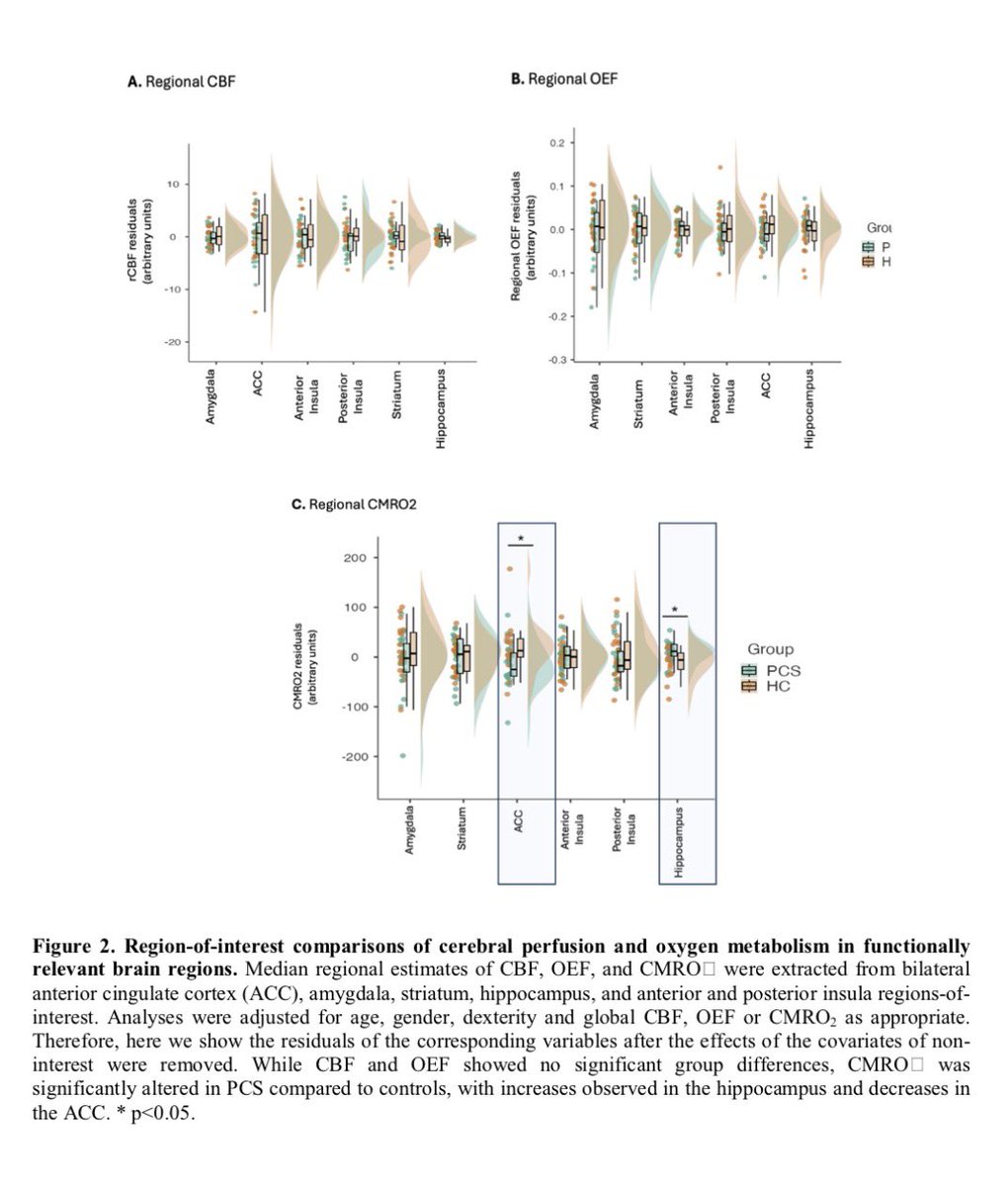
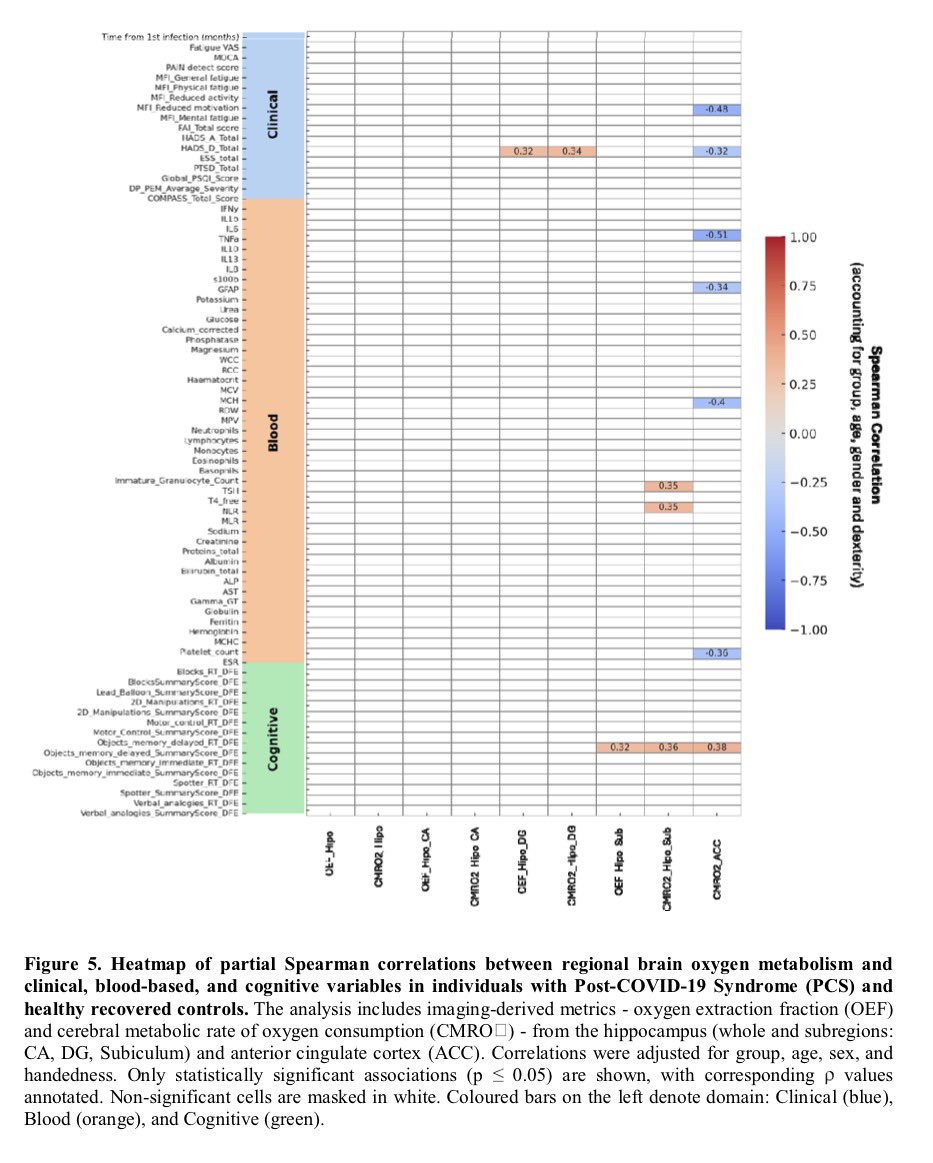
