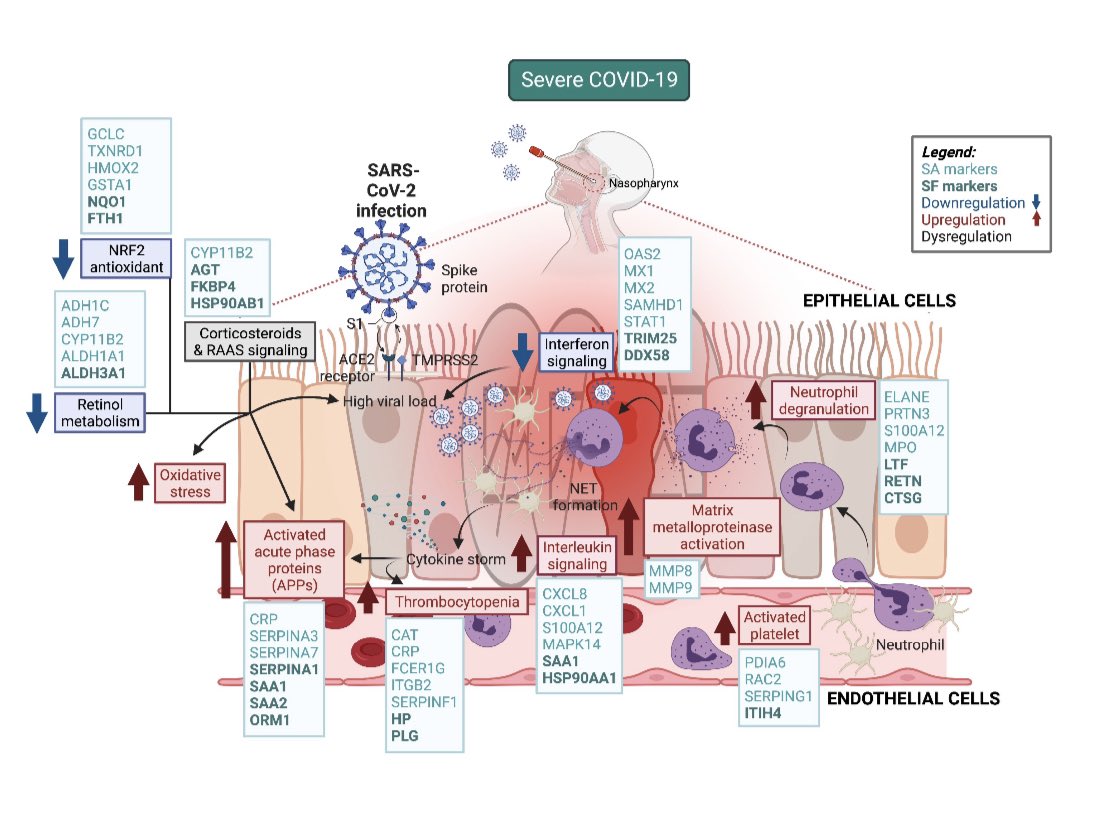
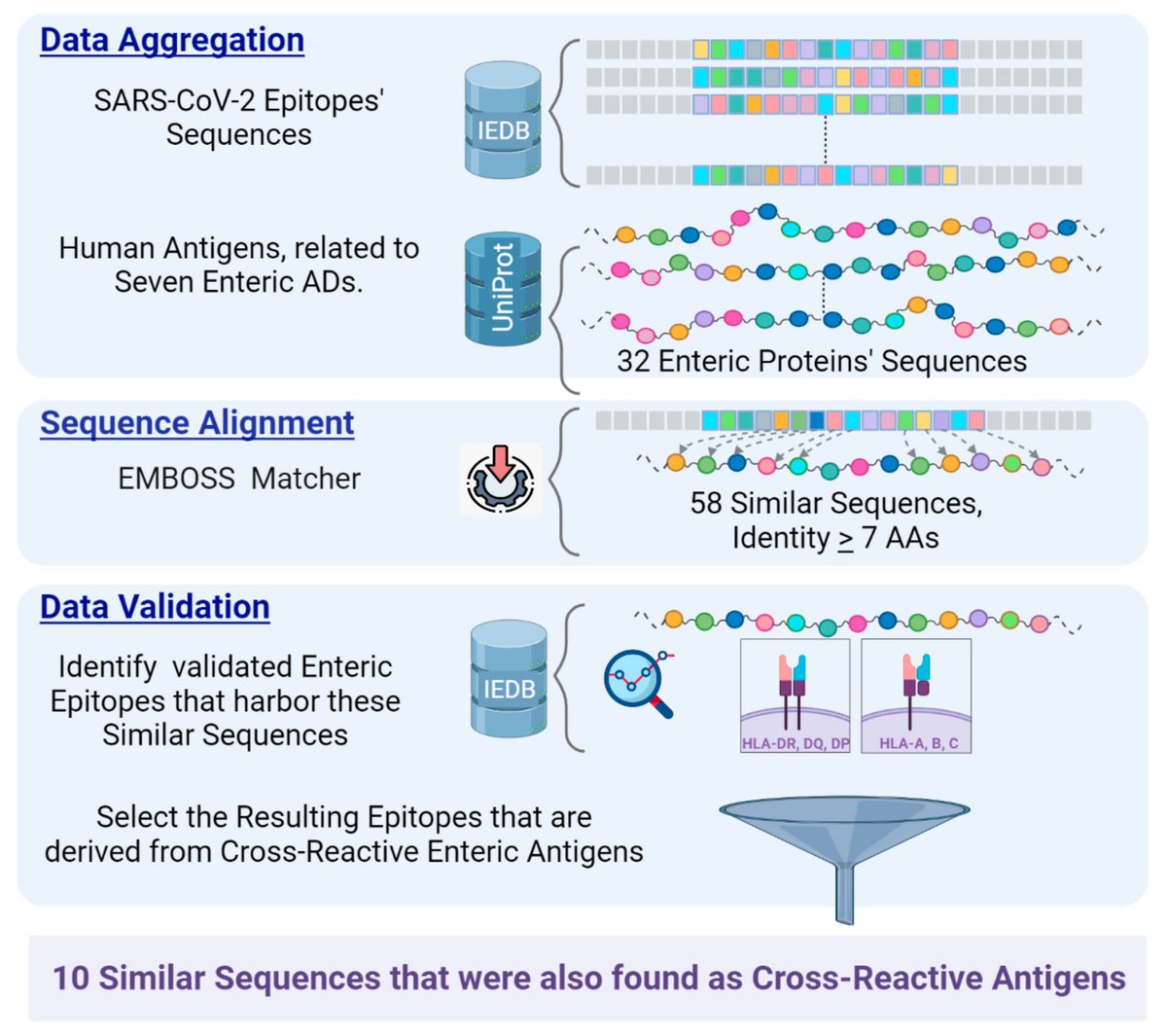
The evolution of SARS-CoV-2 has stunned the virologists & evolutionary biologists all over the world. It has even surpassed the hyper-mutability of the influenza’s H3N2 strain too!
In a new preprint, the evolution of the SARS2 Spike between Dec’23 & Feb’23 is analysed... 1/
In a new preprint, the evolution of the SARS2 Spike between Dec’23 & Feb’23 is analysed... 1/
To summarize the major lineages of SARS-CoV-2 & their Spike evolution during this period, a phylogenetic tree & median-joining network were reconstructed.
By the end of 2022, new variants such as BQ.1.1.10, BA.4.6.3, XBB, & CH.1.1, emerged with higher fitness than BA.5.
2/
By the end of 2022, new variants such as BQ.1.1.10, BA.4.6.3, XBB, & CH.1.1, emerged with higher fitness than BA.5.
