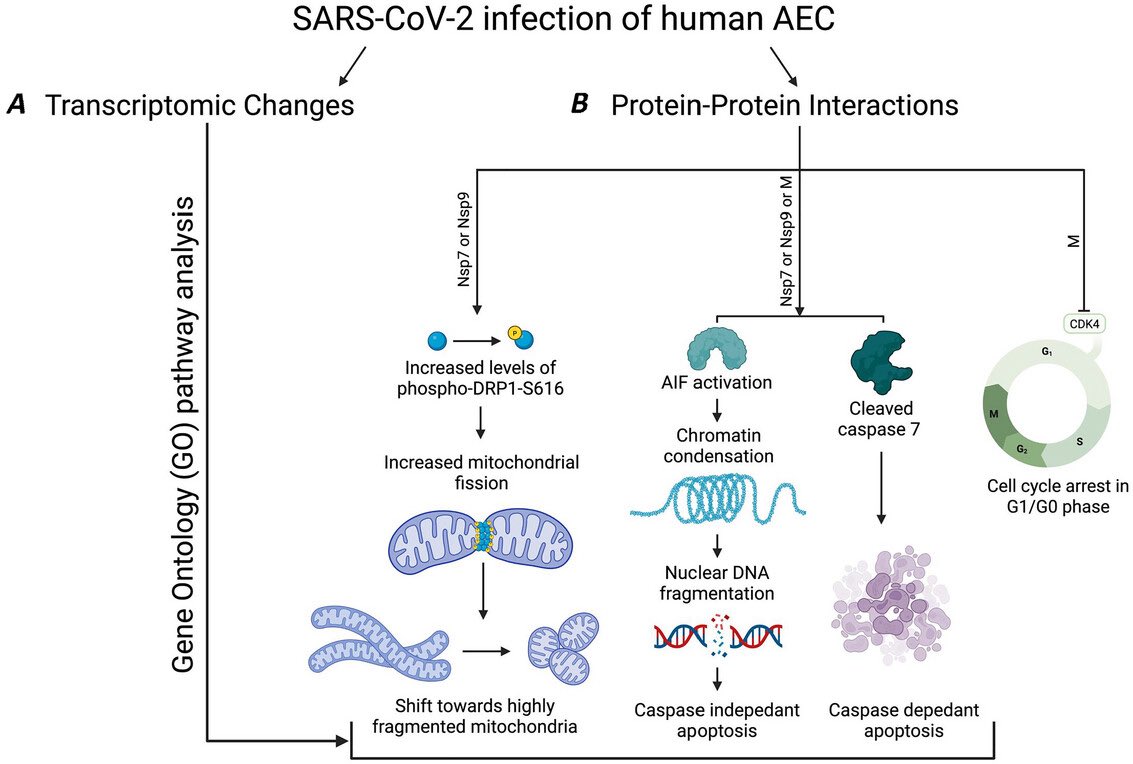
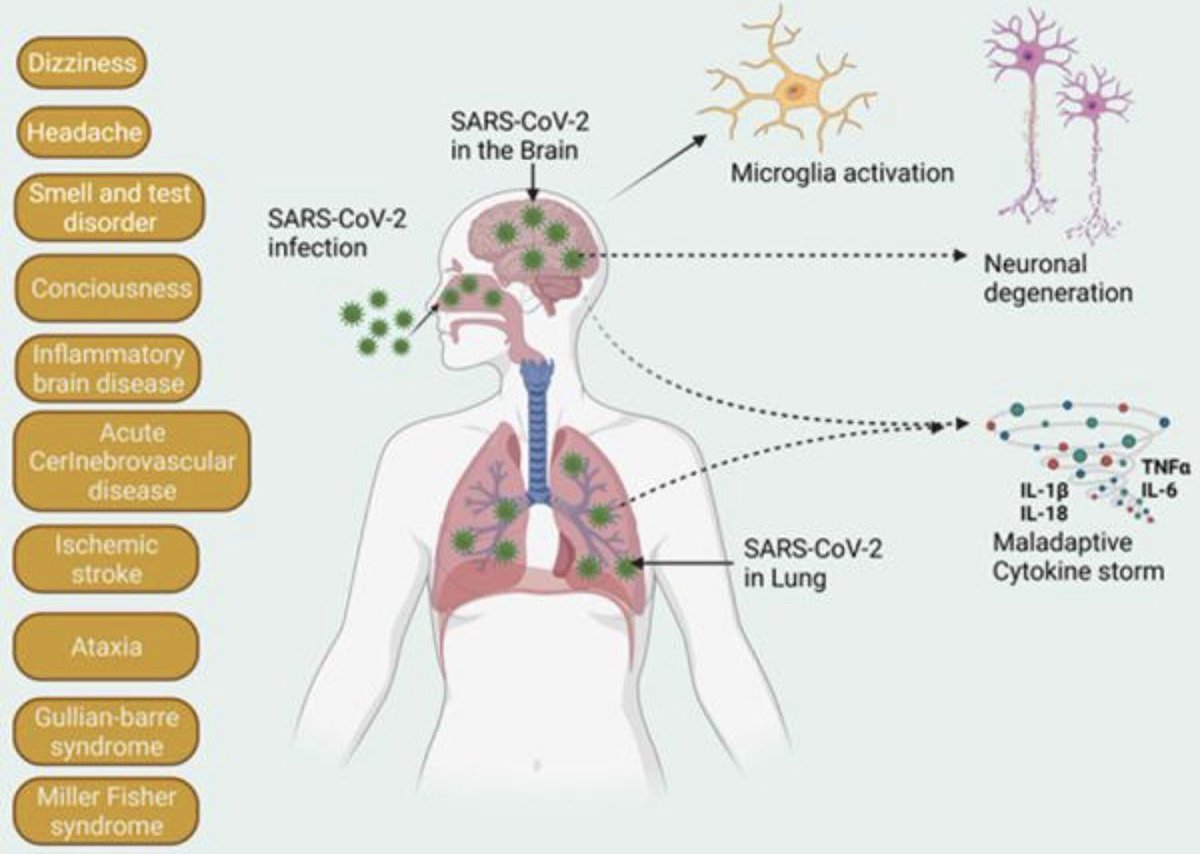
