I was at a restaurant in my hometown (Shaughnessy's Cove in Summerland, BC) and noticed the outdoor air conditioning system.
Whether they know it or not, they are using aerosol science to cool the air. I figured I’d put together a thread to explain how these work.
Whether they know it or not, they are using aerosol science to cool the air. I figured I’d put together a thread to explain how these work.

So, what is actually happening?
By spraying a mist, the outdoor eating area is cooled. Now, the area isn’t cooled because water is being sprayed all over the surfaces, akin to spraying a hose of water everywhere. Something more interesting is happening.
By spraying a mist, the outdoor eating area is cooled. Now, the area isn’t cooled because water is being sprayed all over the surfaces, akin to spraying a hose of water everywhere. Something more interesting is happening.

Mist is sprayed from a hose. Now, mist is simply a large population of individual aerosol droplets. The composition of each droplet is pure (or at least nearly pure) water. The size distribution of the aerosol will range from 50 to <1 microns. 
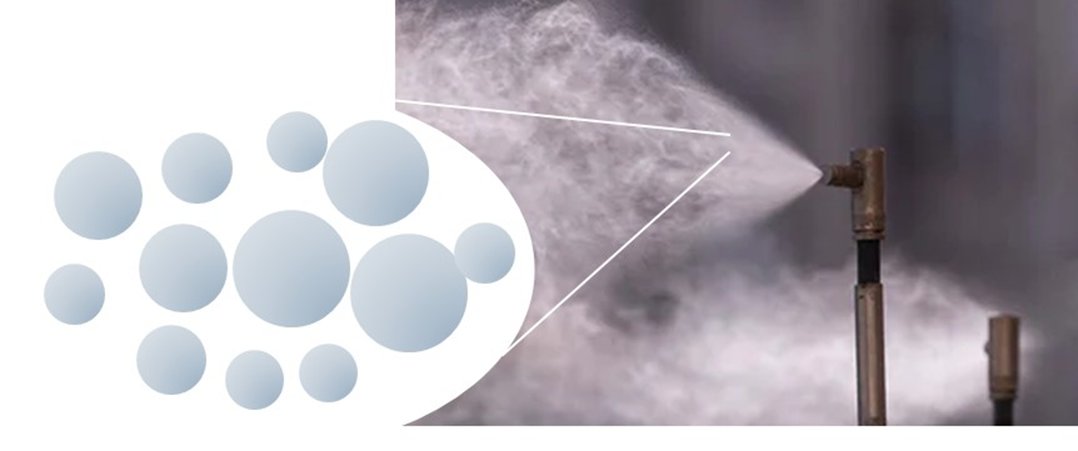
Once pure water is aerosolized, the droplet that is formed will begin to evaporate. All of the water will evaporate until the droplet is gone, where all of the water has converted to vapor.
This is why you won’t get soaked by the spray.
This is why you won’t get soaked by the spray.

The rate that the water droplet evaporates is largely dependent on:
-Droplet size
-Relative humidity
-Temperature
-Droplet size
-Relative humidity
-Temperature

At the same relative humidity and temperature, smaller droplets will change size much faster. Shown here is the size of water droplets evaporating into 80% humidity and 20C. 

None of the previous 3 slides may be all that surprising. And the bigger question is, what does this have to do with how mist cools an outdoor space?
The answer, as It turns out, is “everything”.
The answer, as It turns out, is “everything”.
Context: The body uses sweat to regulate its temperature. As the water in the sweat evaporates from the skin, the surface of the skin is cooled.
Energy is required to convert water from a liquid to a gas. Termed the “energy of vaporization”, this is the energy that is taken up by the water during evaporation.
en.wikipedia.org/wiki/Latent_he…
en.wikipedia.org/wiki/Latent_he…
Temperature is a measure of energy. As energy is transferred from the liquid water to water vapour, the temperature of the surface of the skin is cooled. 

An identical dynamic occurs in an evaporating mist. As each water droplet evaporates, the temperature of the aerosol droplet gets much cooler.
But how much?
But how much?