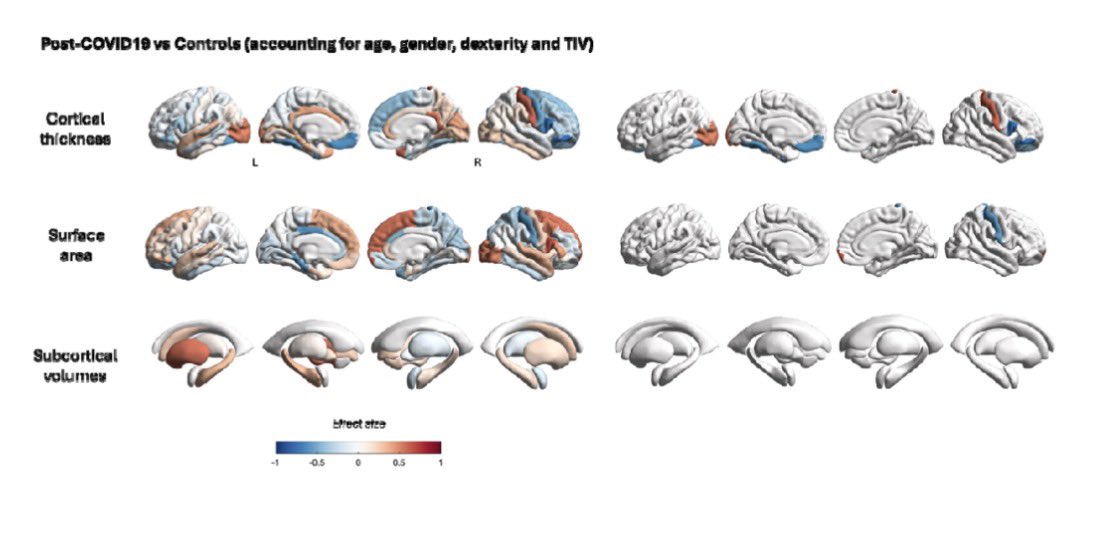
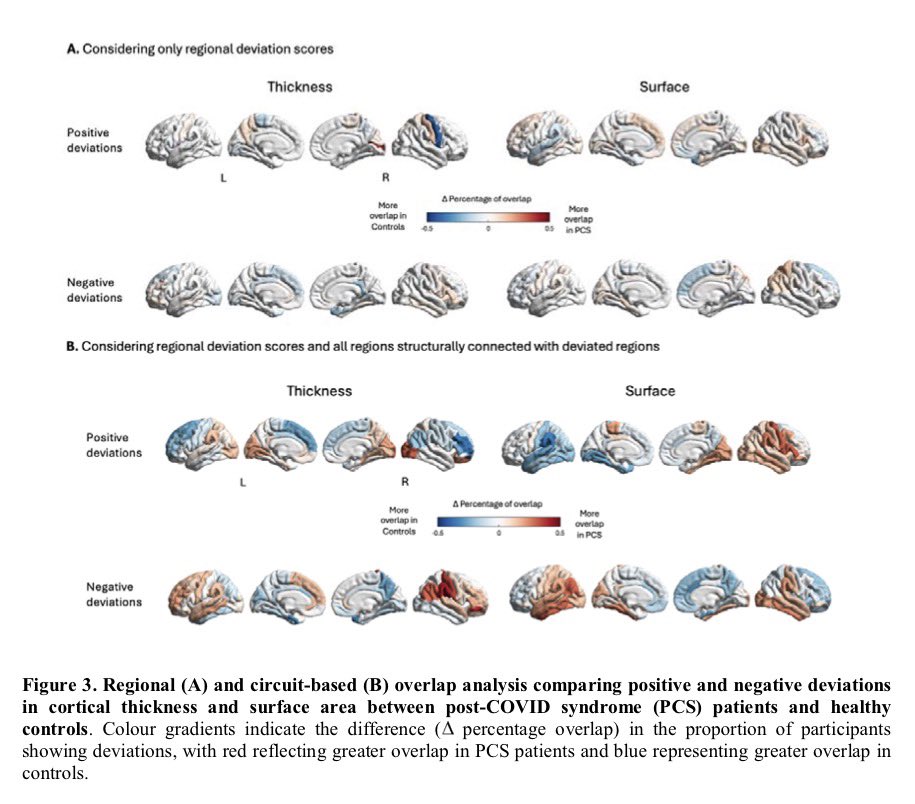
