Addiction is not just about reward.
It reflects predictable dysfunction across brain systems governing behaviour, learning, and control.
Here are 5 core neurobiological processes underlying addiction and relapse: 🧵👇
(Click through to see all 5)
It reflects predictable dysfunction across brain systems governing behaviour, learning, and control.
Here are 5 core neurobiological processes underlying addiction and relapse: 🧵👇
(Click through to see all 5)

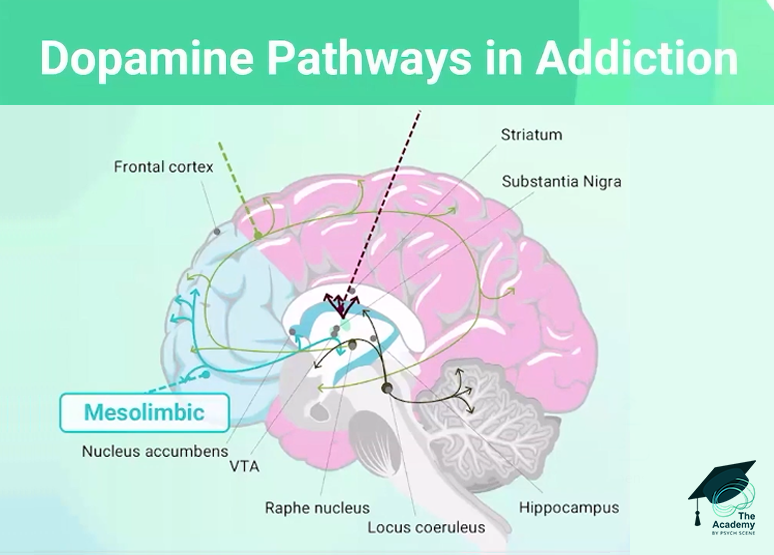
1/ Reward & Motivation
Chronic substance use floods the brain with dopamine, eventually downregulating D2 receptors.
This structurally shifts the brain from "liking" the drug (hedonic pleasure) to intensely "wanting" it (incentive salience).
The biological urge to use becomes overpowering, even when the substance ceases to provide actual pleasure.
Educating patients that the craving they feel is a structural reflex, not a reflection of character, relieves guilt and contextualises relapse.
Chronic substance use floods the brain with dopamine, eventually downregulating D2 receptors.
This structurally shifts the brain from "liking" the drug (hedonic pleasure) to intensely "wanting" it (incentive salience).
The biological urge to use becomes overpowering, even when the substance ceases to provide actual pleasure.
Educating patients that the craving they feel is a structural reflex, not a reflection of character, relieves guilt and contextualises relapse.
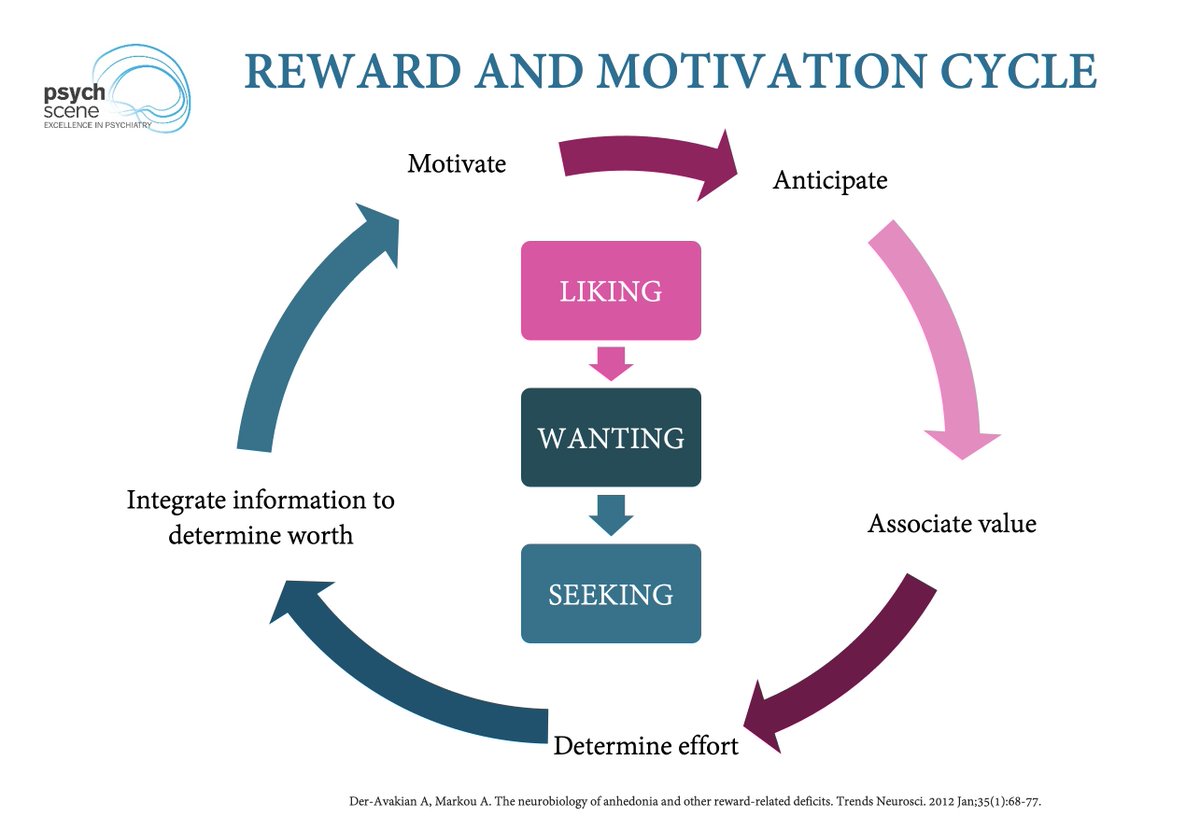
2/ Conditioning
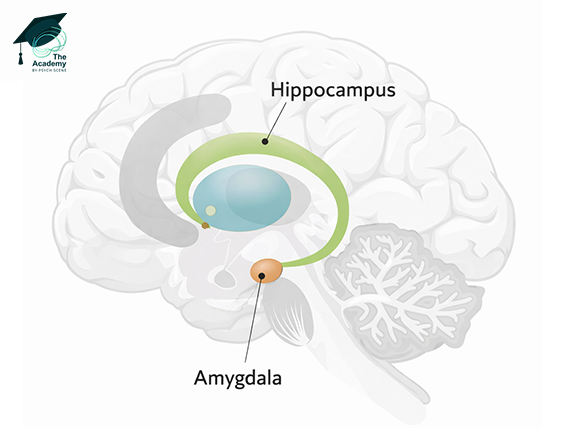
When a substance repeatedly spikes dopamine, the amygdala and hippocampus form pathological associative memories linking the high to neutral environmental cues (places, paraphernalia).
This Pavlovian learning rewires the brain to anticipate the substance, meaning a single stimulus can provoke involuntary cravings years into recovery.
It helps explain why conditioned cues can contribute to craving and relapse risk even after prolonged abstinence.