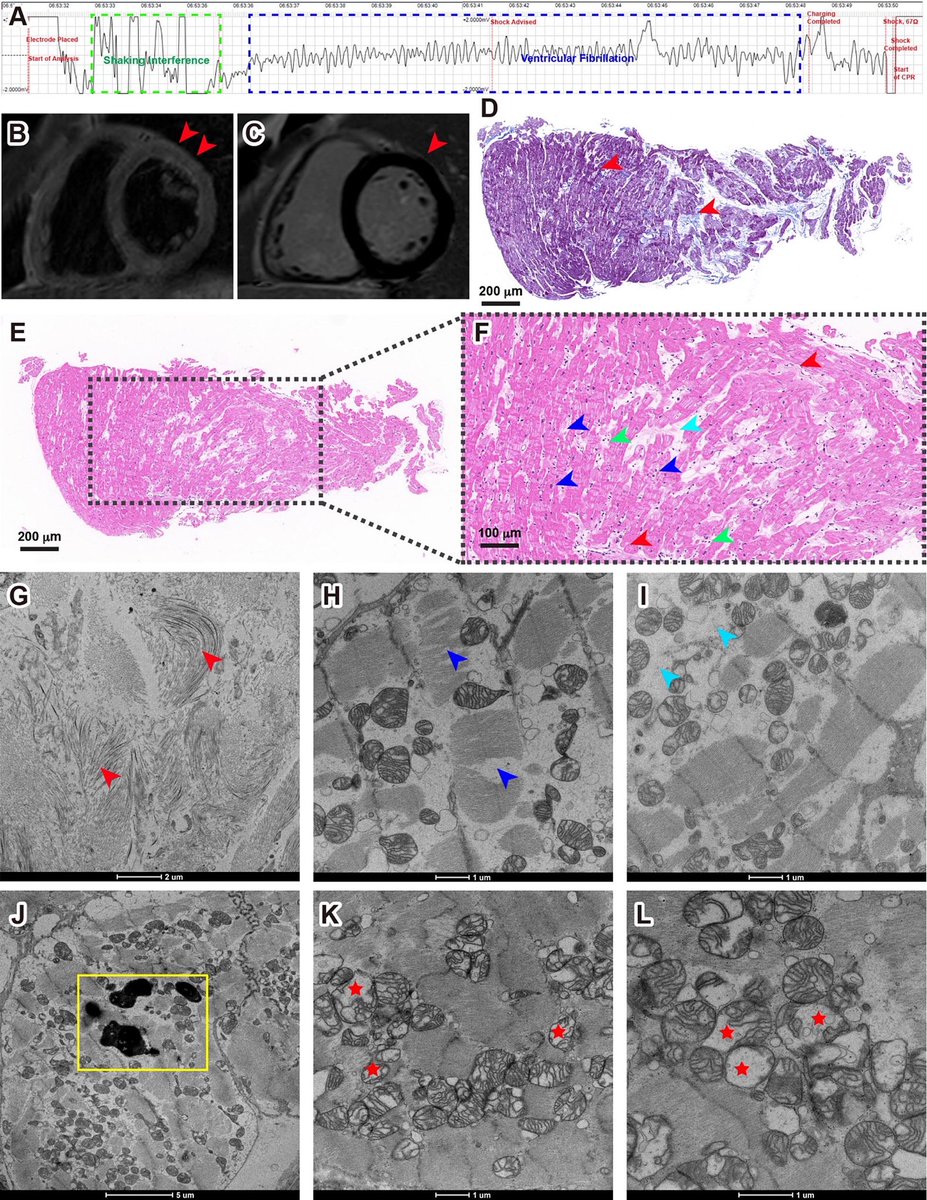
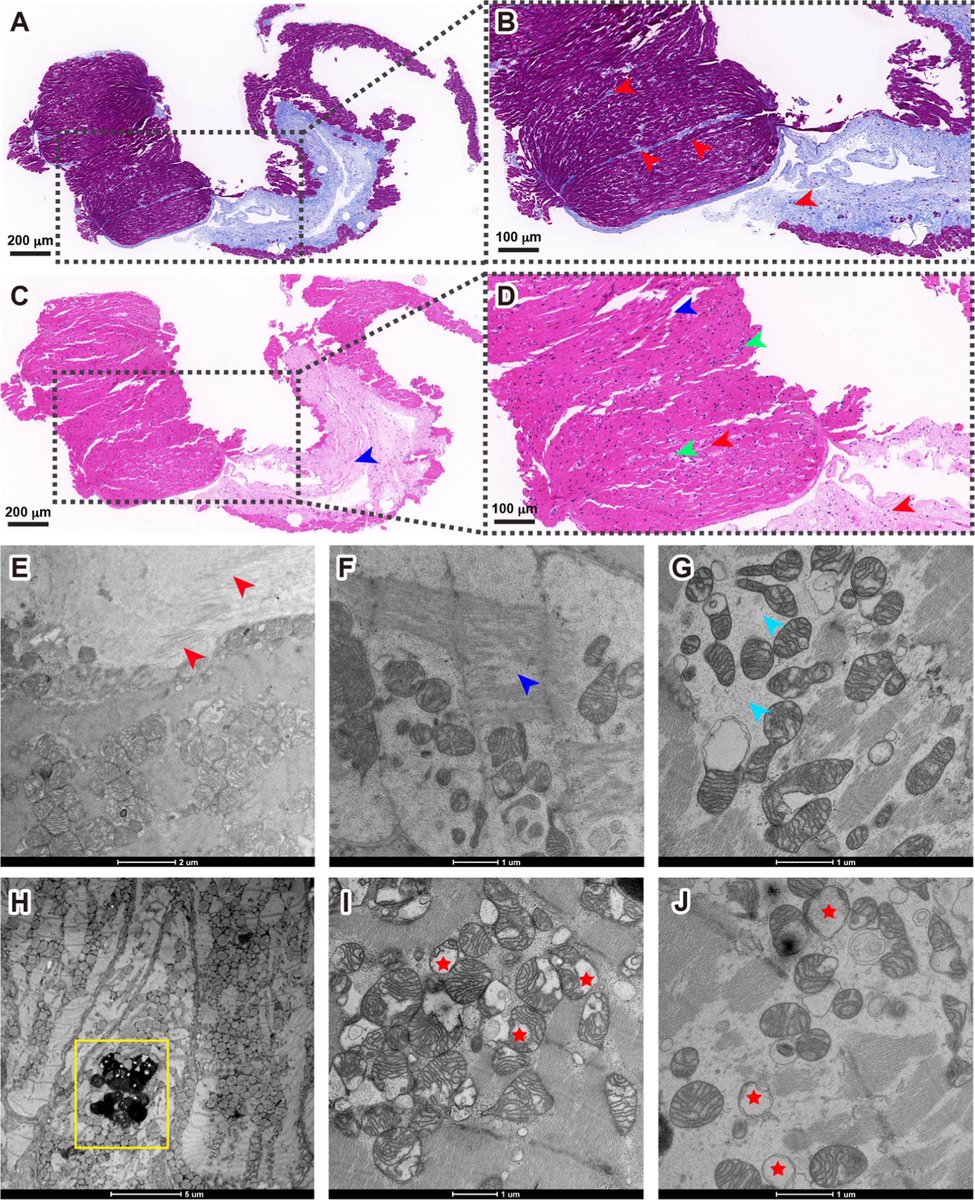
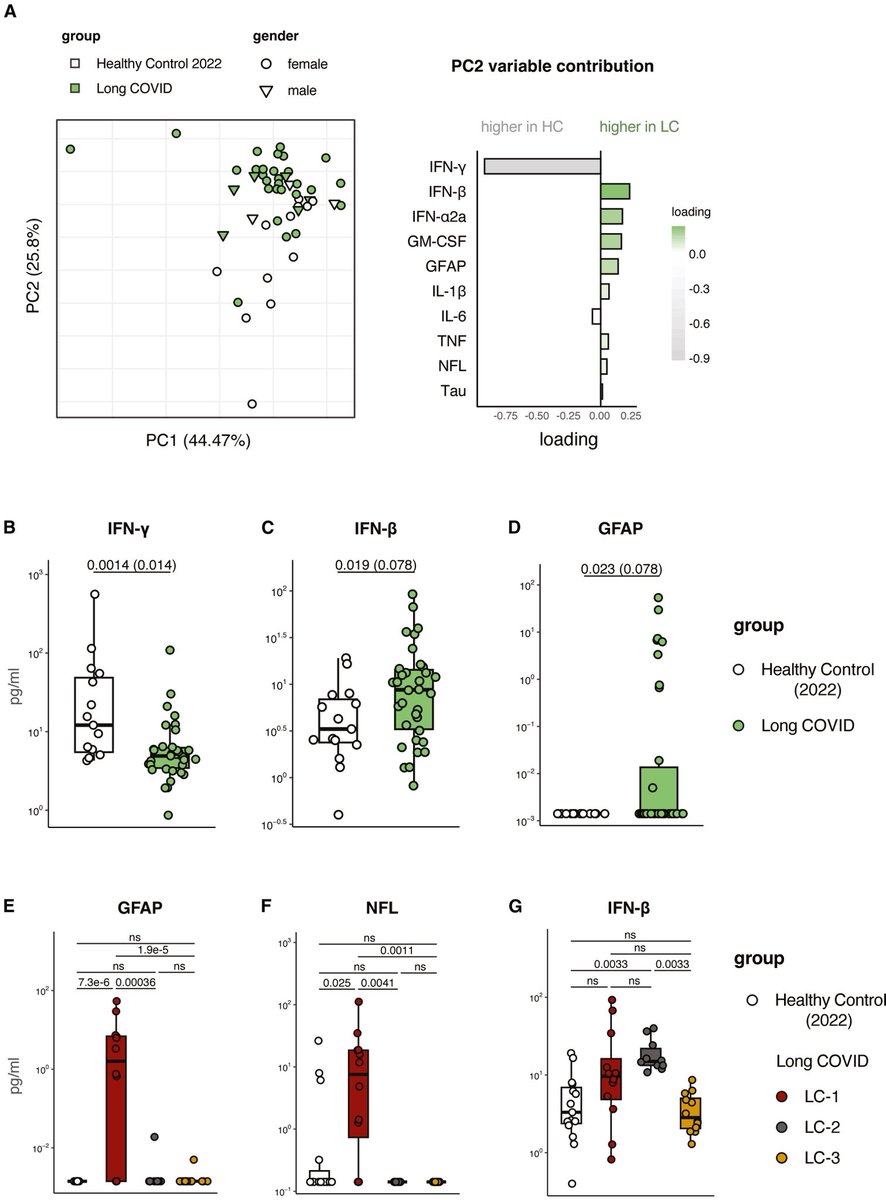