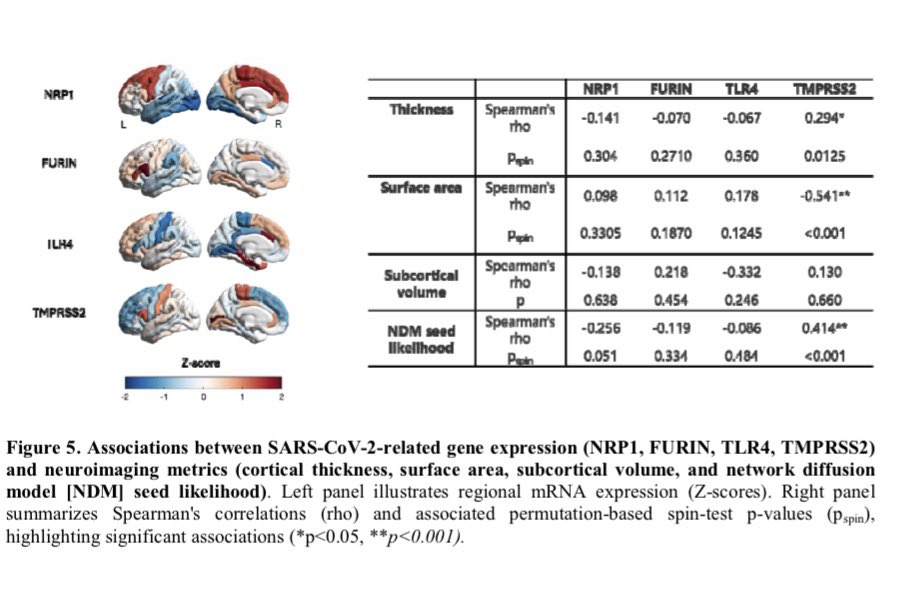
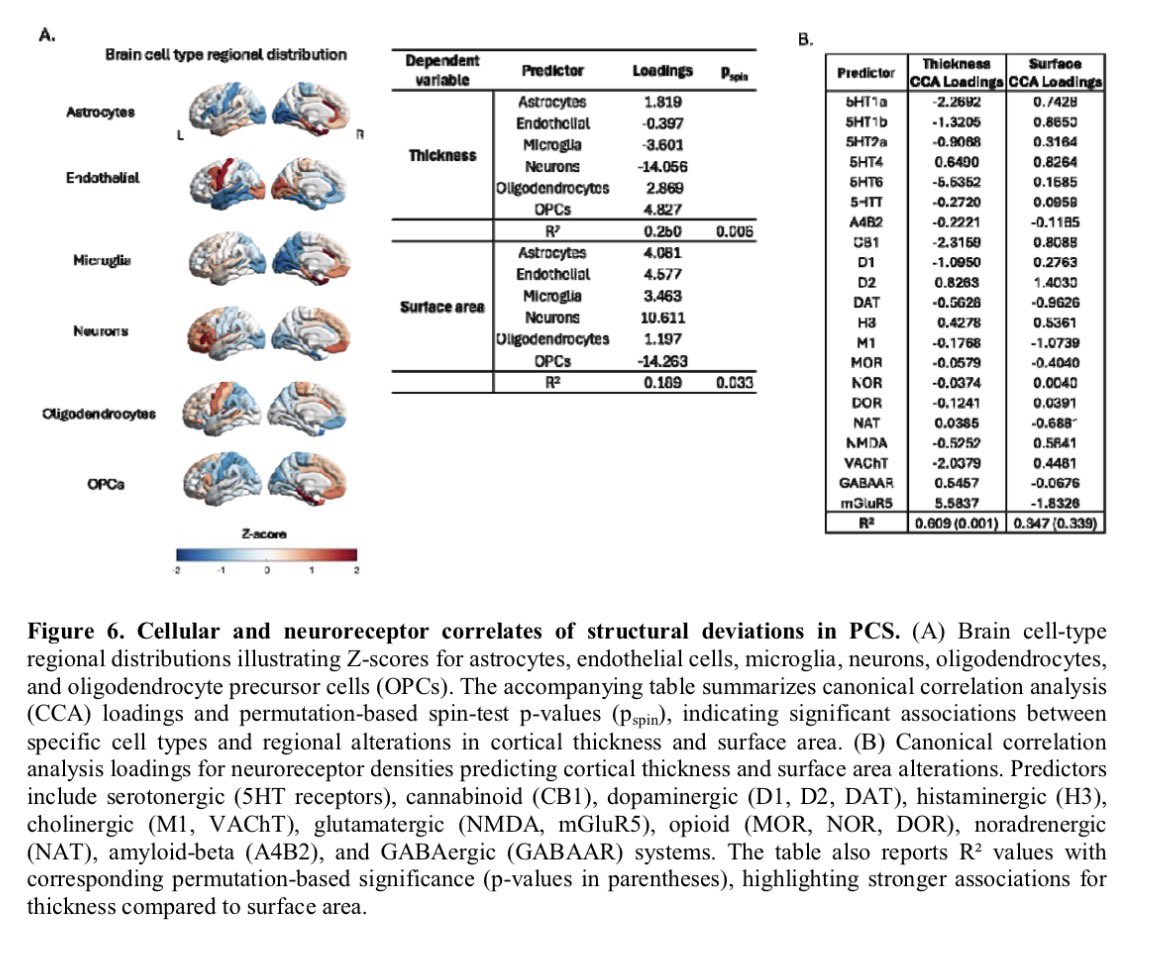
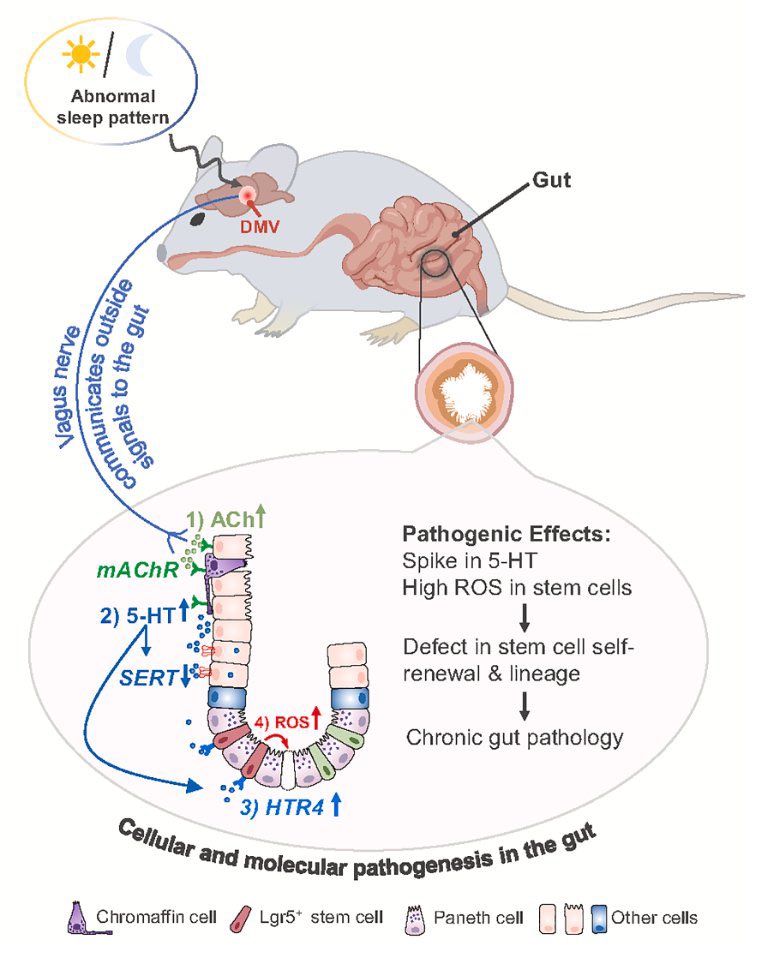
🔥 New research shows that sleep disturbance directly harms intestinal stem cells.
