1/7) We are excited to share our preprint, where we present a method to measure thrombo-inflammation that lead to systemic thrombotic endothelialitis in Long COVID, other post-viral and inflammatory diseases in general: with @dbkell and lead author Anél Thompsonbiorxiv.org/content/10.648…
2/7) Why do we say Long COVID is characterized by thrombotic endothelialitis
In Long COVID, the blood does not behave normally
It becomes more “sticky” and inflammatory.
This affects how blood flows through small vessels and may contribute to many symptoms
In Long COVID, the blood does not behave normally
It becomes more “sticky” and inflammatory.
This affects how blood flows through small vessels and may contribute to many symptoms
3/7) We looked at a key interaction in the blood:
Platelets (cells that help clot)
+
Monocytes (immune cells)
When they stick together, they form platelet–monocyte aggregates (PMAs)
Platelets (cells that help clot)
+
Monocytes (immune cells)
When they stick together, they form platelet–monocyte aggregates (PMAs)
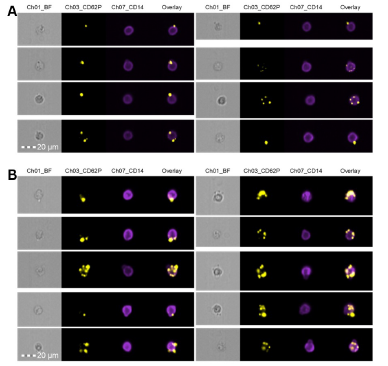
4/7) Why this matters
These aggregates are a sign that:
→ clotting is activated
→ the immune system is activated
→ blood vessels are under stress
In other words, they reflect ongoing inflammation in the circulation or thrombotic endothelialitis
These aggregates are a sign that:
→ clotting is activated
→ the immune system is activated
→ blood vessels are under stress
In other words, they reflect ongoing inflammation in the circulation or thrombotic endothelialitis
5/7) What did we find?
Long COVID patients had much higher levels of these aggregates
(~6 times higher than healthy individuals)
Not just more, but different! In Long COVID, more platelets attach to each immune cell
This suggests a stronger and more persistent activation state
Long COVID patients had much higher levels of these aggregates
(~6 times higher than healthy individuals)
Not just more, but different! In Long COVID, more platelets attach to each immune cell
This suggests a stronger and more persistent activation state
6/7) Why should clinicians care?
These aggregates can be measured from a simple blood sample:
They may help:
• identify patients with active thromboinflammation
• stratify disease severity
• monitor response to treatment
These aggregates can be measured from a simple blood sample:
They may help:
• identify patients with active thromboinflammation
• stratify disease severity
• monitor response to treatment
7/7) what is the bigger picture?
This fits with what we and others have shown:
Long COVID involves
→ inflammation
→ abnormal clotting
→ blood vessel dysfunction
All reinforcing each other resulting in thrombotic endothelialitis. This work provides a practical, measurable biomarker of this process.
This fits with what we and others have shown:
Long COVID involves
→ inflammation
→ abnormal clotting
→ blood vessel dysfunction
All reinforcing each other resulting in thrombotic endothelialitis. This work provides a practical, measurable biomarker of this process.
• • •
Missing some Tweet in this thread? You can try to
force a refresh






