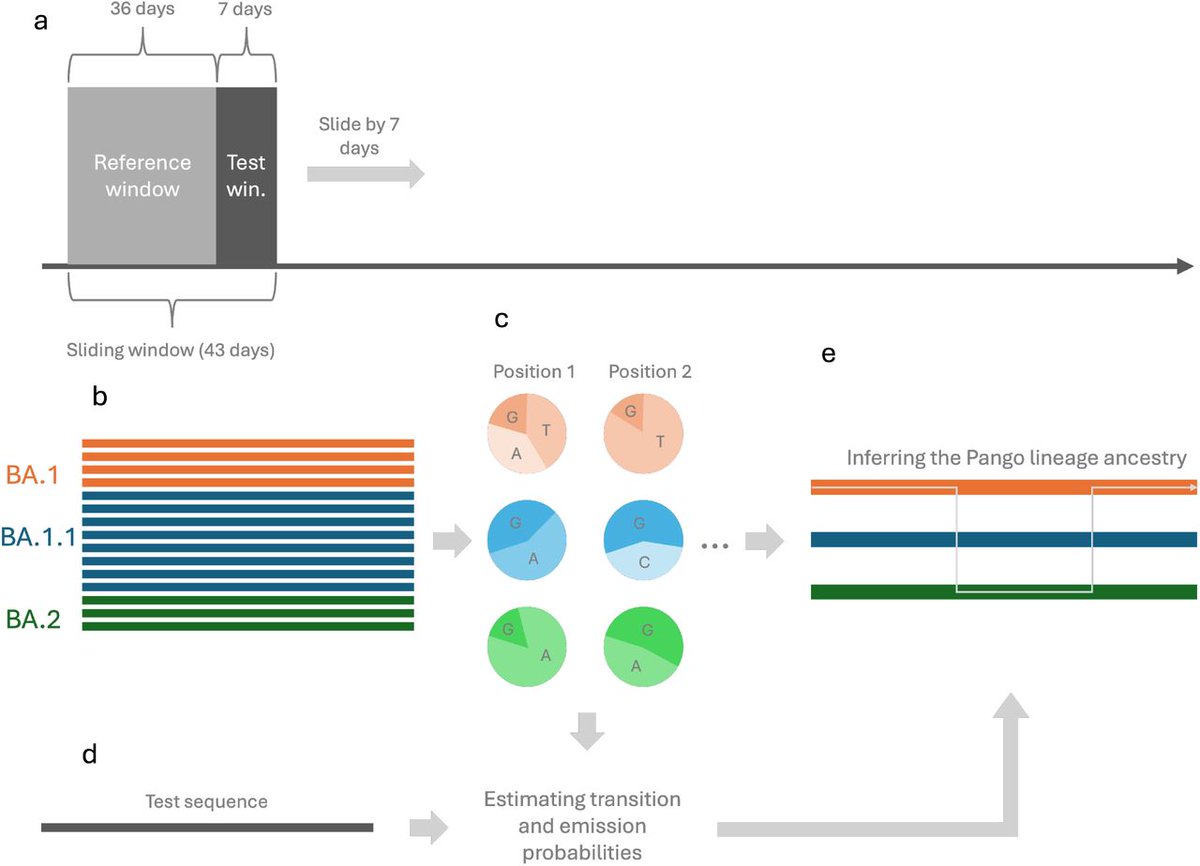
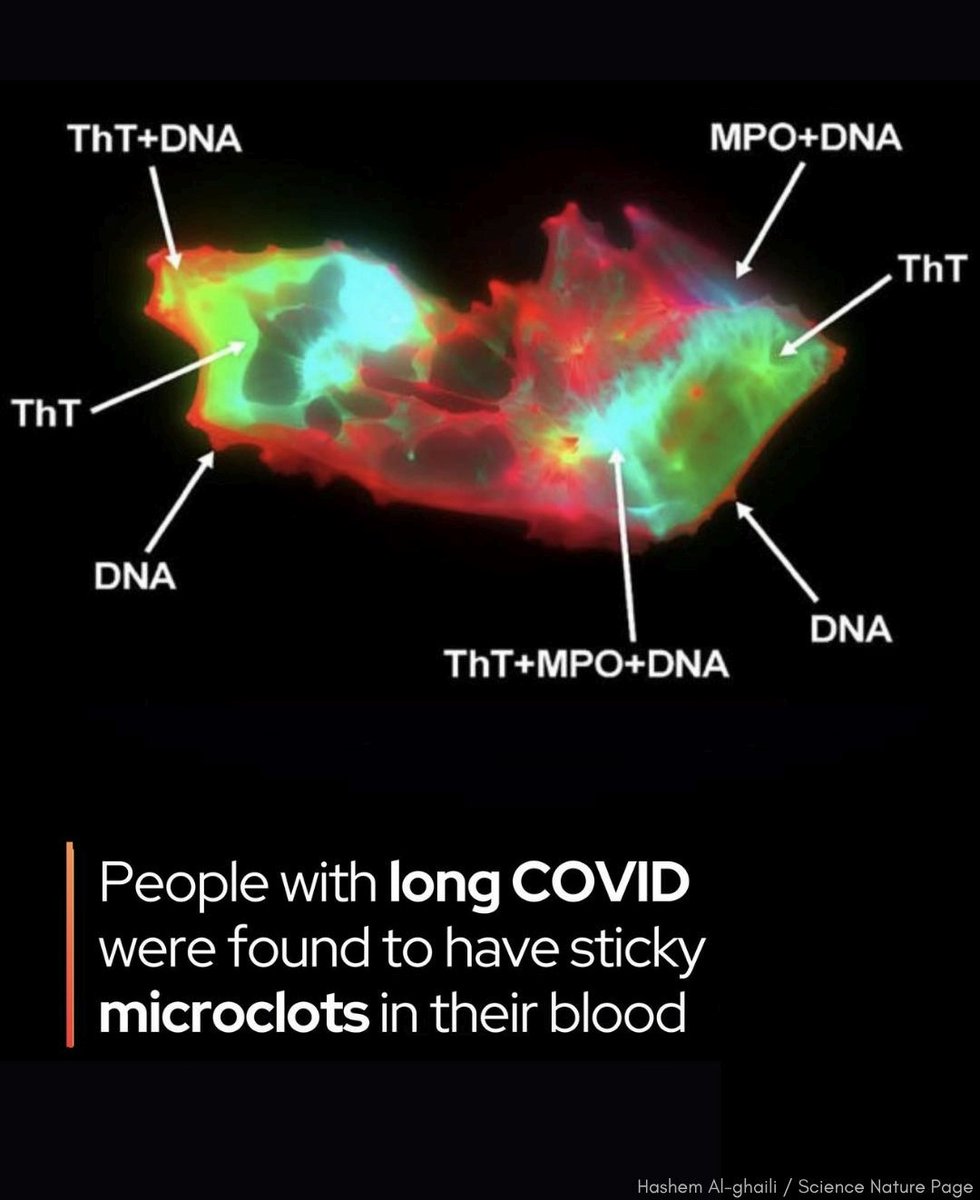
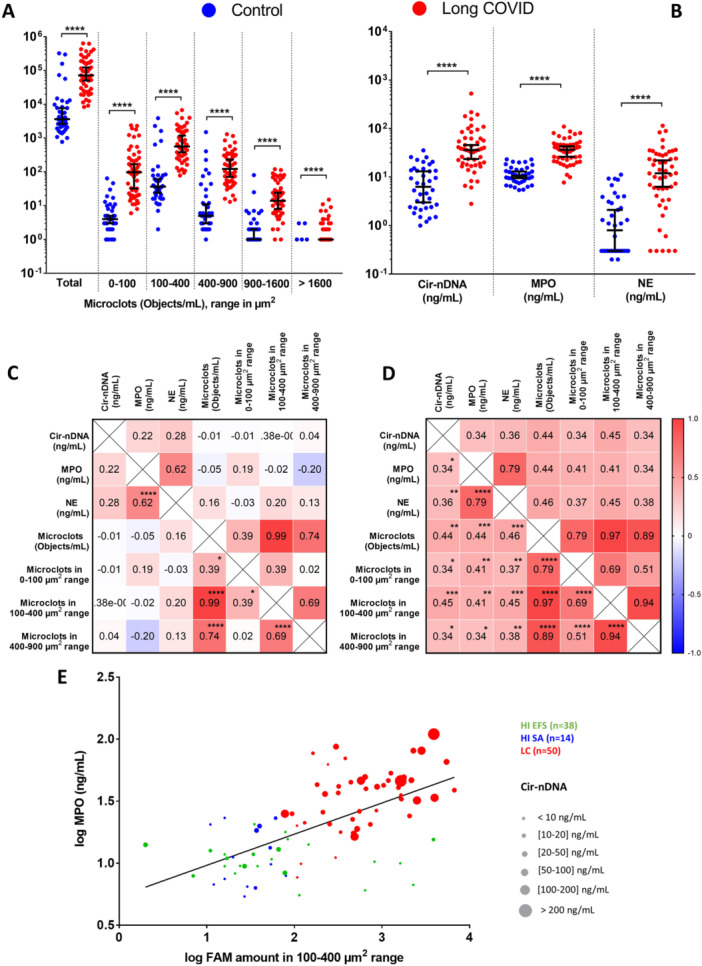
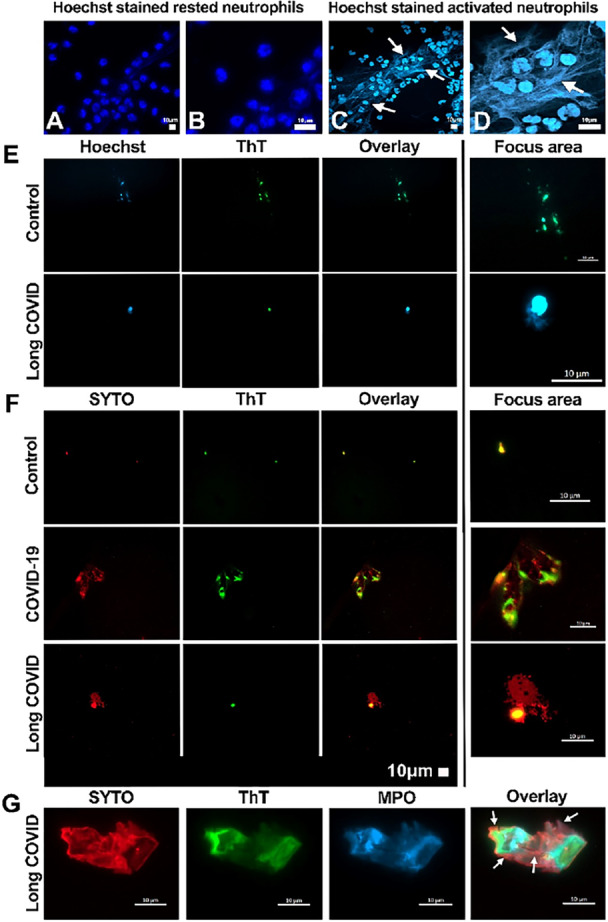
AMAZING SCIENCE ARTICLE !
A SPIKING FEVER
Long neglected, LASSA FEVER is surging in West Africa. Researchers want to know why
In September 2023, 13-year-old Ngozu Ofozor was preparing to go home after spending 2 weeks in the Lassa Fever Isolation Ward science.org/content/articl…