➡️ Acute sleep deprivation impaired stem-cell function in the gut, disrupting normal tissue renewal.
👉 Mechanism uncovered:
➡️ Sleep loss triggered aberrant vagus-nerve signaling from brain to gut, leading to intestinal stem-cell dysfunction.
➡️ This disrupted the gut’s ability to repair itself. 1/
➡️ Acute sleep deprivation impaired stem-cell function in the gut, disrupting normal tissue renewal.
👉 Mechanism uncovered:
➡️ Sleep loss triggered aberrant vagus-nerve signaling from brain to gut, leading to intestinal stem-cell dysfunction.
➡️ This disrupted the gut’s ability to repair itself. 1/
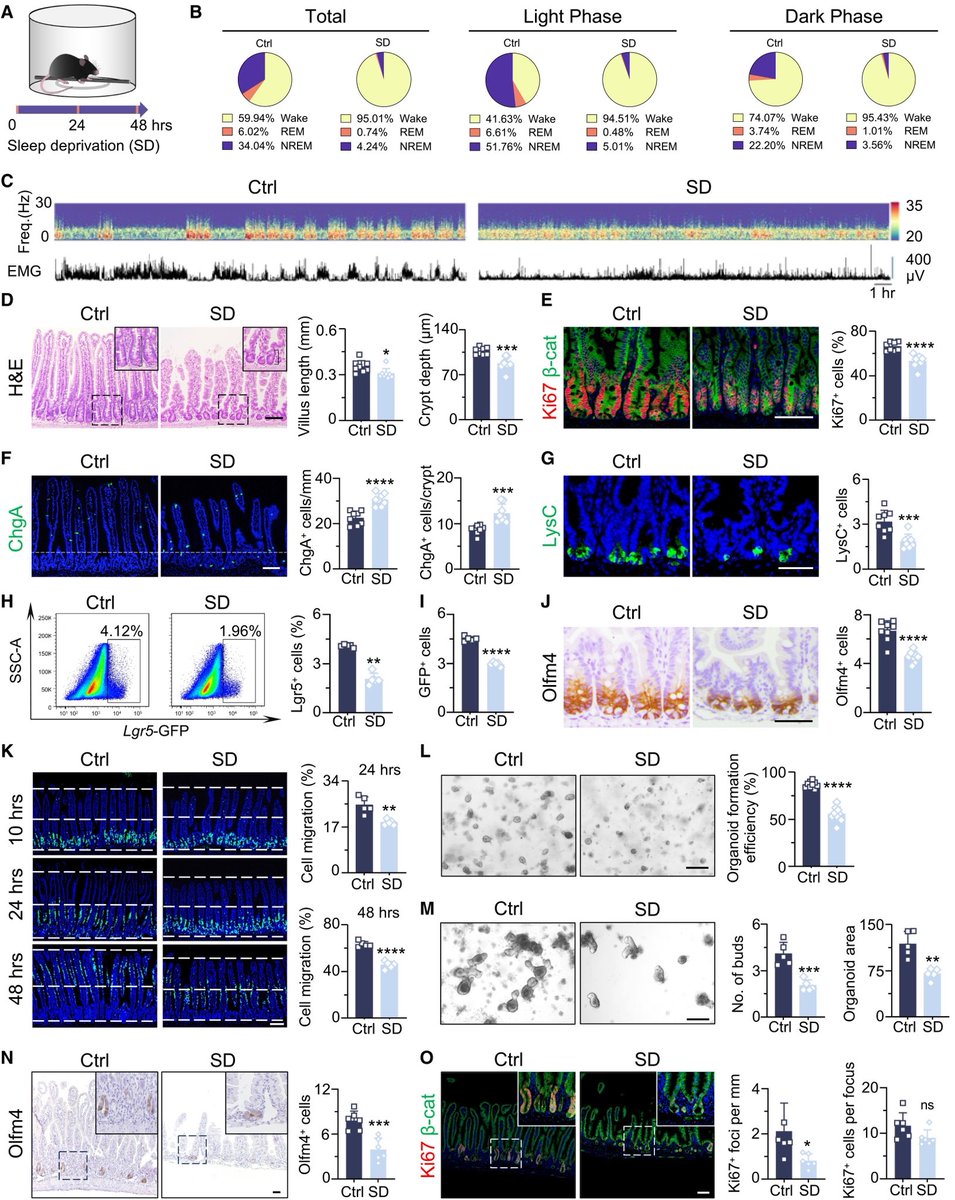
Key cellular changes after sleep deprivation:
• Shortened crypt-villus architecture
• Loss of Paneth cells
• Impaired intestinal stem-cell activity