When a substance repeatedly spikes dopamine, the amygdala and hippocampus form pathological associative memories linking the high to neutral environmental cues (places, paraphernalia).
This Pavlovian learning rewires the brain to anticipate the substance, meaning a single stimulus can provoke involuntary cravings years into recovery.
It helps explain why conditioned cues can contribute to craving and relapse risk even after prolonged abstinence.
3/ Self-Regulation Impairment
Chronic use physically impairs the prefrontal cortex, dismantling the brain’s "brake" system.
This deficit reduces impulse inhibition, shifting behaviour to rigid, automatic habits mediated by the dorsal striatum.
Interventions must actively aim to strengthen executive function and disrupt automatic routines to help rebuild the patient's compromised cognitive control.
Chronic use physically impairs the prefrontal cortex, dismantling the brain’s "brake" system.
This deficit reduces impulse inhibition, shifting behaviour to rigid, automatic habits mediated by the dorsal striatum.
Interventions must actively aim to strengthen executive function and disrupt automatic routines to help rebuild the patient's compromised cognitive control.

4/ Negative Mood and Stress Reactivity
Late-stage addiction relies on negative reinforcement.
The extended amygdala activates stress pathways, inducing severe psychological agony known as allostasis.
Patients no longer use to chase a high; they use compulsively to escape this withdrawal-induced dysphoria.
Recognising this shift highlights that treatment must prioritise the physiological stabilisation of the stress axis, rather than just reward blockade.
Late-stage addiction relies on negative reinforcement.
The extended amygdala activates stress pathways, inducing severe psychological agony known as allostasis.
Patients no longer use to chase a high; they use compulsively to escape this withdrawal-induced dysphoria.
Recognising this shift highlights that treatment must prioritise the physiological stabilisation of the stress axis, rather than just reward blockade.