Shown here is the temperature of a 10 micronwater droplet evaporating into different relative humidities. In dry air, the droplet will reach temperatures well below freezing. 
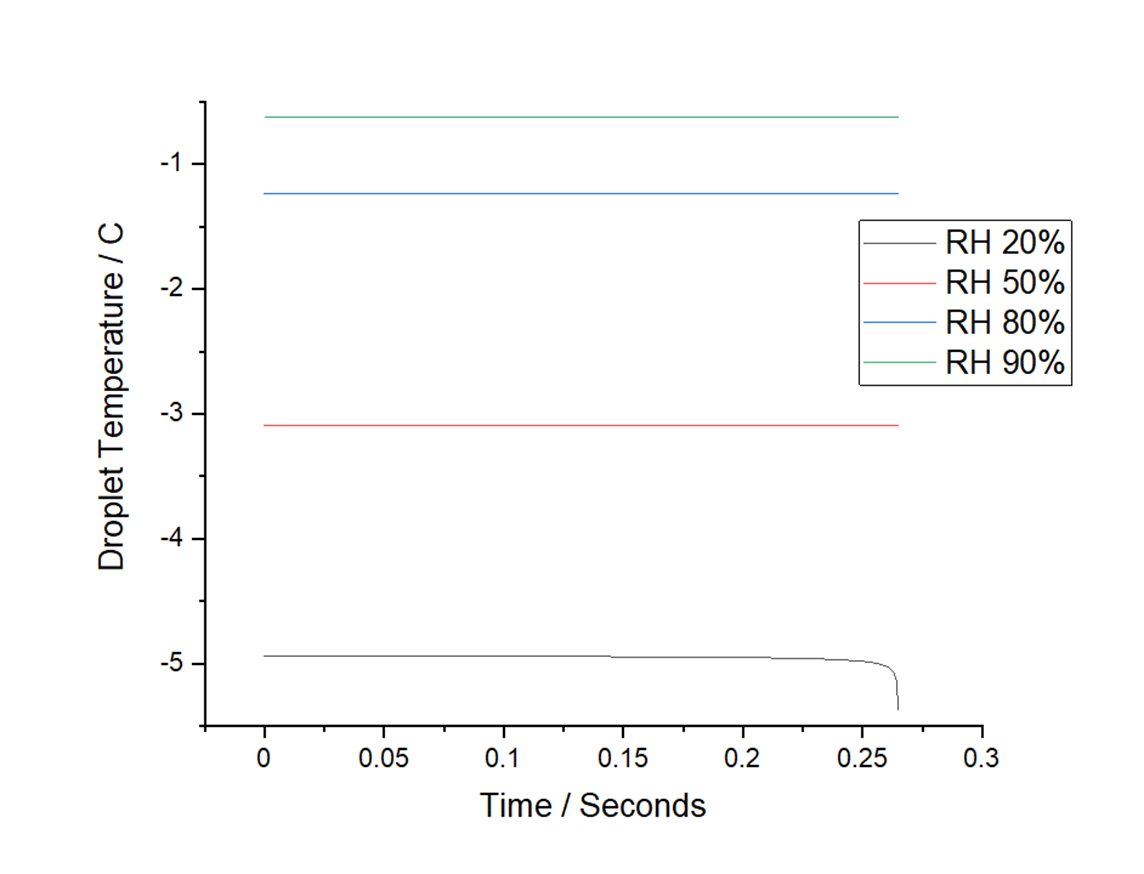
Fun Fact/Aside: Pure water droplets do not freeze at 0 C. Pure water droplets can reach as low as -37C before forming an ice crystal.
en.wikipedia.org/wiki/Supercool…
en.wikipedia.org/wiki/Supercool…
Alright, so the reason why these misting systems work so well is that the aerosol is incredibly cold. This cold mist in turn will lower the temperature of the surrounding air, leading to a comfortable environment. 

I hope you found that interesting. At the very least, you have a couple fun facts you can use on the patio this summer.
If you have any questions, please let me know.
If you have any questions, please let me know.
• • •
Missing some Tweet in this thread? You can try to
force a refresh