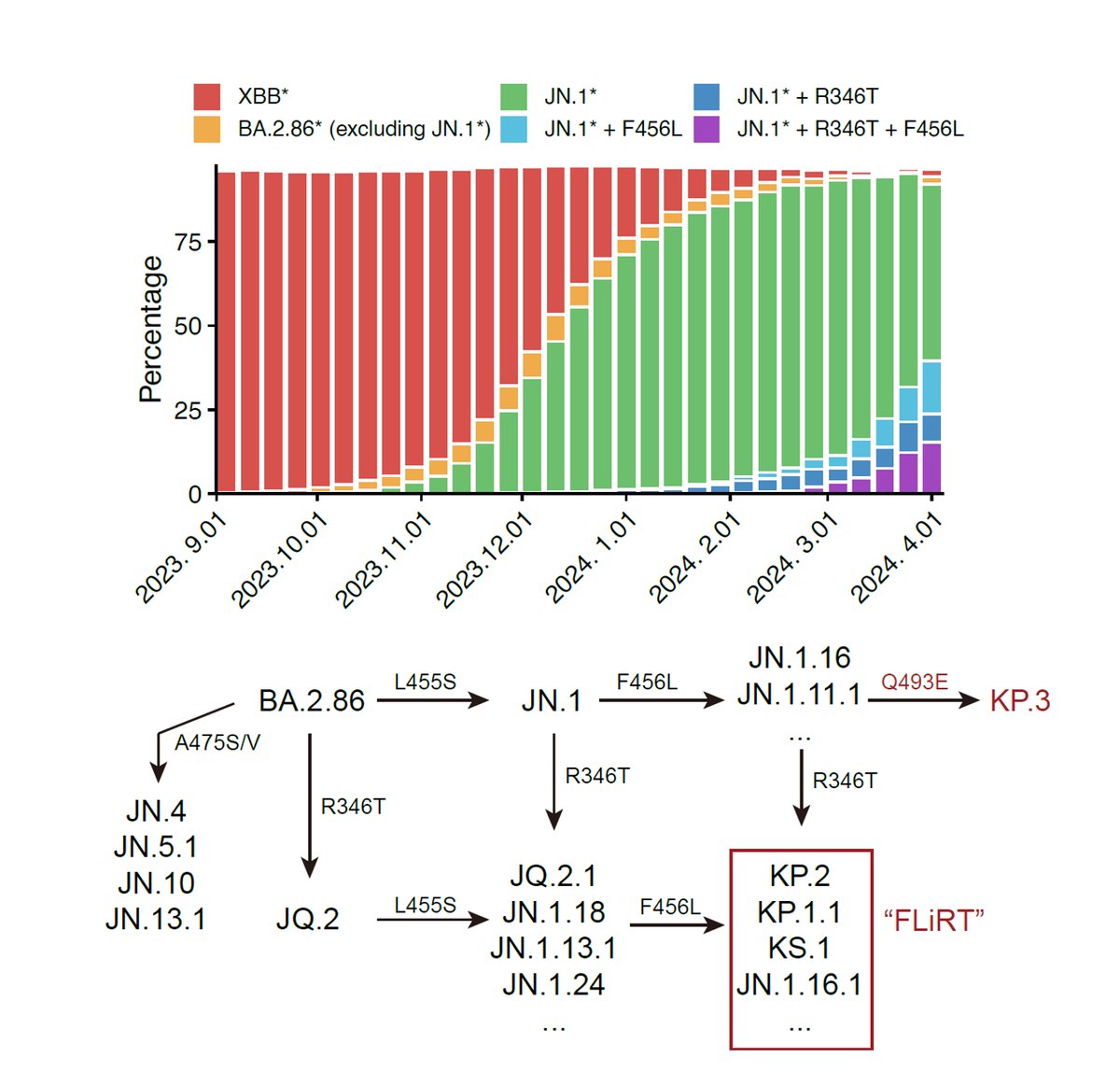
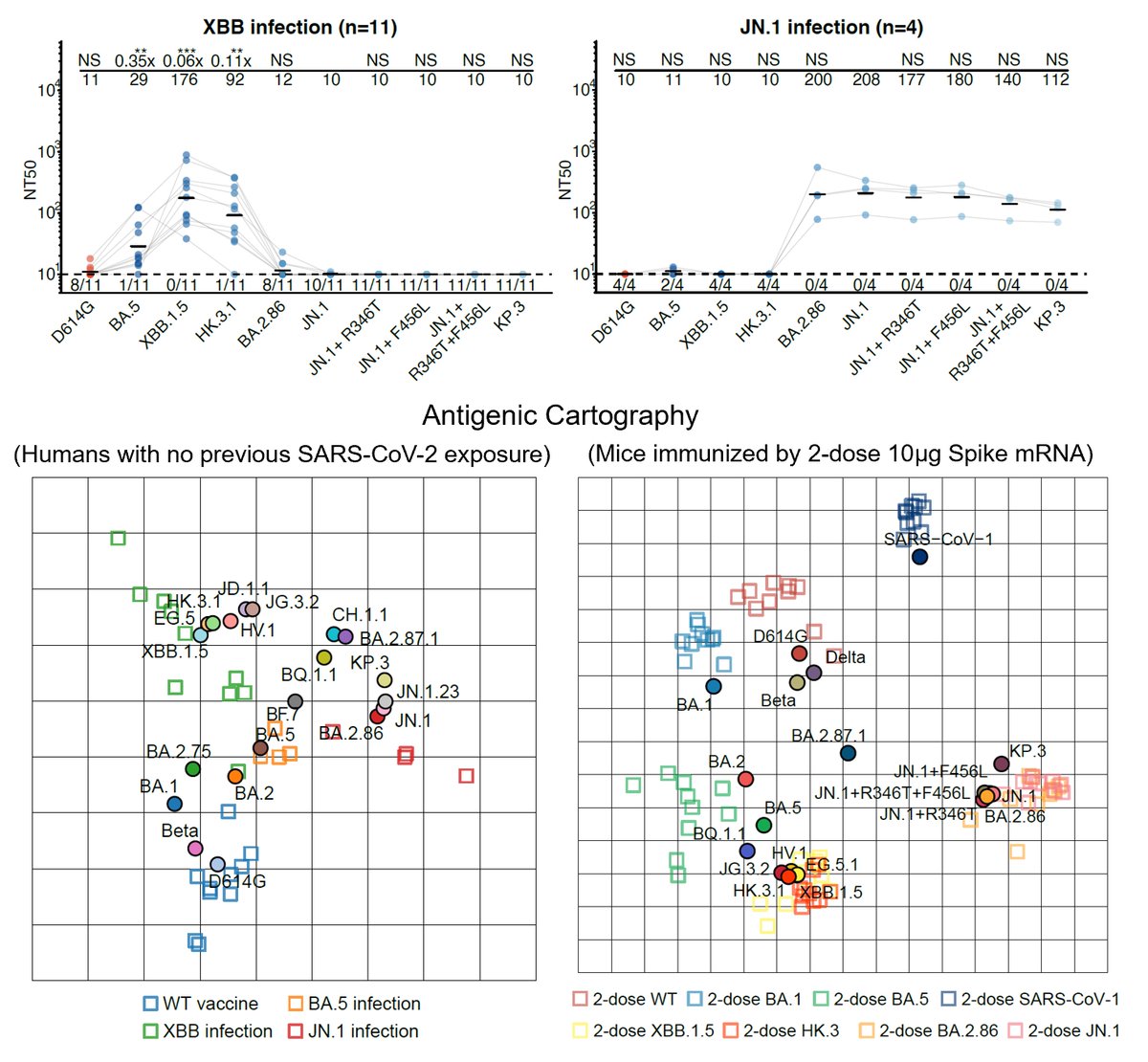
Sharing some new experimental data on BA.2.86:
1) BA.2.86 is antigenically distinct compared to XBB.1.5.
2) BA.2.86 can significantly escape XBB-infection/vaccination induced antibodies.
3) However, the infectivity of BA.2.86 may be much lower than XBB.1.5 and EG.5. (1/n)
1) BA.2.86 is antigenically distinct compared to XBB.1.5.
2) BA.2.86 can significantly escape XBB-infection/vaccination induced antibodies.
3) However, the infectivity of BA.2.86 may be much lower than XBB.1.5 and EG.5. (1/n)

