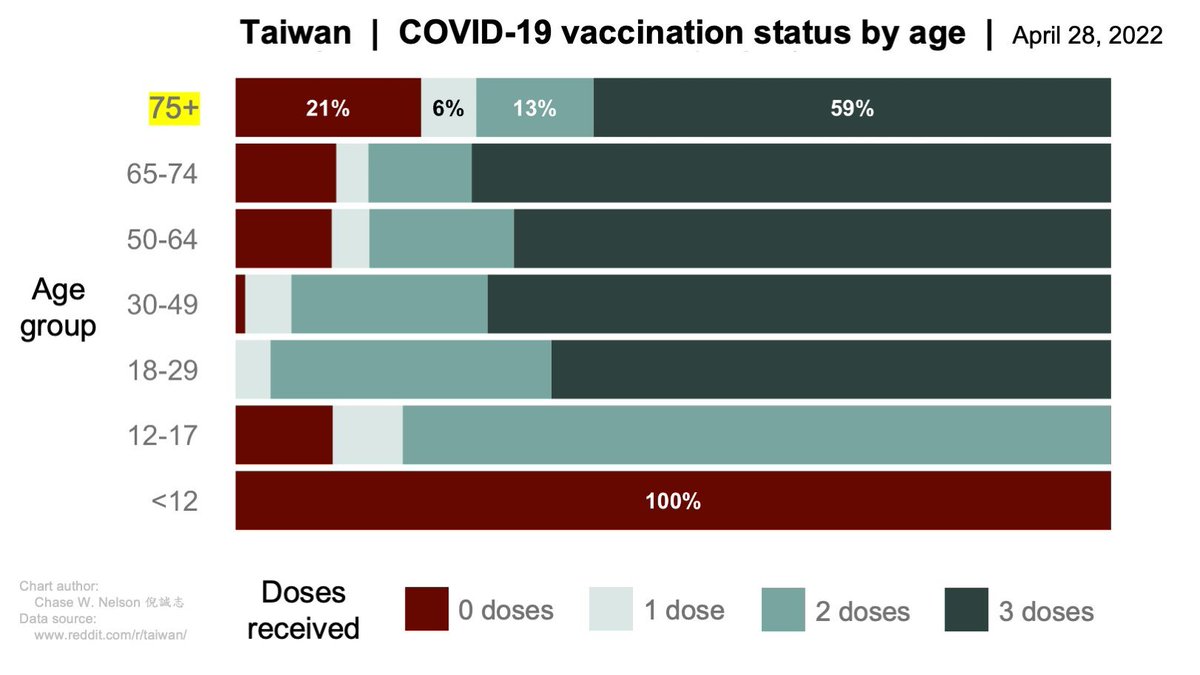
FDA limits #JnJ vaccine to only those who can't receive other vaccines due to risk of thrombocytopenia syndrome (TTS), aka vaccine-induced thrombocytic thrombocytopenia (VITT), aka the rare vaccine clot syndrome we've known about since 3/2021
cnn.com/2022/05/05/hea…
cnn.com/2022/05/05/hea…
The cause of this rare effect is the development of anti-PIF4 antibodies, but the mechanism of that is, surprisingly, still unknown. The same effect is seen in AZ, and J&J is Ad26 while AZ is a chimp Ad, so it's likely common to many-most primate adenoviruses.
It's been suggested Ad capsids bind and precipitate PIF4, triggering an immune reaction against PIF4. However a simpler mechanism that I have yet to see ruled out would be simply the production of antibodies to some Ad capsid protein that in rare cases cross-react to PIF4
The practical effect of today's FDA action is to remove J&J as a booster option for anyone still entertaining that.
That will leave Novavax as the only RNA alternative for boosters in the US, if FDA approves. Wonder how work on the Omicron-adapted Novavax booster is going.
That will leave Novavax as the only RNA alternative for boosters in the US, if FDA approves. Wonder how work on the Omicron-adapted Novavax booster is going.
• • •
Missing some Tweet in this thread? You can try to
force a refresh