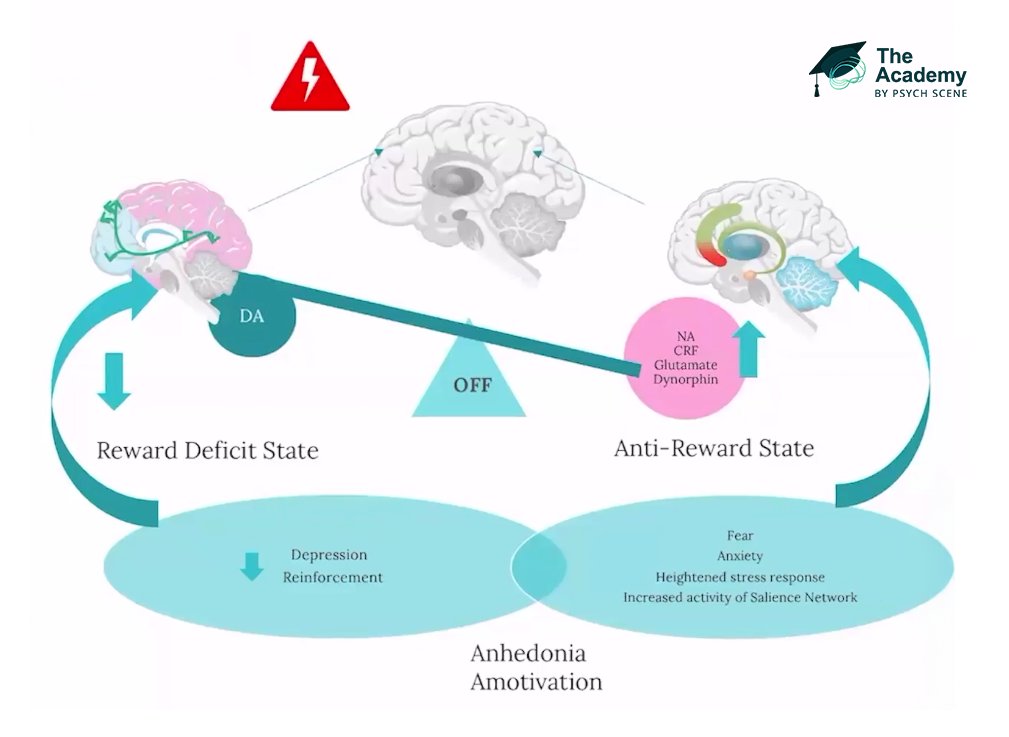
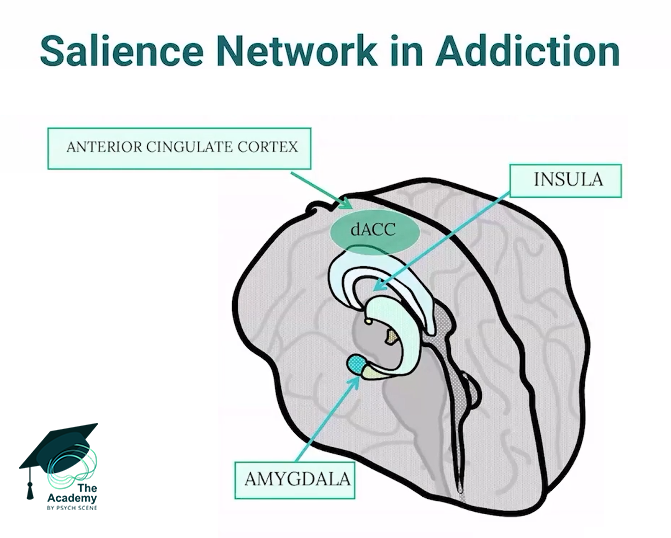
5/ Interoceptive Awareness
Addiction disrupts the insular cortex, which governs how we perceive internal bodily states.
This sensory dysregulation “causes” patients to misinterpret normal physiological signals, like mild anxiety or a racing heart, as severe withdrawal or an intense craving.
Incorporating mindfulness and biofeedback in treatment helps patients accurately reinterpret these somatic signals, preventing normal stress from leading to a relapse.
Addiction disrupts the insular cortex, which governs how we perceive internal bodily states.
This sensory dysregulation “causes” patients to misinterpret normal physiological signals, like mild anxiety or a racing heart, as severe withdrawal or an intense craving.
Incorporating mindfulness and biofeedback in treatment helps patients accurately reinterpret these somatic signals, preventing normal stress from leading to a relapse.
5 Core Neurobiological Processes Underlying Addiction and Relapse
1. Reward & Motivation: D2 downregulation shifts pleasure to intense “wanting”; psychoeducation reframes cravings and reduces guilt.
2. Conditioning: Learned associations mean cues (e.g., people, places, emotions) can trigger relapse even after abstinence.
3. Self-Regulation Impairment: PFC dysfunction weakens impulse control; interventions must rebuild executive function.
Continue...
1. Reward & Motivation: D2 downregulation shifts pleasure to intense “wanting”; psychoeducation reframes cravings and reduces guilt.
2. Conditioning: Learned associations mean cues (e.g., people, places, emotions) can trigger relapse even after abstinence.
3. Self-Regulation Impairment: PFC dysfunction weakens impulse control; interventions must rebuild executive function.
Continue...
4. Negative Mood & Stress: Late-stage addiction is associated with negative reinforcement via the extended amygdala (allostasis). Prioritise physiological stabilisation of the stress axis.
5. Interoceptive Awareness: Insular dysregulation misinterprets normal somatic signals as severe cravings. Incorporate biofeedback and mindfulness to help patients accurately reinterpret these cues.
5. Interoceptive Awareness: Insular dysregulation misinterprets normal somatic signals as severe cravings. Incorporate biofeedback and mindfulness to help patients accurately reinterpret these cues.
To learn more about addiction and its implications in clinical practice, click the link below and check our course in The Academy.
psychscene.co/419Eg9X
psychscene.co/419Eg9X
• • •
Missing some Tweet in this thread? You can try to
force a refresh