$NWBO #DCVaxL New name #murcidencel
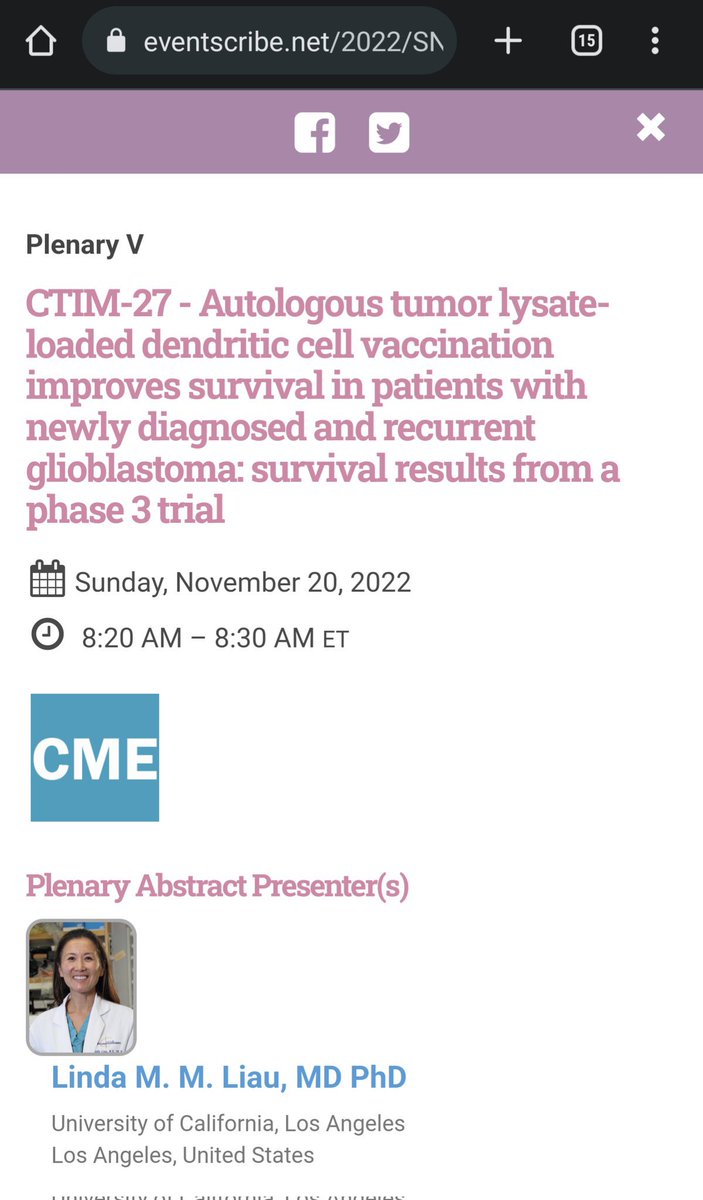
SNO Abstract, just released, for presentation by #DrLindaLiau on November 20th:
Background: Standard of care (SOC) and patient survival in glioblastoma have changed little in the past 17 years.
SNO Abstract, just released, for presentation by #DrLindaLiau on November 20th:
Background: Standard of care (SOC) and patient survival in glioblastoma have changed little in the past 17 years.
We evaluated in a phase 3 trial whether adding an autologous tumor lysate-loaded dendritic cell vaccine (murcidencel) to SOC extends survival. Patients and
Methods: Newly diagnosed glioblastoma patients were randomized 2:1 to either murcidencel or placebo.
Methods: Newly diagnosed glioblastoma patients were randomized 2:1 to either murcidencel or placebo.
Under a crossover design, all patients could receive murcidencel following tumor recurrence. All parties remained blinded regarding treatments before recurrence. Patients thus received murcidencel at new diagnosis (nGBM) or at recurrence (rGBM) following crossover from placebo.
The primary and secondary endpoints compare overall survival (OS) with contemporaneous, matched external controls. Four sets of analyses were conducted to ensure rigorous matching of the controls, reduce biases, and confirm the robustness of the results.
Results: 331 patients were enrolled. With the crossover, 89% received murcidencel. Median OS (mOS) for nGBM patients (n = 232) was 19.3 months from randomization (22.4 months from surgery) with murcidencel vs. 16.5 months from randomization in the controls (HR = 0.80, p = 0.002).
Survival at 48 months from randomization was 15.7% vs. 9.9%, and at 60 months was 13% vs. 5.7%. For rGBM (n = 64), mOS was 13.2 months from relapse vs. 7.8 months in the controls (HR = 0.58, p < 0.001).
Survival at 24 months post-recurrence was 20.7% vs. 9.6%, and at 30 months post-recurrence was 11.1% vs 5.1%.
In nGBM patients with methylated MGMT (n = 90), mOS was 30.2 months from randomization (33 months from surgery) with murcidencel vs. 21.3 months from randomization in the controls (HR = 0.74, p = 0.027).
The treatment was well tolerated, with only 5 serious adverse events deemed at least possibly related to the vaccine.
Conclusion: Clinically meaningful and statistically significant survival extension was seen in both nGBM and rGBM patients treated with murcidencel and SOC compared with contemporaneous, matched external controls who received SOC alone.
Upcoming presentation for Dr. Liau, for NWBO, at the SNO (Society for Neuro-Oncology) conference, November 20th:

Thread by @biosectinvestor :
https://twitter.com/biosectinvestor/status/1591088744140255234?s=20&t=0VdDESFM94BZsbbWjxjYUw
$NWBO #DCVaxL #murcidencel
Investors don't know what journal will likely publish the $NWBO #DCVaxL #murcidencel final Phase 3 results for #Glioblastoma, but speculation is rampant:
@threadreaderapp unroll
• • •
Missing some Tweet in this thread? You can try to
force a refresh




