A new preprint (June 2025) shows that microclots formed during COVID-19 can obstruct capillaries and impair microcirculation.
And remarkably, an earlier peer-reviewed study reached the same conclusion - by a completely different method. 🧵
And remarkably, an earlier peer-reviewed study reached the same conclusion - by a completely different method. 🧵
In the new study by Kell, Pretorius et al., SARS-CoV-2 is associated with abnormal blood clotting that produces fibrinaloid microclots - clots containing amyloid (!) fibrin that are resistant to fibrinolysis (breakdown). preprints.org/manuscript/202…
These microclots are made of amyloid-type fibrin - a misfolded version that resists not only natural fibrinolysis, but also standard clot-busting drugs.
In other words: your body can’t easily remove them. Neither can your meds.
In other words: your body can’t easily remove them. Neither can your meds.
Using laser Doppler and laser speckle imaging (LDI/LSI), the researchers found:
elevated blood pressure
reduced capillary blood flow
Consistent with capillary obstruction and rarefaction.
This suggests high blood pressure is an effect, not the cause!
elevated blood pressure
reduced capillary blood flow
Consistent with capillary obstruction and rarefaction.
This suggests high blood pressure is an effect, not the cause!
Now compare this with a prior peer-reviewed study (Jalal et al., Scientific Reports, 2025):
Researchers trained an AI model (EfficientNet) to detect abnormalities in nailfold capillary images: dilated, hemorrhagic, and disorganized vessels nature.com/articles/s4159…
Researchers trained an AI model (EfficientNet) to detect abnormalities in nailfold capillary images: dilated, hemorrhagic, and disorganized vessels nature.com/articles/s4159…
The AI model (CE-NFCNet) achieved perfect accuracy in distinguishing healthy vs abnormal capillaries- even in noisy images (eg low contrast, dark skin, glare).
Another line of evidence pointing to microvascular damage.
Another line of evidence pointing to microvascular damage.
Two different technologies - one functional (blood flow), one structural (capillary morphology) - arrive at the same insight:
COVID-19 leaves a lasting mark on the microvasculature, even in mild or asymptomatic cases.
COVID-19 leaves a lasting mark on the microvasculature, even in mild or asymptomatic cases.
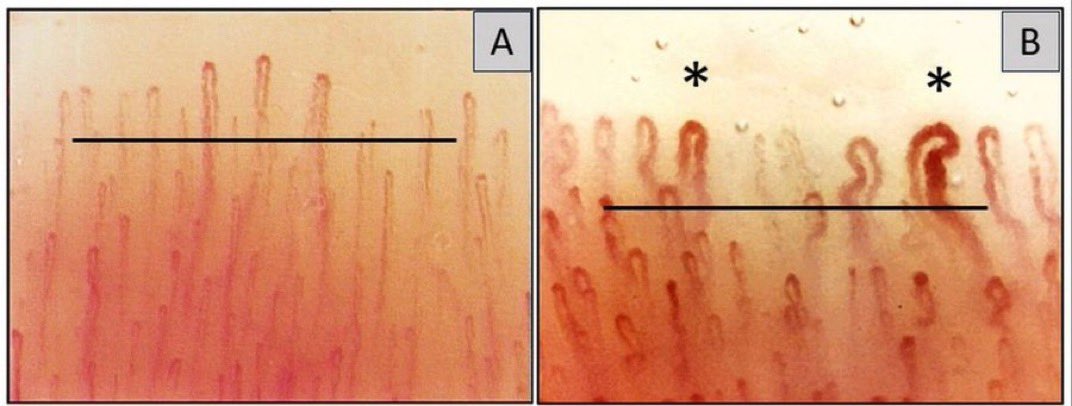
On the left: healthy nailfold capillaries.
On the right: capillaries one year after COVID.
The person on the right was never diagnosed with Long COVID!
On the right: capillaries one year after COVID.
The person on the right was never diagnosed with Long COVID!