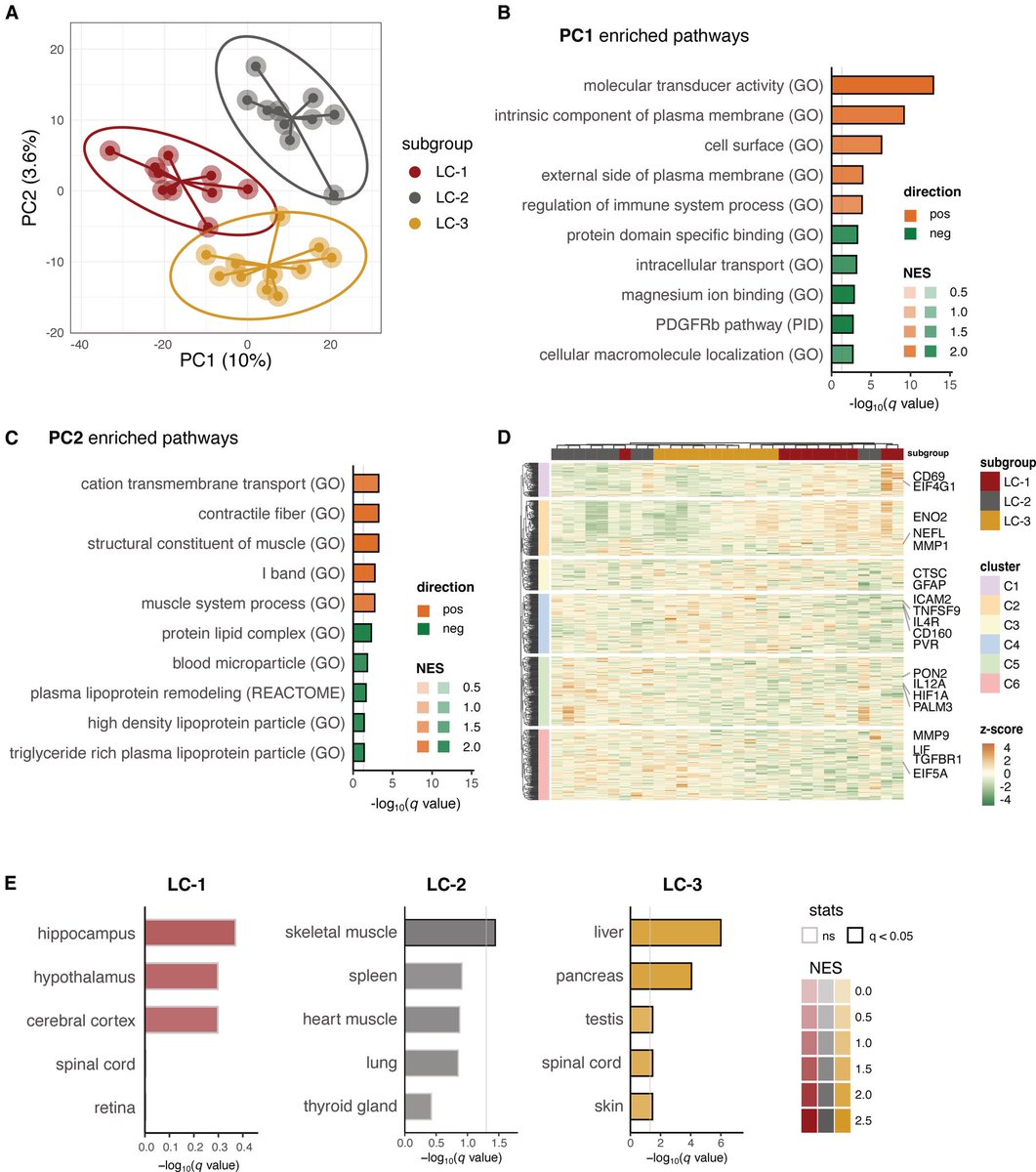
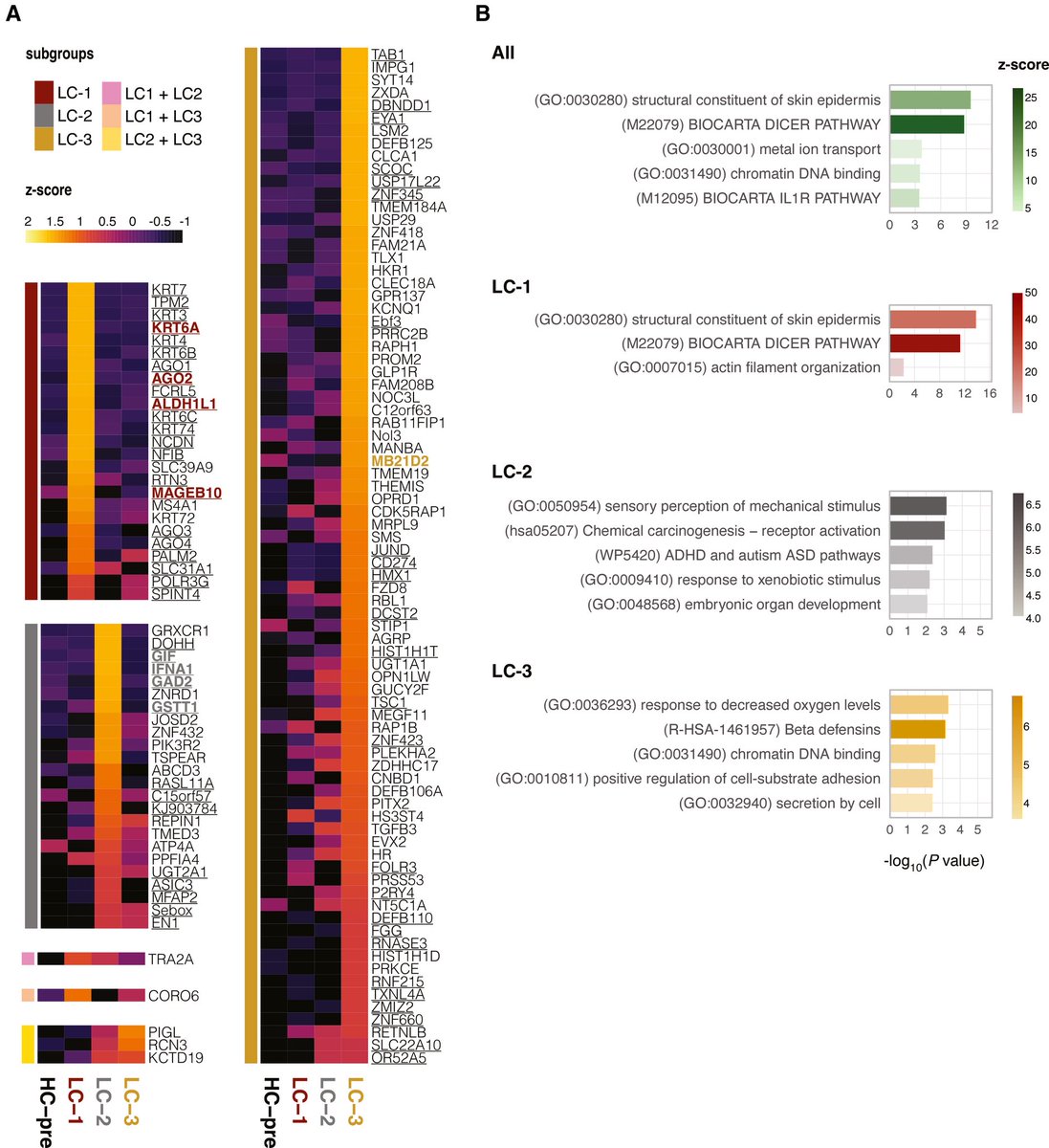