All critical for maintaining gut health. 2/
• Shortened crypt-villus architecture
• Loss of Paneth cells
• Impaired intestinal stem-cell activity
All critical for maintaining gut health. 2/
👉 Important implication:
➡️ Sleep disorders may contribute to chronic gastrointestinal diseases by altering brain-to-gut neural signaling and stem-cell function.
⚠️ Sleep is not just rest—it is essential for tissue regeneration. 3/
➡️ Sleep disorders may contribute to chronic gastrointestinal diseases by altering brain-to-gut neural signaling and stem-cell function.
⚠️ Sleep is not just rest—it is essential for tissue regeneration. 3/
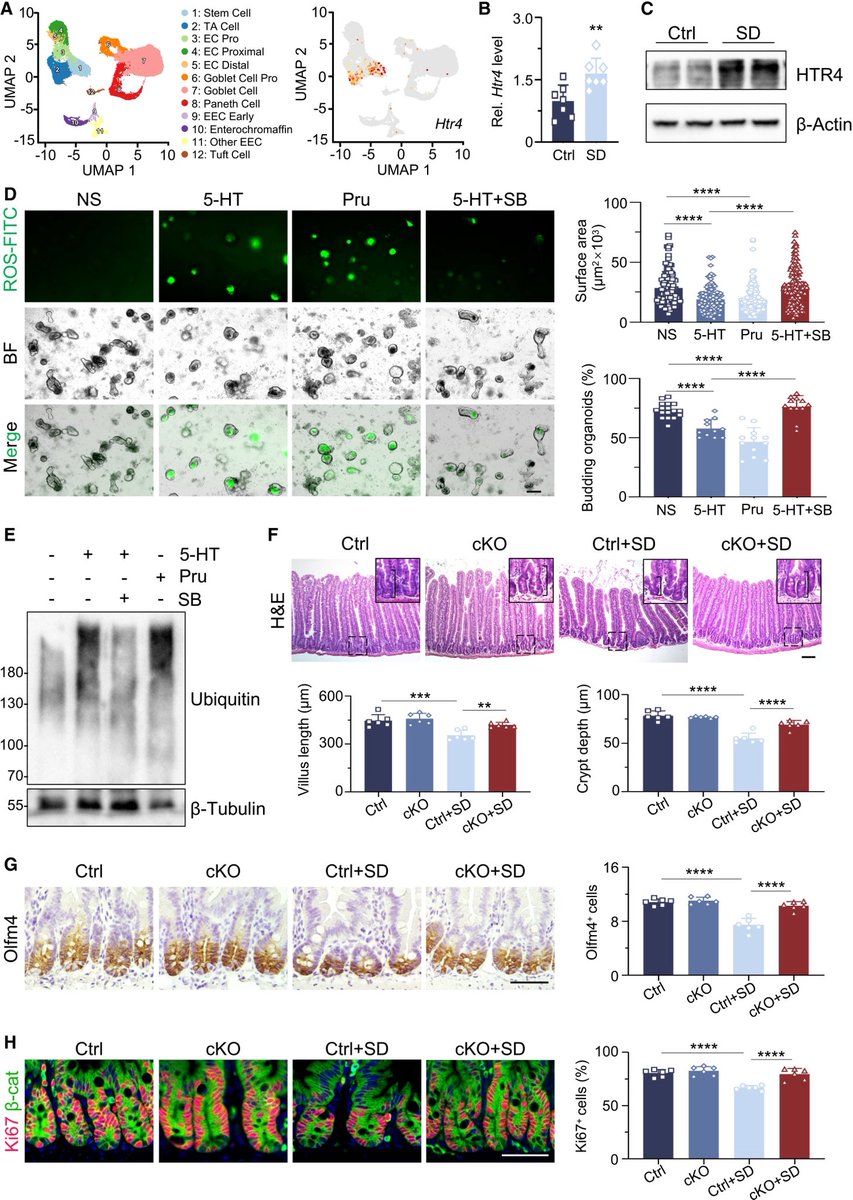
👉 Big picture:
➡️ The study reveals a direct brain–gut–stem cell axis, linking sleep quality to intestinal repair and long-term health.