A SPIKING FEVER
Long neglected, LASSA FEVER is surging in West Africa. Researchers want to know why
In September 2023, 13-year-old Ngozu Ofozor was preparing to go home after spending 2 weeks in the Lassa Fever Isolation Ward science.org/content/articl…

2) Lassa fever has long been concentrated in endemic areas in Nigeria, Guinea ... But in recent years, cases of the deadly hemorrhagic disease have been popping up in other parts of West Africa. With climate change and population growth, the virus is expected to extend its reach 

3) Scientists isolated the virus from 14 specimens of multimammate mouse, Mastomys natalensis, so-called because of its long rows of nipples for nursing big litters.
science.org/doi/10.1126/sc…

science.org/doi/10.1126/sc…

4) Since 2016, several other rodent species in Nigeria, Guinea, Ghana, and Benin have been shown to harbor the virus. But M. natalensis remains the main driver of deadly outbreaks
pubmed.ncbi.nlm.nih.gov/27140942/

pubmed.ncbi.nlm.nih.gov/27140942/

5) Survivors’ stories
Many of those who recover from Lassa fever have dealt with life-changing consequences. Some are shunned by their communities, and many have permanent hearing loss. Health care workers face a particularly high risk of infection.
Many of those who recover from Lassa fever have dealt with life-changing consequences. Some are shunned by their communities, and many have permanent hearing loss. Health care workers face a particularly high risk of infection.

6) With a large team of African, European, and U.S. researchers, Sabeti and Happi analyzed blood samples collected earlier from Lassa fever patients at ISTH and KGH and sequenced 183 viral genomes, as well as 11 more from M. natalensis field samples.
nature.com/articles/s4156…

nature.com/articles/s4156…

7) Now, 17 years after Sabeti’s initial observation, she, Happi, and colleagues, in their new study, published this month in Nature Microbiology shed light on the long-standing mystery of why some people develop severe disease and some mild
nature.com/articles/s4146…
nature.com/articles/s4146…
8) Most attention has focused on ecological factors that favor virus circulation, perhaps by influencing its survival outside the host, or rodent behavior. Rainfall and to a lesser extent temperature seem to be key. 

9) By 2070, under a “moderate” climate change scenario, areas suitable for Lassa virus circulation could cover most of the region between Guinea and Nigeria. Susceptible regions could also appear for the first time in parts of Central and East Africa. 

10) CONCLUSION
No one expects the Lassa virus to spark a global pandemic—unlike respiratory viruses such as SARS-CoV-2 that easily spread from one person to another. But for those who study the disease and treat its victims, the steady march of the disease is alarming
No one expects the Lassa virus to spark a global pandemic—unlike respiratory viruses such as SARS-CoV-2 that easily spread from one person to another. But for those who study the disease and treat its victims, the steady march of the disease is alarming
• • •
Missing some Tweet in this thread? You can try to
force a refresh