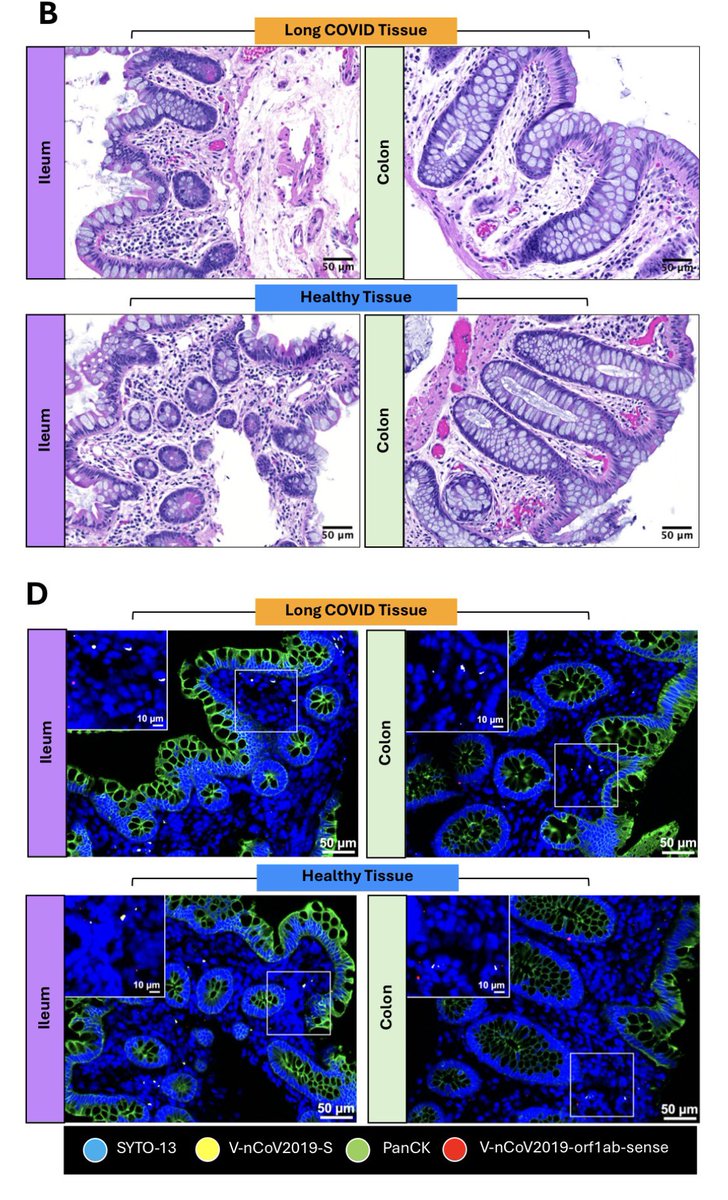
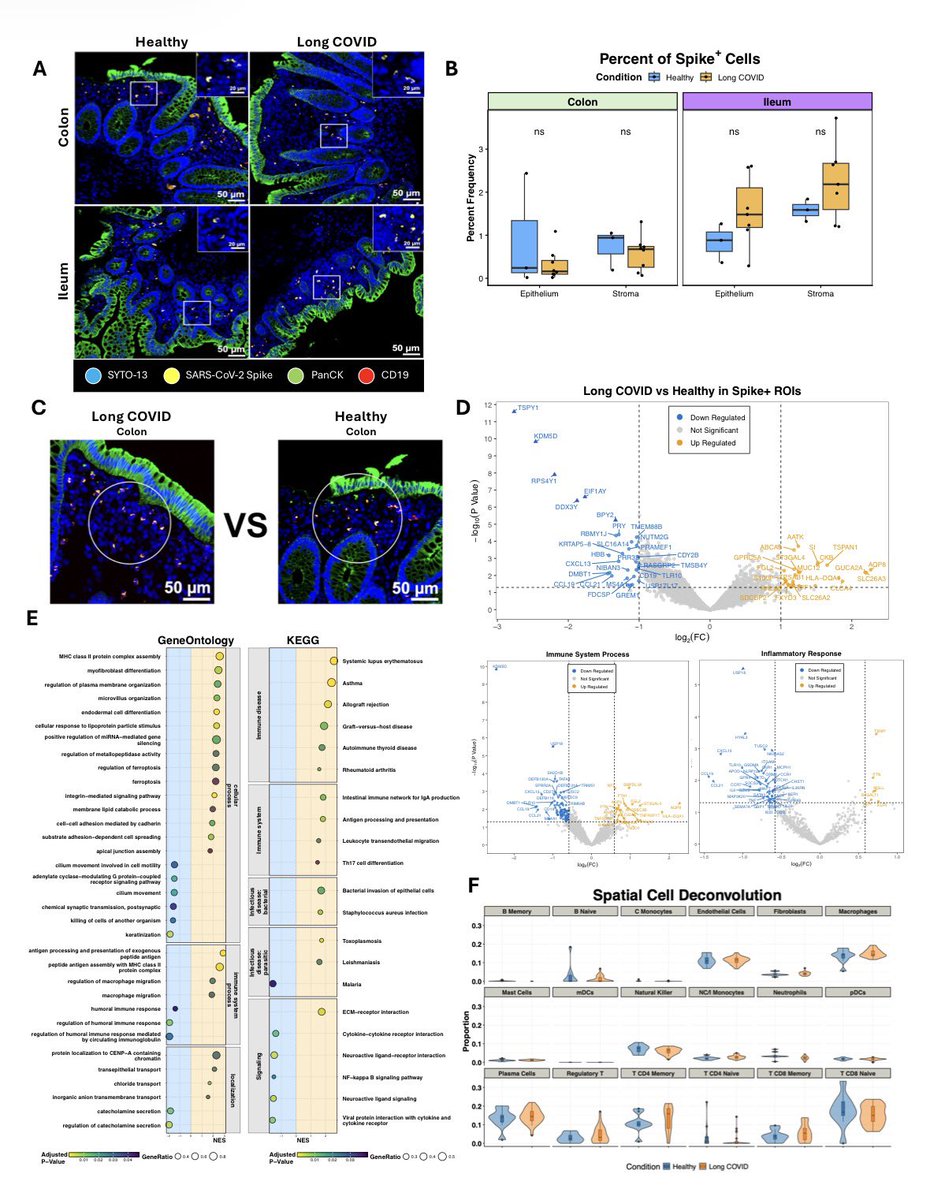
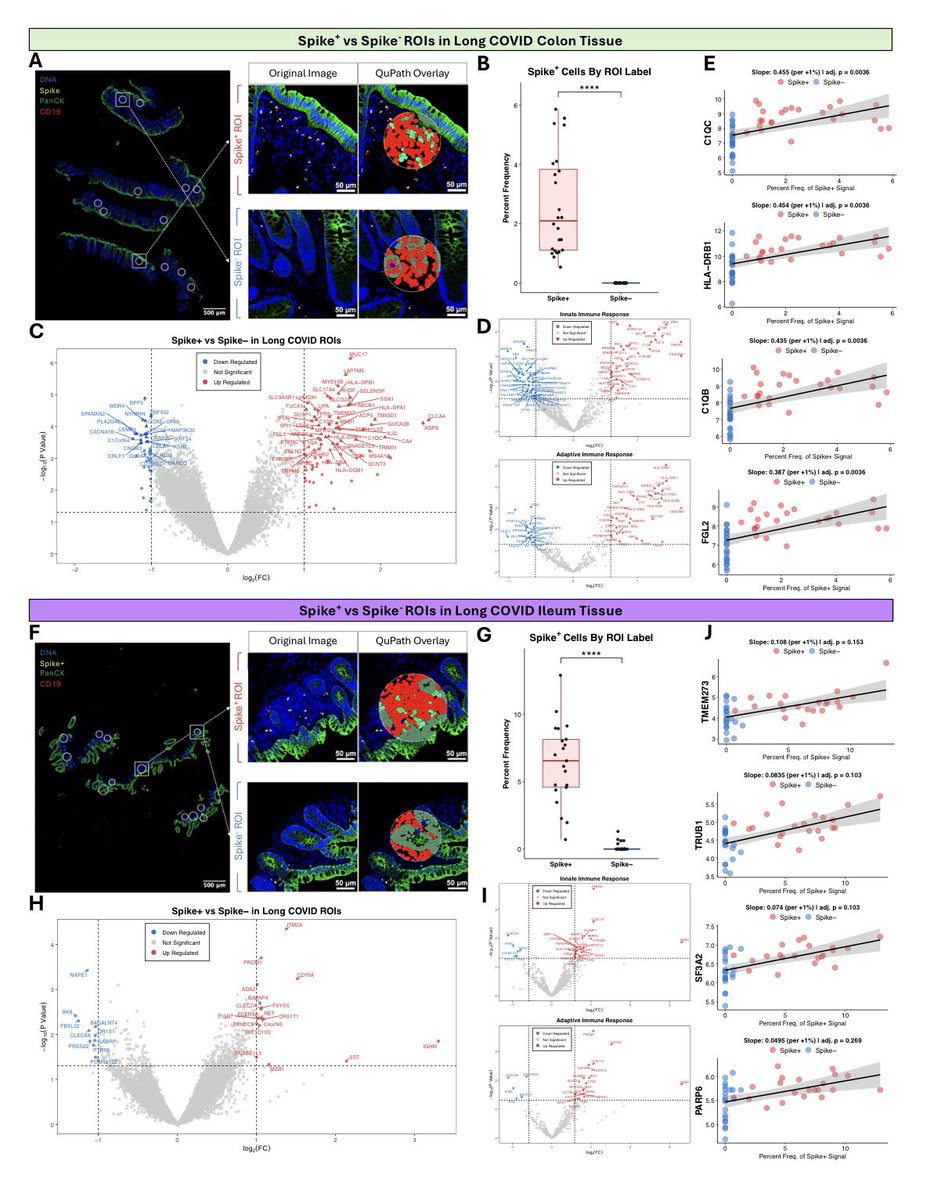
A new Yale study has found a promising target for treating the brain fog that can follow COVID-19 and offers new insight into a hypothesis about the origin of Alzheimer's disease.
