Autoimmune Encephalitis (AE) is a relatively rare neuroinflammatory disorder.
However, what makes AE so hard for clinicians to spot isn't just its rarity.
Rather, it is that it can look psychiatric early.
To avoid misdiagnosis, here’s a 3-point AE diagnostic criteria clinicians should know:🧵👇
However, what makes AE so hard for clinicians to spot isn't just its rarity.
Rather, it is that it can look psychiatric early.
To avoid misdiagnosis, here’s a 3-point AE diagnostic criteria clinicians should know:🧵👇
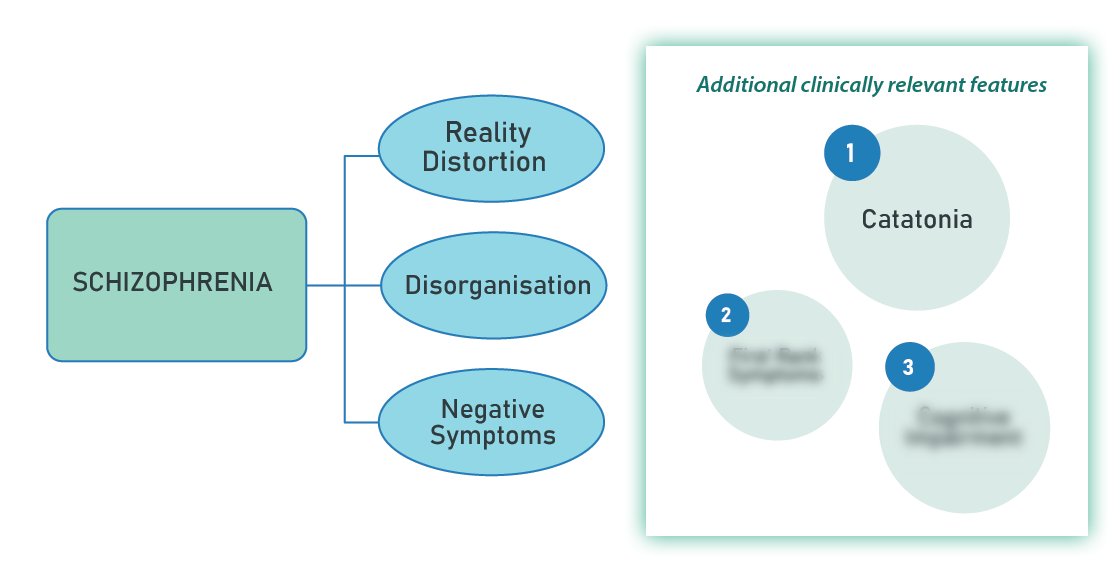
AE diagnosis can be considered when all three of the following criteria are met.
1/ Subacute Onset
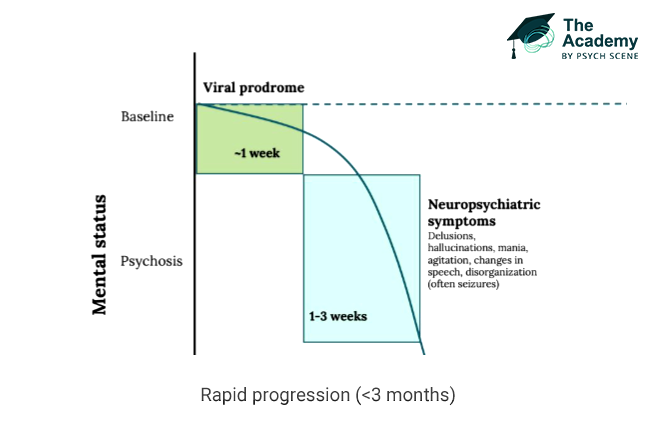
The first criterion is a subacute onset, defined as a rapid progression over less than 3 months.
Clinically, this may present as:
- working memory deficits or short-term memory loss
- altered mental status
- psychiatric symptoms
The key signal is not just symptom type, but how quickly the syndrome evolves.
1/ Subacute Onset
The first criterion is a subacute onset, defined as a rapid progression over less than 3 months.
Clinically, this may present as:
- working memory deficits or short-term memory loss
- altered mental status
- psychiatric symptoms
The key signal is not just symptom type, but how quickly the syndrome evolves.