➡️ Another reminder that sleep is a biological necessity, not a luxury. 4/
➡️ The study reveals a direct brain–gut–stem cell axis, linking sleep quality to intestinal repair and long-term health.
➡️ Another reminder that sleep is a biological necessity, not a luxury. 4/
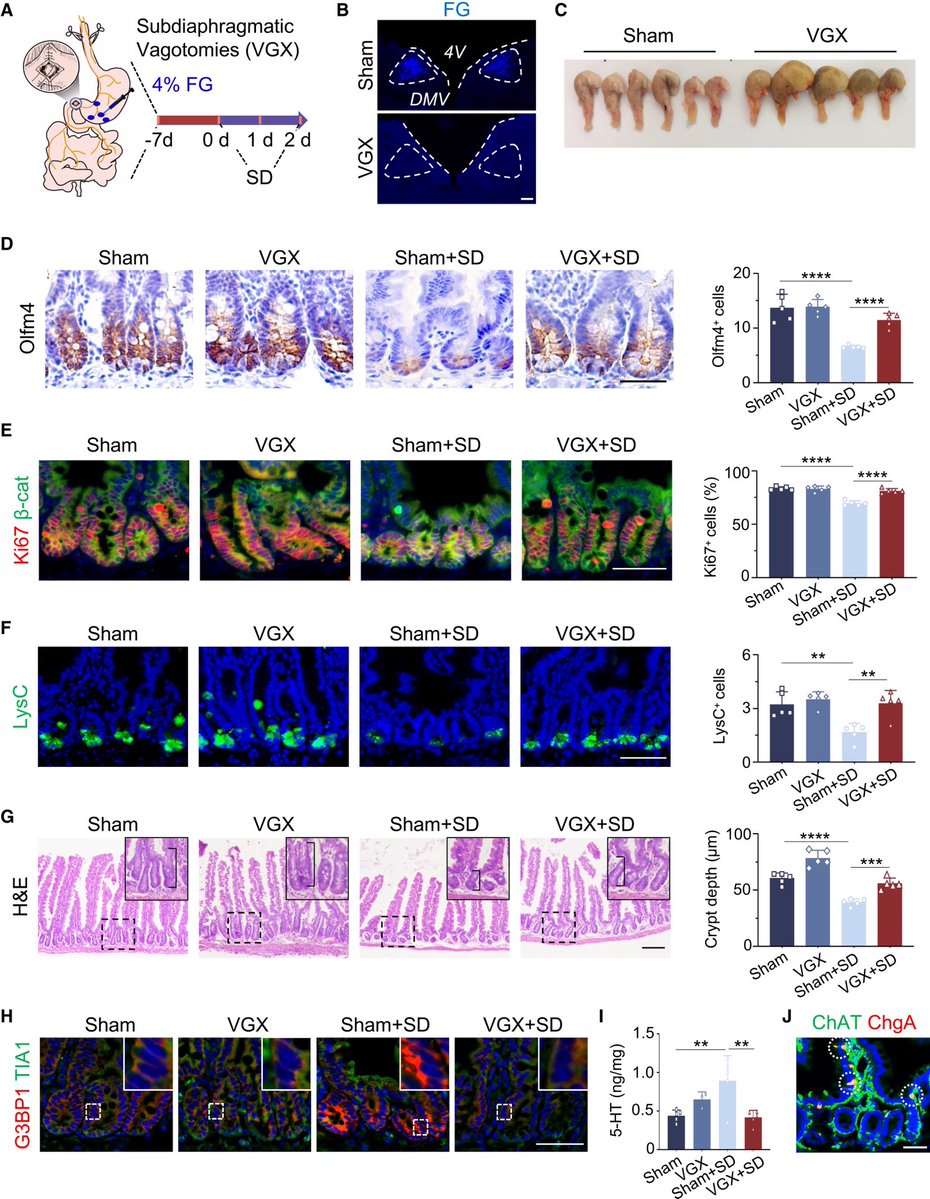
👉 Plain-language summary:
➡️ The study shows that lack of sleep can harm the intestine at a cellular level.