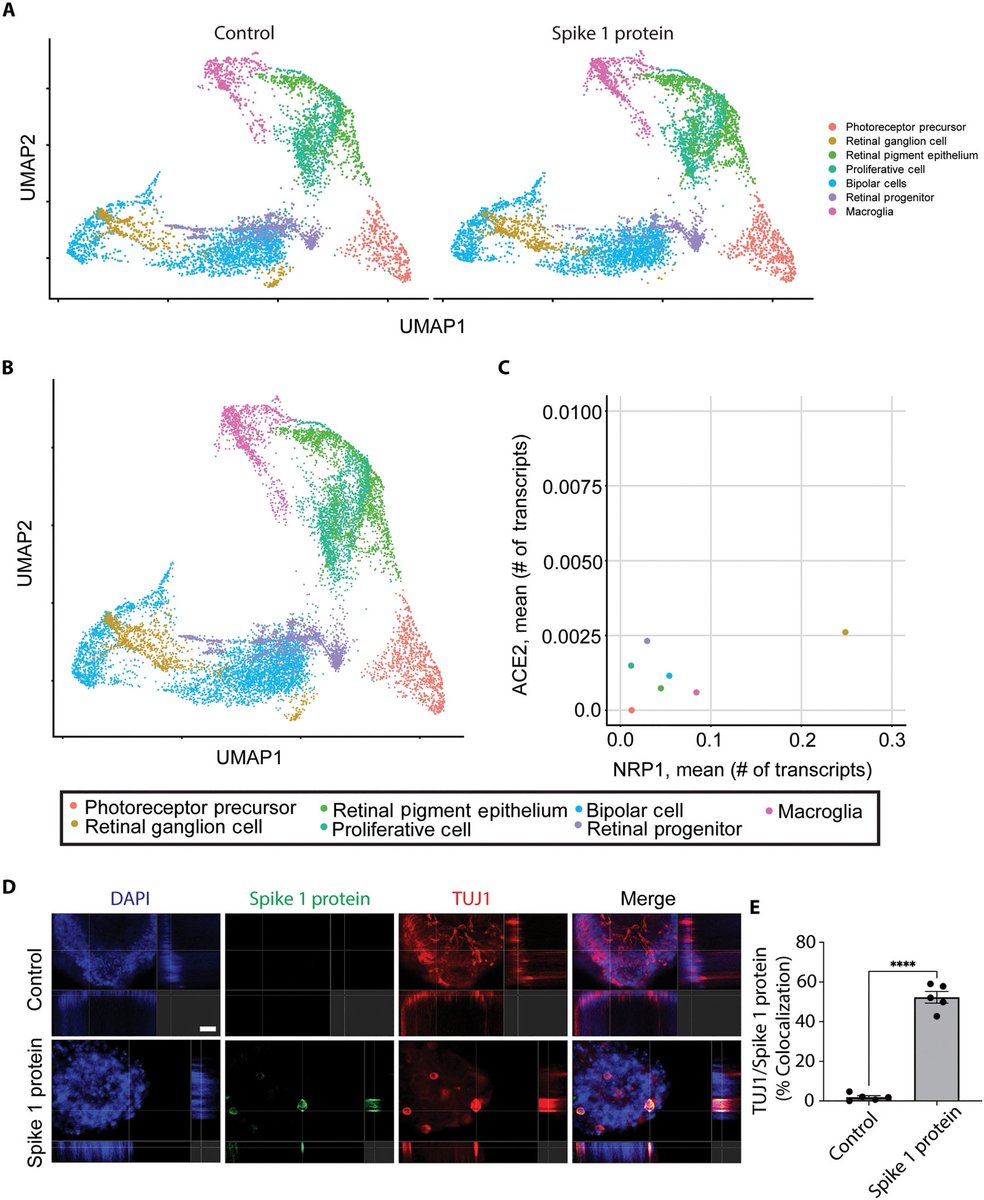
Researchers obtained viable postmortem human retinal tissue and generated human retinal organoids that contain electrophysiologically active neurons.
They demonstrated that SARS-CoV-2 induced amyloid-β extracellular protein aggregates in human retinal explants and retinal organoids. 1/
Researchers obtained viable postmortem human retinal tissue and generated human retinal organoids that contain electrophysiologically active neurons.
They demonstrated that SARS-CoV-2 induced amyloid-β extracellular protein aggregates in human retinal explants and retinal organoids. 1/

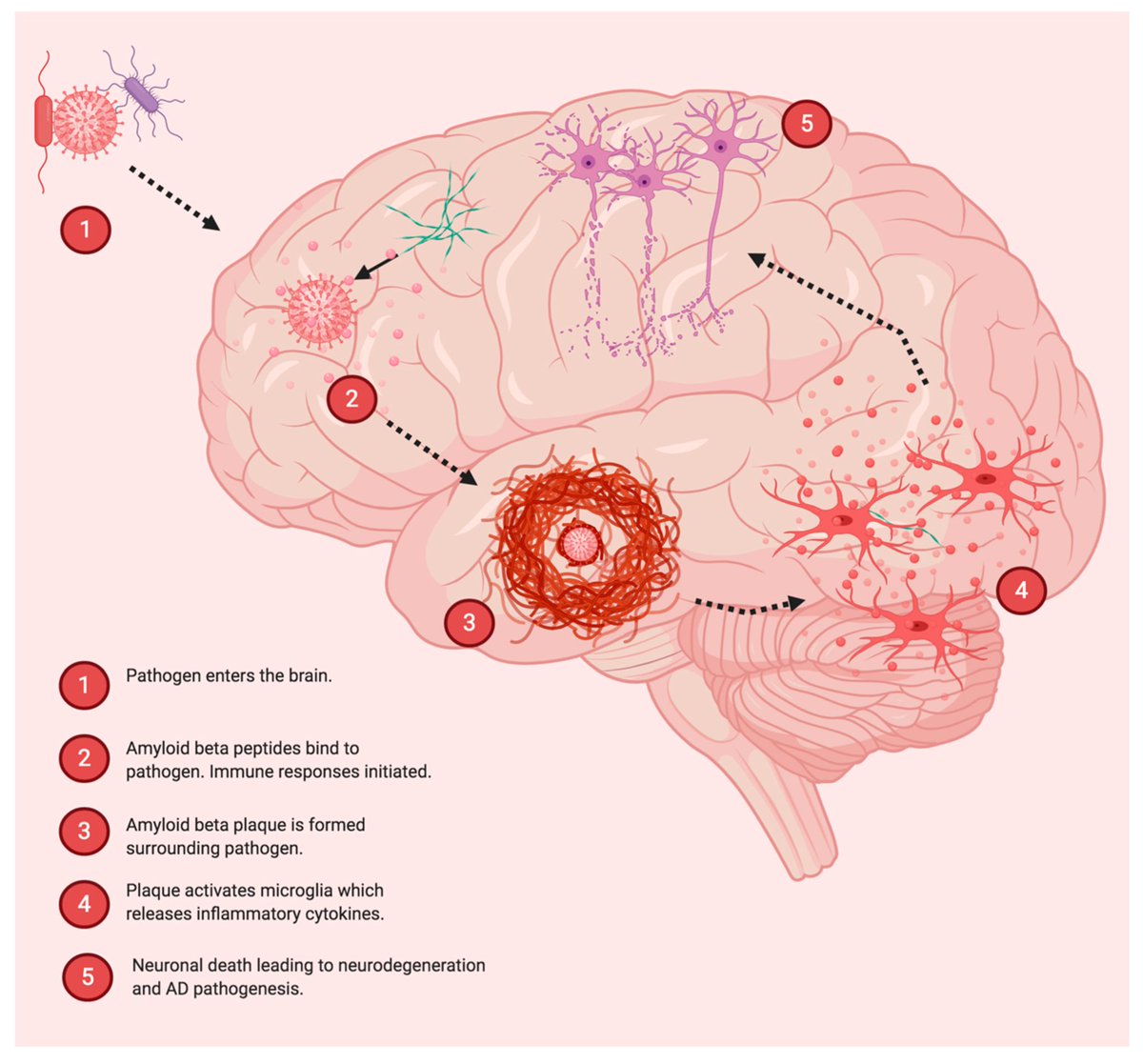
While the etiology of Alzheimer’s disease remains unknown, there is growing support for the amyloid-β antimicrobial hypothesis.
