The final version is out! Viruses lead to untemplated amyloid formation of proteins in human cerebrospinal fluid even after UV-inactivation, resulting in a drastic depletion of soluble proteins. 1/6
pubs.acs.org/doi/10.1021/ac…
pubs.acs.org/doi/10.1021/ac…
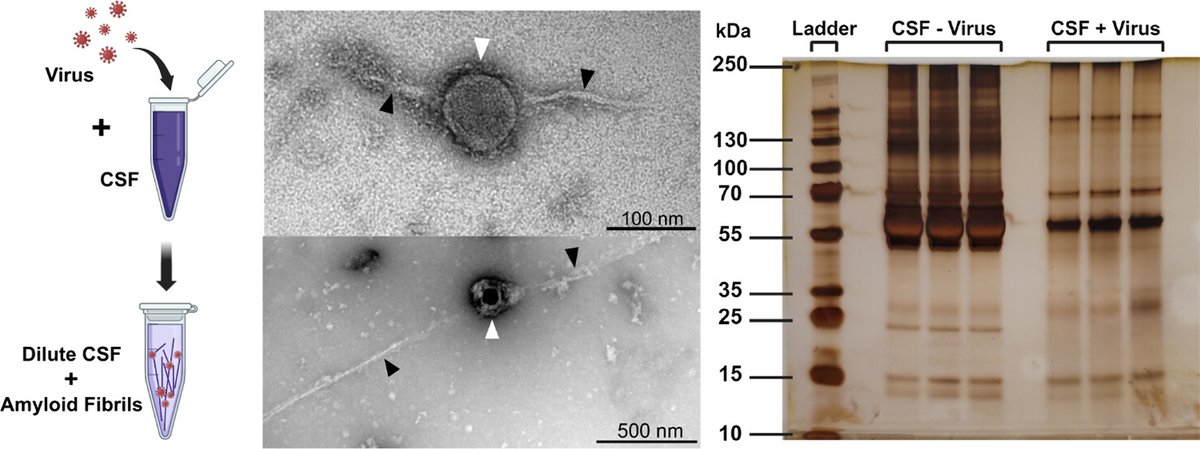
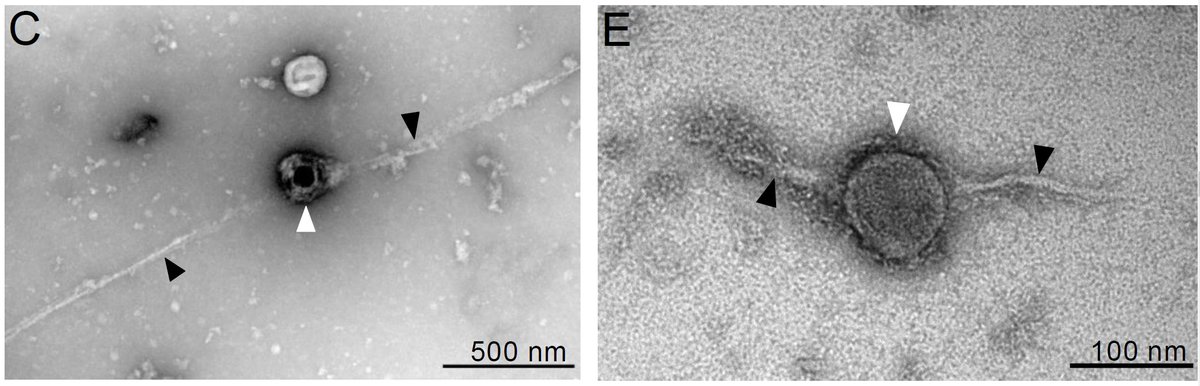
The resistance of amyloidogenic agents to UV inactivation was the foundation of the protein-only prion hypothesis. We show that, while UV inactivates viral nucleic acids, it does not affect the ability of viruses to act as catalytic surfaces that induce amyloid aggregation. 2/6
Our results support the notion that protein aggregation is a function of the recipient environment, where a subset of proteins are more susceptible to aggregation triggers due to their high concentration (supersaturation) & their ability to interact with nucleating surfaces. 3/6
It also links viral-catalyzed protein aggregation to protein loss-of-function, where this reaction leads to what can only be described as catastrophic protein depletion, something that will certainly be devastating to any biological environment in which it occurs. 4/6
We emphasize that our results are only mechanistical, we don't link a certain virus to a certain disease. However, it removes a major mechanistic barrier that prevented viruses from being considered as potential triggers of amyloid aggregation in neurodegenerative disorders. 5/6
The prion hypothesis, which unduly excluded viruses, mischaracterized a phenomenon of phase transition as replication, limited our ability to understand & treat these diseases & completely messed up the concept of biological information. More here: 6/6theprecipitate.blogspot.com/2024/10/what-i…
• • •
Missing some Tweet in this thread? You can try to
force a refresh