2/
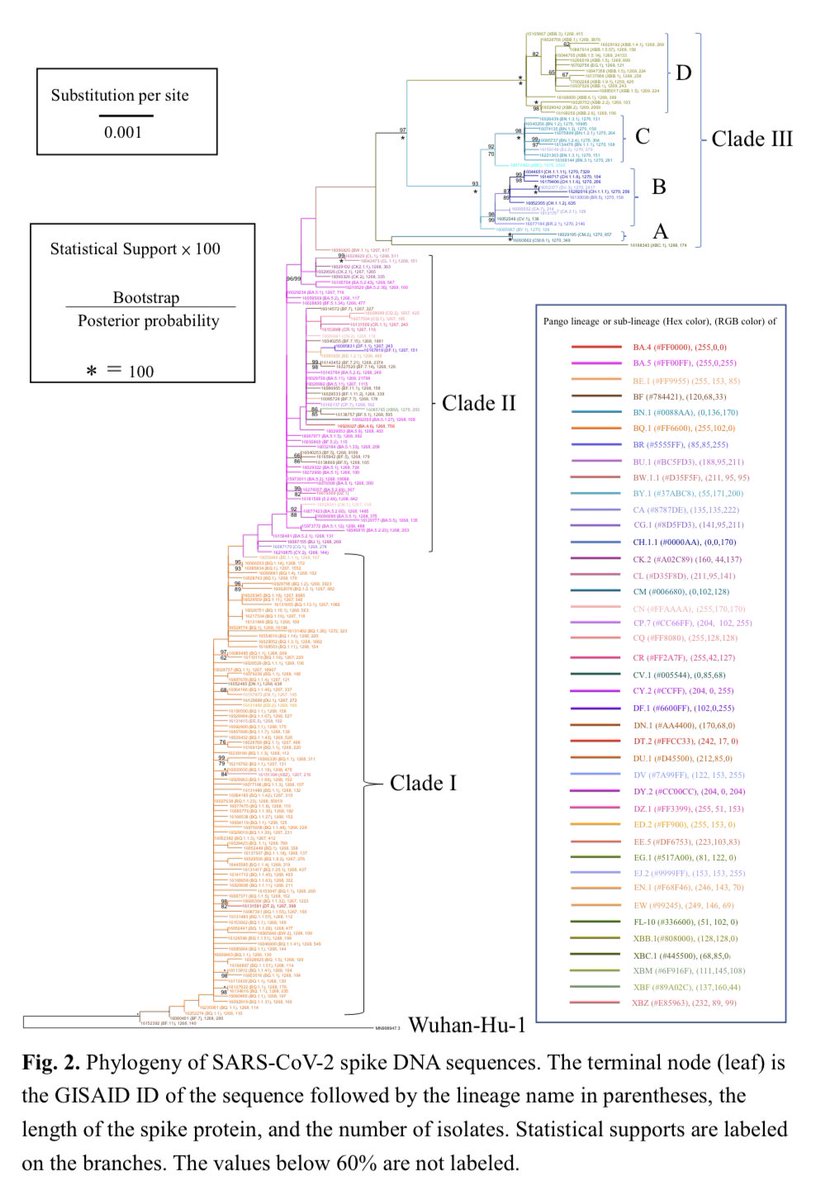
The phylogenetic tree of the spike DNA sequences revealed that the majority of variants belonged to three major lineages: BA.2 (BA.1.1.529.2), BA.5 (BA.1.1.529.5), and XBB. The median network showed that these lineages had at least six major diversifying centers. 3/ 
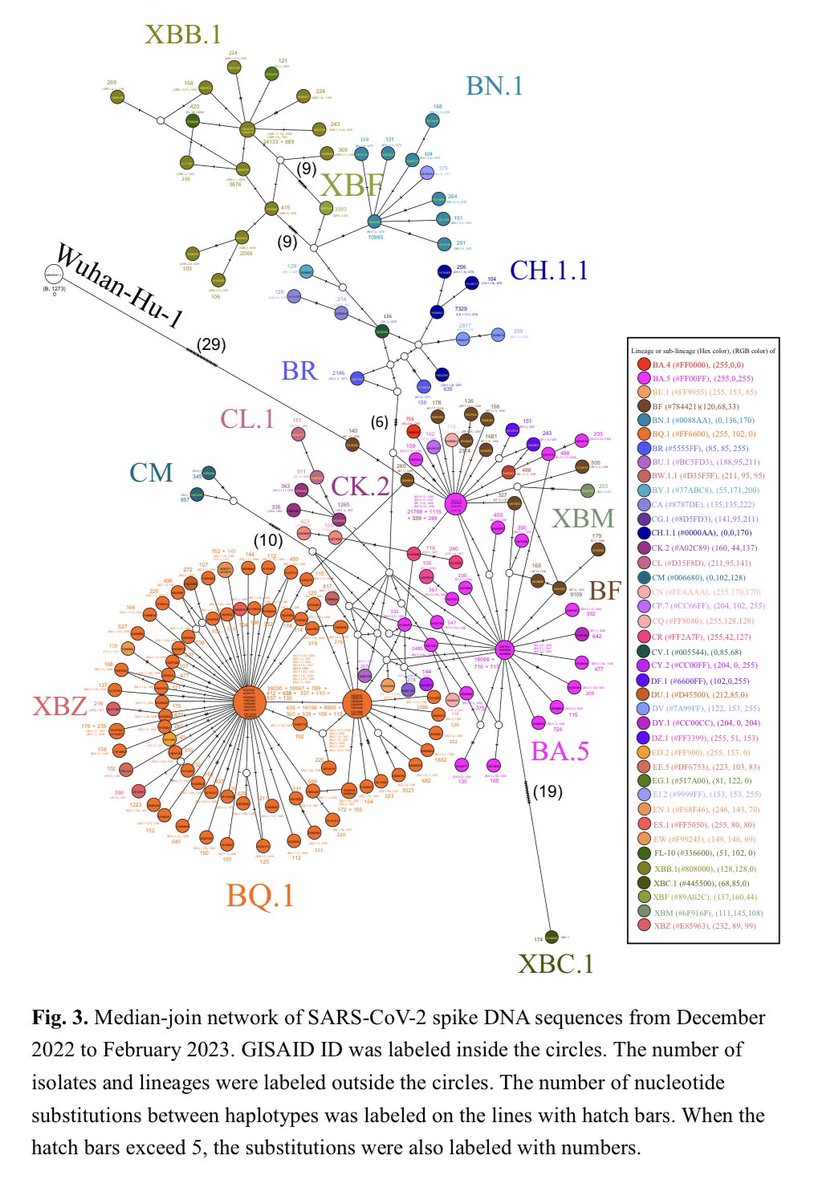
The spike DNA sequences of these diversifying centers had the representative accession IDs (EPI_ISL_) of 16040256 (BN.1.2), 15970311 (BA.5), 16028739 (BA.5.11), 16028774 (BQ.1), 16027638 (BQ.1.1.23), and 16044705 (XBB.1.5). 4/
Selection analyses revealed 26 amino-acid sites under positive selection.
These sites included L5, V83, W152, G181, N185, V213, H245, Y248, D253, S255, S256, G257, R346, R408, K444, V445, G446, N450, L452, N460, F486, Q613, Q675, T883, P1162, and V1264. 5/
These sites included L5, V83, W152, G181, N185, V213, H245, Y248, D253, S255, S256, G257, R346, R408, K444, V445, G446, N450, L452, N460, F486, Q613, Q675, T883, P1162, and V1264. 5/
Continued surveillance and research are necessary to monitor the evolution and potential impact of these variants on public health. 6/
biorxiv.org/content/10.110…
biorxiv.org/content/10.110…
• • •
Missing some Tweet in this thread? You can try to
force a refresh

 Read on Twitter
Read on Twitter