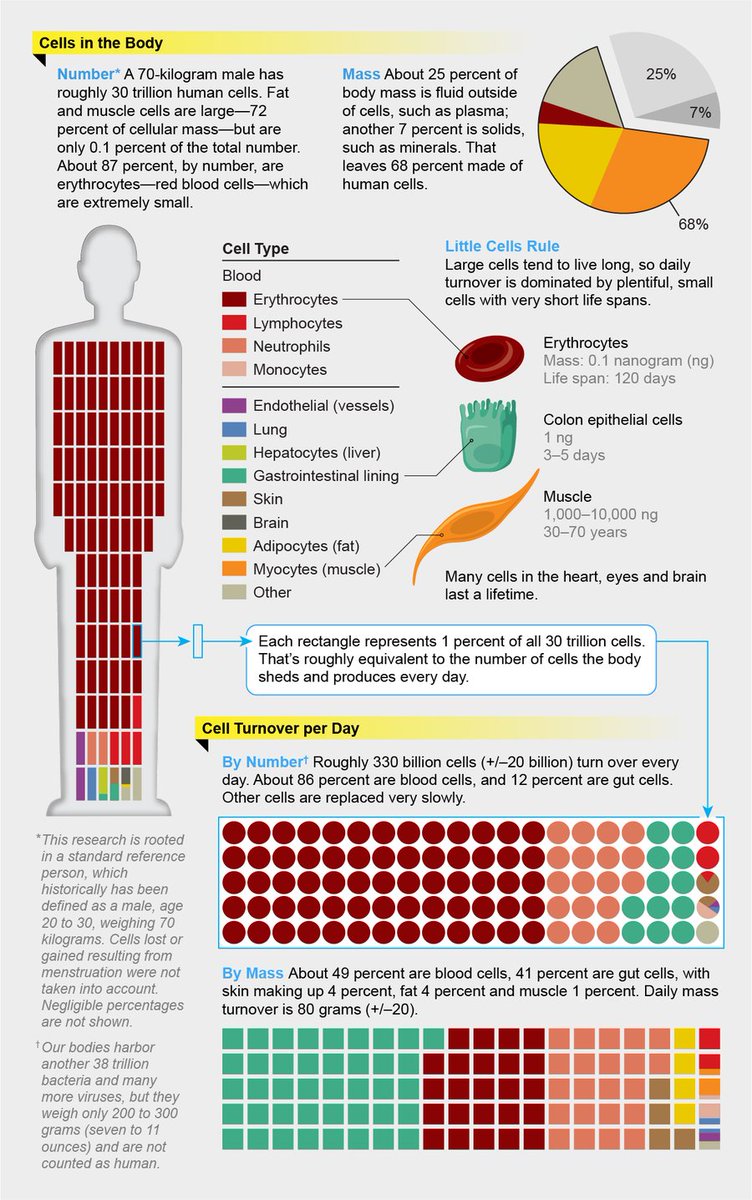
AIRBORNE transmission of respiratory VIRUSES
(in few graphs, layman terms)
"In contrast to droplets, aerosols can linger in air for hours and travel beyond 1 to 2 m from the infected individual who exhales them, causing new infections at both short and long ranges."
(in few graphs, layman terms)
"In contrast to droplets, aerosols can linger in air for hours and travel beyond 1 to 2 m from the infected individual who exhales them, causing new infections at both short and long ranges."
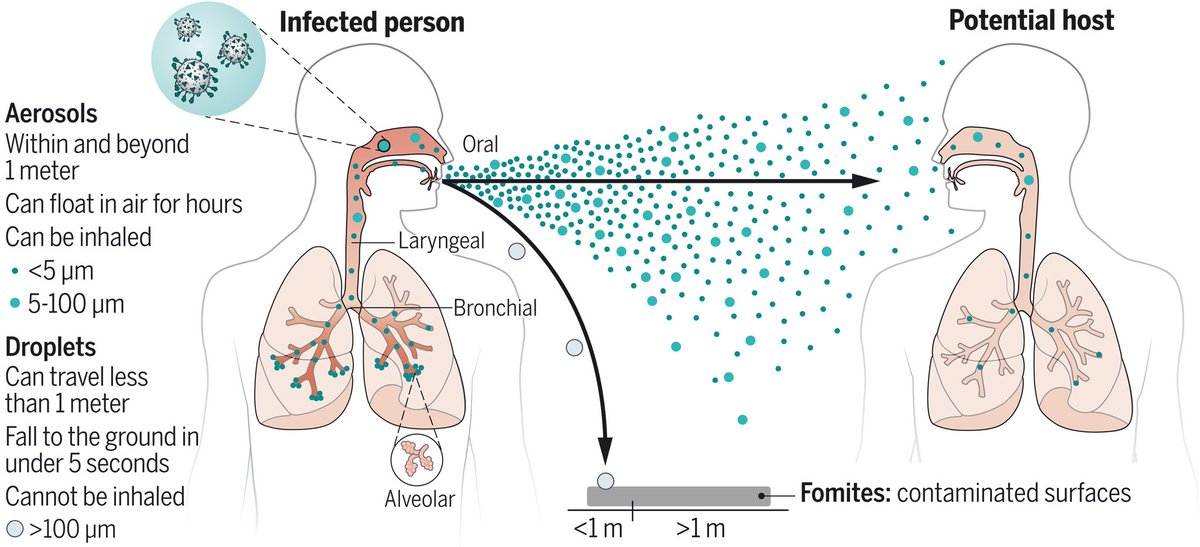
2) "Phases involved in the airborne transmission of virus-laden aerosols include (i) generation and exhalation; (ii) transport; and (iii) inhalation, deposition, and infection." 

3) "The behavior and fate of virus-laden aerosols are inherently governed by their characteristic properties, including physical size, viral load, infectivity, other chemical components in the aerosol, electrostatic charge, pH, and the air-liquid interfacial properties" 

3) "How long can aerosols linger in air?
For example, the time required for an aerosol of 100, 5, or 1 μm to fall to the ground (or surfaces) from a height of 1.5 m is 5 s, 33 min, or 12.2 hours, respectively."
For example, the time required for an aerosol of 100, 5, or 1 μm to fall to the ground (or surfaces) from a height of 1.5 m is 5 s, 33 min, or 12.2 hours, respectively."

4) "The movement of aerosols is more strongly influenced by airflow direction and pattern, type of ventilation, and air filtration and disinfection" 

5) "Small aerosols tend to deposit in the tracheobronchial and alveolar regions on the basis of gravitational sedimentation and Brownian diffusion." 

6) Thanks for reading 🙏
Source : science.org/doi/10.1126/sc…
FYI
@MeetJess @white_bite @DavidJoffe64 @1goodtern @HarrySpoelstra
Source : science.org/doi/10.1126/sc…
FYI
@MeetJess @white_bite @DavidJoffe64 @1goodtern @HarrySpoelstra

• • •
Missing some Tweet in this thread? You can try to
force a refresh