By using pseudovirus neutralization assay and antigenic cartography (based on mRNA immunized mouse serum), we found that BA.2.86 is antigenically distinct from WT, BA.2, BA.5, and XBB.1.5. This means that XBB-induced antibodies cannot well recognize and neutralize BA.2.86. (2/n) 

Indeed, BA.2.86 can induce significant antibody evasion of plasma isolated from convalescents who experienced XBB breakthrough infection or reinfections. BA.2.86's immune evasion capability even exceeds EG.5 and is comparable to "FLip" variants (XBB.1.5 + L455F & F456L). (3/n) 

As for monoclonal neutralizing antibody (NAb) drugs, all approved antibodies can't neutralize BA.2.86 well, but SA55 remains effective (clinical phase II-III). Interestingly, the E554K mutation carried by BA.2.86 can escape SD1-targeting NAb, represented by S3H3. (4/n) 

We also tested a panel of XBB.1.5-effective NAbs against XBB.1.5-based pseudoviruses carrying single BA.2.86 RBD mutations. Results showed that N450D, K356T, L452W, A484K, V483del, V445H are the key mutations responsible for BA.2.86's enhanced immune evasion than XBB.1.5. (5/n) 

Importantly, we found that BA.2.86 exhibits lower cell infectivity compared to XBB.1.5 and EG.5, which may affect its transmission. The lower infectivity may be contributed by K356T, V483del, and E554K. Note that here the infectivity is measured through pseudovirus assays. (6/n) 

In sum, we found that BA.2.86 is antigenically distinct from XBB.1.5 and can escape XBB-induced neutralizing antibodies. The updated vaccine's efficacy against BA.2.86 should be closely monitored; however, BA.2.86 may not prevail very fast due to its lower infectivity. (7/n)
• • •
Missing some Tweet in this thread? You can try to
force a refresh