. (7/n) 

Next, we asked whether mRNA vax or natural SARS-CoV-2 infection establishes lung-resident CD8 T cells. While both induced comparable circulating CD8 T cells (IV+), natural infection >> vax in establishing tissue-resident CD8 T cells (IV-). #mucosalimmunity (8/n) 

How well does the mRNA vax or primary infection protect against VOC, and how much of that depends on CD8 T cells? Great news is that the mRNA vax or prior infection protected 100% of mice, even after CD8 T depletion at the time of challenge. (9/n) 

In the lungs of these mice, we found that both mRNA vax mice and convalescent mice were completely protected from disease with original strain (WA1) and the B.1.351 virus. Even without CD8 T, all vax & convalescent mice eliminated infectious virus (G).(10/n) 

Further, by immunizing with varying doses of the mRNA vax, @BenIsraelow found a strong correlation between anti-spike IgG levels, neutralizing Ab and protection against COVID-19 disease. (11/n) 

In conclusion, while T cells were sufficient for the clearance of primary infection, they were not required for protection against reinfection or vaccine-mediated protection. (12/n)
We did not test the sufficiency of T cells in vaccine-mediated protection. However, a very nice study by @Masopust_Vezys shows the promise of adding T cell antigens to vaccines. (13/n)
pubmed.ncbi.nlm.nih.gov/34193597/
pubmed.ncbi.nlm.nih.gov/34193597/
While we did not test the Delta variant, with its high viral load and transmission capacity, vaccines that induce mucosal immunity (TRM, IgA) may become important to better prevent infection and transmission. (14/n)
news.yale.edu/2021/09/02/imm…
news.yale.edu/2021/09/02/imm…
As usual, this project was enabled by a great team work. Led by the amazing @BenIsraelow @YaleIDFellows with key collaborators @tianyangmao @sneakyvirus1 @ericsongg @biobridget & @SaadOmer3 🙏🏼 (end)
• • •
Missing some Tweet in this thread? You can try to
force a refresh