Sleep deprivation sends abnormal signals from the brain to the gut through the vagus nerve, which disrupts the function of intestinal stem cells that normally repair the gut lining.
This damage includes loss of key support cells and reduced ability of the intestine to regenerate. The findings suggest that good sleep is essential for maintaining gut health and long-term body function. 5/5
cell.com/cell-stem-cell…
➡️ The study shows that lack of sleep can harm the intestine at a cellular level.
Sleep deprivation sends abnormal signals from the brain to the gut through the vagus nerve, which disrupts the function of intestinal stem cells that normally repair the gut lining.
This damage includes loss of key support cells and reduced ability of the intestine to regenerate. The findings suggest that good sleep is essential for maintaining gut health and long-term body function. 5/5
cell.com/cell-stem-cell…
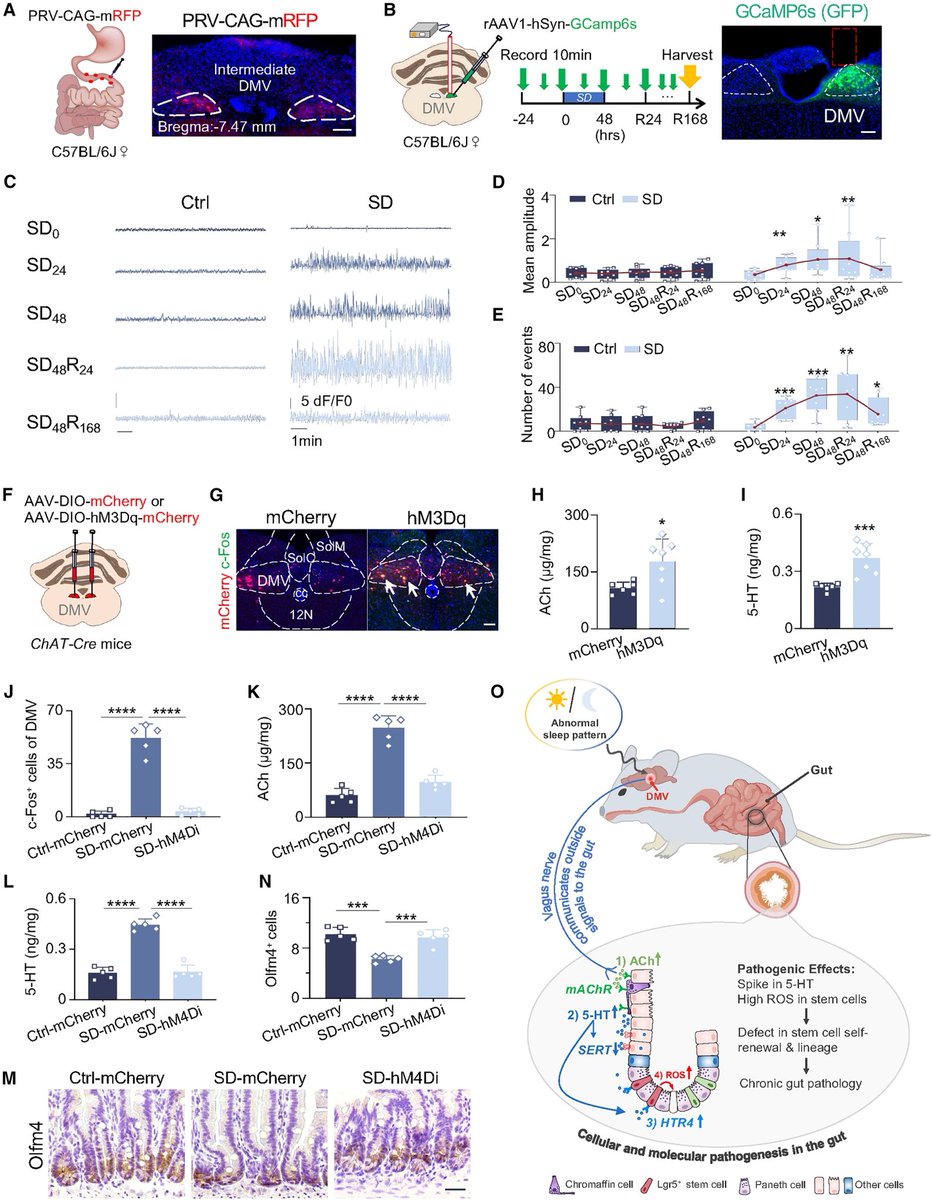
• • •
Missing some Tweet in this thread? You can try to
force a refresh