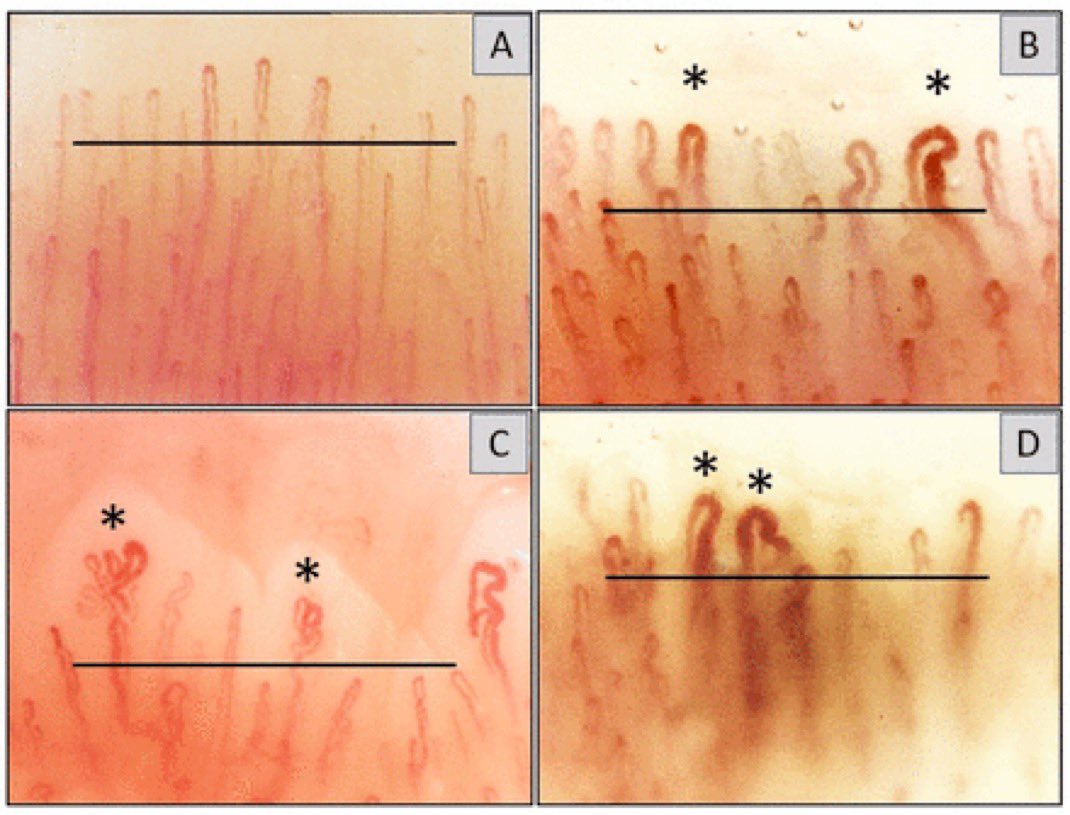
Below:
A healthy individual
B asymptomatic COVID
C & D two patients with Long COVID
Different clinical stories, but a shared vascular footprint.
A healthy individual
B asymptomatic COVID
C & D two patients with Long COVID
Different clinical stories, but a shared vascular footprint.
What we see in the skin (nailfold) is a window into systemic microcirculation.
If the capillaries are damaged here, it’s highly likely that similar damage exists in the lungs, brain, heart, kidneys - we just can’t visualize it as easily.
If the capillaries are damaged here, it’s highly likely that similar damage exists in the lungs, brain, heart, kidneys - we just can’t visualize it as easily.
These changes are:
not rare
not limited to severe COVID
not always reversible
Capillary networks do not regenerate easily - if at all.
not rare
not limited to severe COVID
not always reversible
Capillary networks do not regenerate easily - if at all.
In future waves of SARS-CoV-2 - wearing a respirator isn’t extreme.
It’s one of the few ways to protect a system that doesn’t repair itself well - your microcirculation.
It’s one of the few ways to protect a system that doesn’t repair itself well - your microcirculation.
And while we're connecting dots - let’s go back to 2020.
Already then, a pediatric study showed that most children infected with SARS-CoV-2 had lab evidence of endothelial injury, even with mild or no symptoms.
Already then, a pediatric study showed that most children infected with SARS-CoV-2 had lab evidence of endothelial injury, even with mild or no symptoms.
In this Blood Advances study from CHOP (Children’s Hospital of Philadelphia),
86% of children met diagnostic criteria for microangiopathy (TMA) - based on elevated sC5b‑9,
a marker of complement-mediated capillary damage.
Even in regular COVID cases.
86% of children met diagnostic criteria for microangiopathy (TMA) - based on elevated sC5b‑9,
a marker of complement-mediated capillary damage.
Even in regular COVID cases.
Worth noting: the 2020 CHOP study was done in hospitalized kids (COVID or MIS-C).
But importantly, many had no obvious symptoms of vascular injury - yet still showed strong lab signs (↑sC5b‑9).
Suggests subclinical damage may be broader than we think.
But importantly, many had no obvious symptoms of vascular injury - yet still showed strong lab signs (↑sC5b‑9).
Suggests subclinical damage may be broader than we think.
Later studies confirmed this:
increased circulating endothelial cells (CECs)
elevated MCP-1, VEGF-A, IL-8
= signs of ongoing endothelial activation and vascular inflammation - even in pediatric COVID.
increased circulating endothelial cells (CECs)
elevated MCP-1, VEGF-A, IL-8
= signs of ongoing endothelial activation and vascular inflammation - even in pediatric COVID.
All of this helps explain why:
breathlessness can linger
microvascular imaging looks abnormal
cognitive function dips
even when “routine tests” come back normal.
We're not just fighting a virus - but its systemic footprint.
breathlessness can linger
microvascular imaging looks abnormal
cognitive function dips
even when “routine tests” come back normal.
We're not just fighting a virus - but its systemic footprint.
• • •
Missing some Tweet in this thread? You can try to
force a refresh




