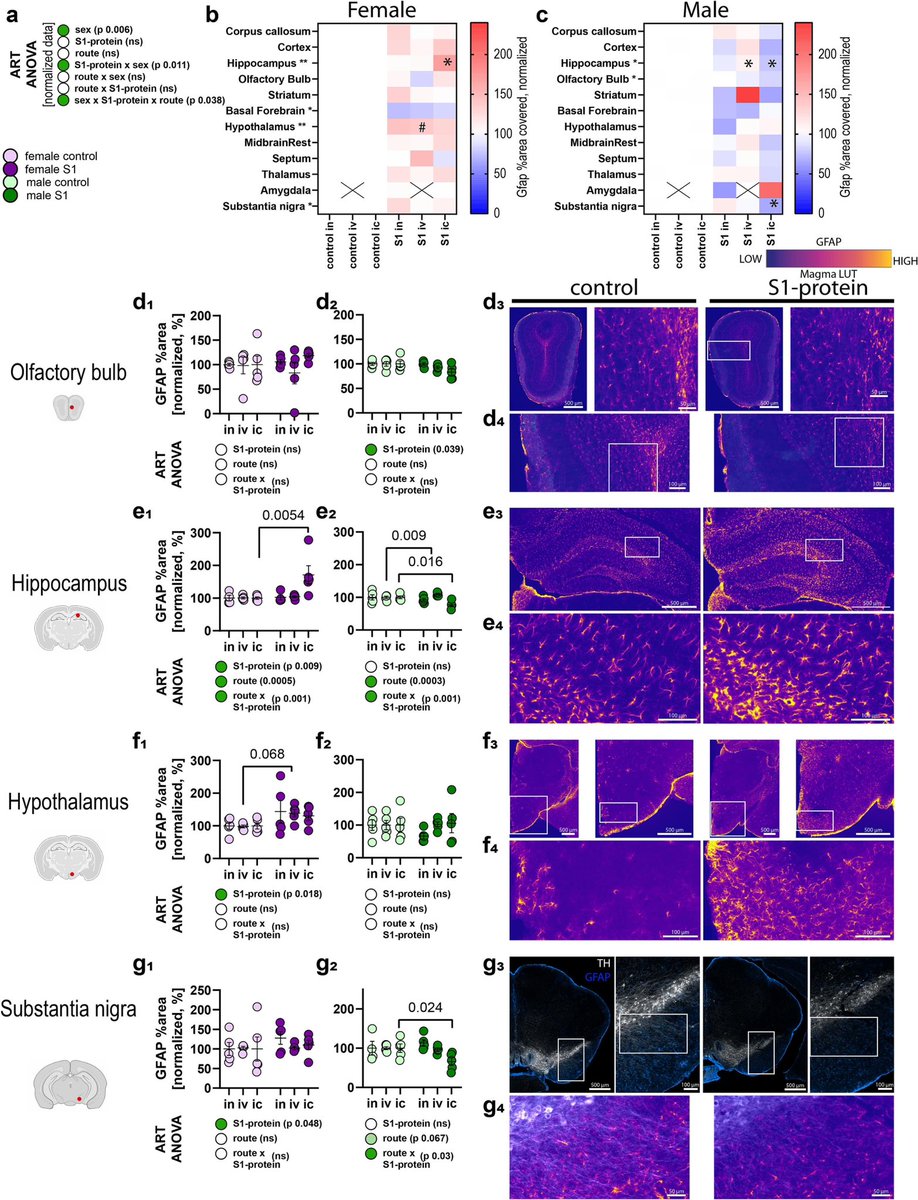
Now >5 years out from onset of SARS-CoV-2 pandemic, the mechanisms driving #LongCOVID are just beginning to be elucidated. Adipose tissue has been proposed as a potential reservoir for viral persistence and tissue dysfunction contributing to symptomology seen in LongCOVID 1/ 

Stanford researchers found that COVID infection causes lasting gene expression changes in subcutaneous fat, depleting immune pathways and increasing Hox gene activity, but these changes were not linked to #LongCOVID. 2/ 

No persistent SARS-CoV-2 RNA was detected in adipose tissue, suggesting that fat is not a viral reservoir. However, adipose transcription remodeling after COVID infection may affect metabolism and inflammation without directly causing LongCOVID. 3/3
biorxiv.org/content/10.110…
biorxiv.org/content/10.110…

• • •
Missing some Tweet in this thread? You can try to
force a refresh