Amyloid-β, the main component of amyloid plaques in Alzheimer’s disease, has been shown to be generated in the presence of microbes.
Entrapment of microbes by aggregated amyloid-β may serve as an innate immune response to pathogenic infections. 2/
Amyloid-β, the main component of amyloid plaques in Alzheimer’s disease, has been shown to be generated in the presence of microbes.
Entrapment of microbes by aggregated amyloid-β may serve as an innate immune response to pathogenic infections. 2/
Lastly, pharmacological inhibition of neuropilin-1 resulted in reduced amyloid-β deposition in human retinal explants treated with SARS-CoV-2 Spike 1 protein.
These results suggest that Spike 1 protein, during infection with SARS-CoV-2, can induce amyloid-β aggregation, which may be associated with the neurological symptoms experienced in COVID-19. 3/
These results suggest that Spike 1 protein, during infection with SARS-CoV-2, can induce amyloid-β aggregation, which may be associated with the neurological symptoms experienced in COVID-19. 3/

In conclusion, these data suggest that COVID-19 infection, through the SARS-CoV-2 Spike 1 protein, leads to Alzheimer’s disease–related pathology. Inhibition of the cognate receptor for SARS-CoV-2 Spike 1 protein in neurons, NRP1, decreases amyloid-β accumulation in ex vivo human neural tissue. 4/
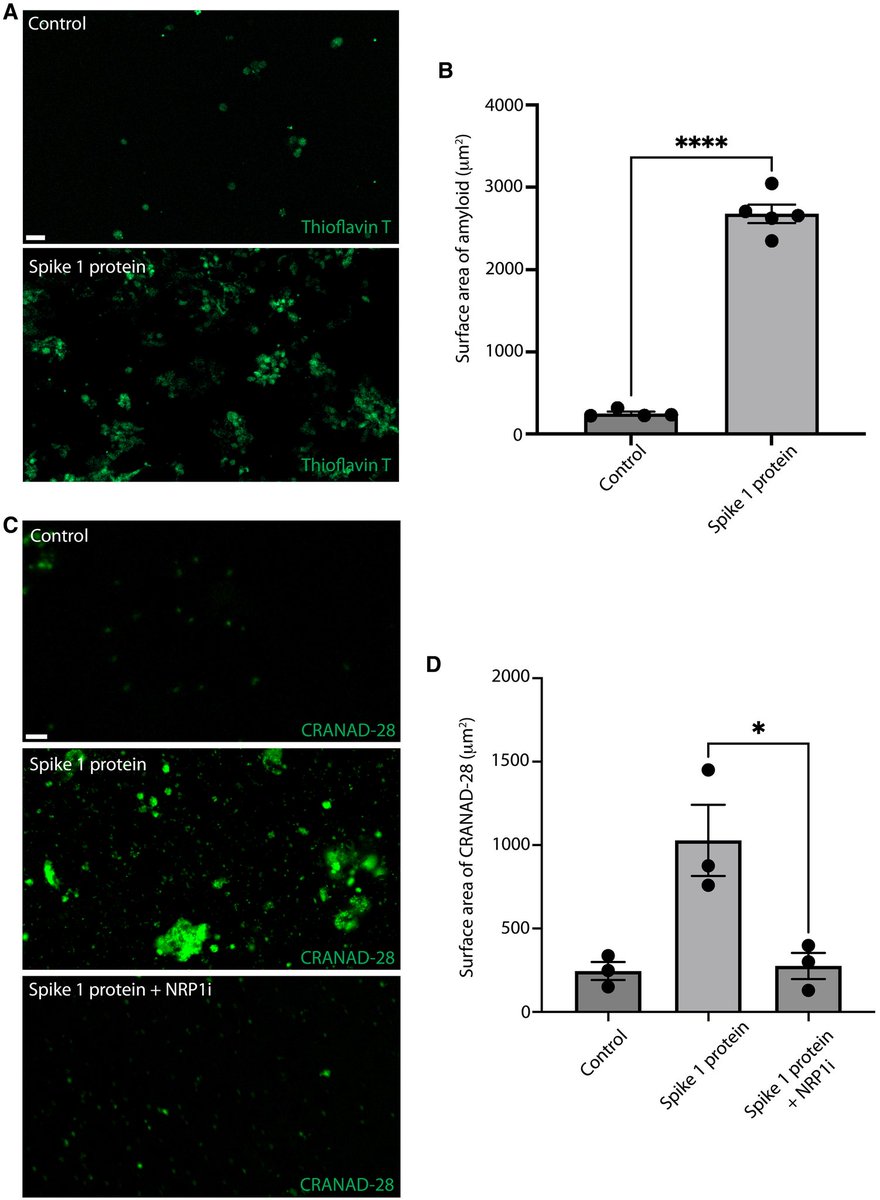
These investigations may provide insight into approaches to prevent the buildup of amyloid-β associated with viral infections.
Future studies will be aimed at identifying therapies to promote residual clearance or reduce excessive amyloid-β deposition post–COVID-19 infection. 5/
Future studies will be aimed at identifying therapies to promote residual clearance or reduce excessive amyloid-β deposition post–COVID-19 infection. 5/
Last, these studies suggest a potential approach to clinically define CNS Long Covid syndromes by measuring retinal function and amyloid-β burden. 6/6
science.org/doi/10.1126/sc…
science.org/doi/10.1126/sc…
• • •
Missing some Tweet in this thread? You can try to
force a refresh