2/ At Least One Supportive Neurological or Paraclinical Feature
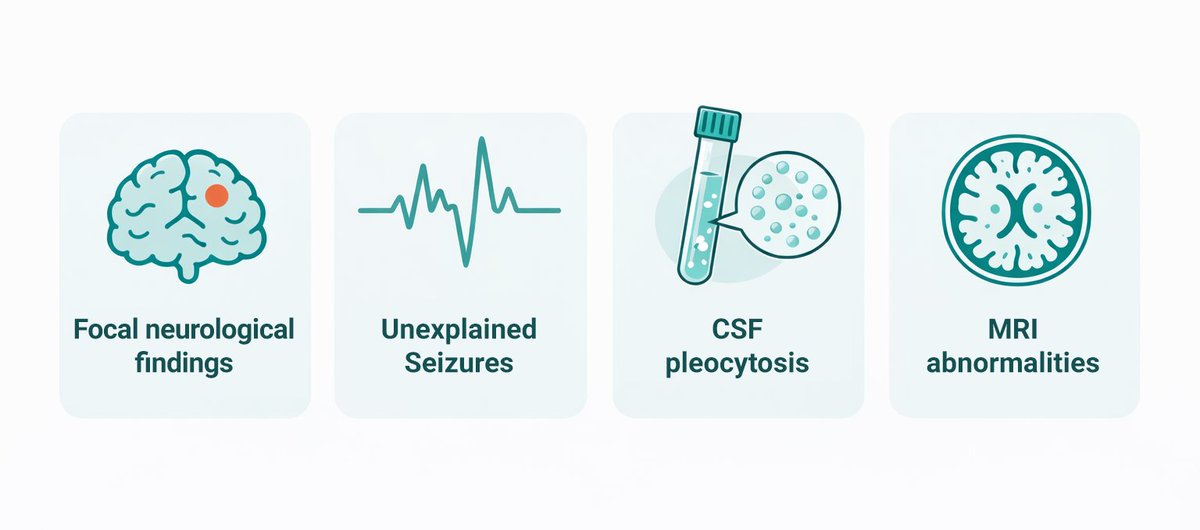
The second criterion requires at least one of the following:
- new focal CNS findings
- seizures not explained by a previously known seizure disorder
- CSF pleocytosis
- MRI features suggestive of encephalitis
This is where the picture starts to move beyond a purely psychiatric presentation.
The second criterion requires at least one of the following:
- new focal CNS findings
- seizures not explained by a previously known seizure disorder
- CSF pleocytosis
- MRI features suggestive of encephalitis
This is where the picture starts to move beyond a purely psychiatric presentation.
3/ Exclude Alternative Causes
The third criterion requires reasonable exclusion of alternative causes.
So the task is not only to identify a compatible syndrome, but to show that another explanation does not account for it.
Here, altered mental status may include reduced or altered consciousness, lethargy, or personality change.
Supportive MRI findings may include T2-FLAIR hyperintensity in the medial temporal lobes or in multifocal grey or white matter areas compatible with inflammation.
The third criterion requires reasonable exclusion of alternative causes.
So the task is not only to identify a compatible syndrome, but to show that another explanation does not account for it.
Here, altered mental status may include reduced or altered consciousness, lethargy, or personality change.
Supportive MRI findings may include T2-FLAIR hyperintensity in the medial temporal lobes or in multifocal grey or white matter areas compatible with inflammation.
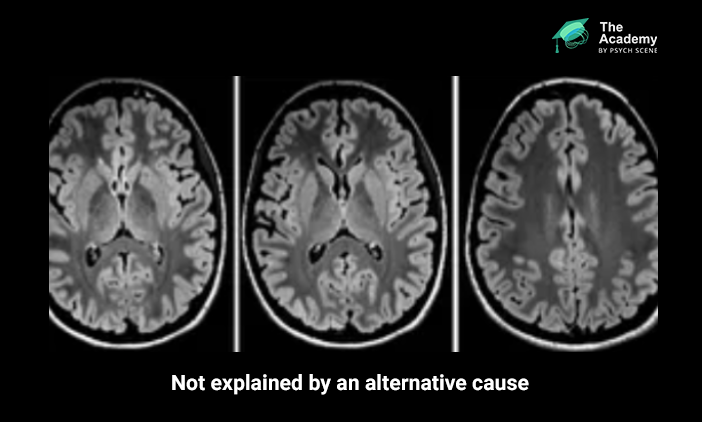
What happens if the clinical picture is phenotypically indicative, but one of the first three criteria is not entirely met?
A diagnosis of definite limbic encephalitis can still be confirmed in these instances, but it requires the explicit detection of highly specific antibodies against cell-surface, synaptic, or onconeural proteins.
A diagnosis of definite limbic encephalitis can still be confirmed in these instances, but it requires the explicit detection of highly specific antibodies against cell-surface, synaptic, or onconeural proteins.

3-Point Diagnostic Criteria for Autoimmune Encephalitis
A possible diagnosis of autoimmune encephalitis requires all three of these criteria:
1. Subacute Onset: Progression of memory deficits, altered mental status, or psychiatric symptoms in under 3 months.
2. Supportive Features: Presence of new focal CNS findings, unexplained seizures, CSF pleocytosis, or MRI abnormalities.
3. Reasonable Exclusion: Systematic elimination of alternative infectious, toxic, or metabolic aetiologies.
A possible diagnosis of autoimmune encephalitis requires all three of these criteria:
1. Subacute Onset: Progression of memory deficits, altered mental status, or psychiatric symptoms in under 3 months.
2. Supportive Features: Presence of new focal CNS findings, unexplained seizures, CSF pleocytosis, or MRI abnormalities.
3. Reasonable Exclusion: Systematic elimination of alternative infectious, toxic, or metabolic aetiologies.
💡 Psych Scene Tip: In suspected AE, shift from symptom description to syndrome verification.
If psychiatric symptoms are rapidly evolving, ask whether there is at least one supportive neurological or paraclinical feature, then actively rule out competing explanations rather than defaulting to a primary psychiatric label.
If psychiatric symptoms are rapidly evolving, ask whether there is at least one supportive neurological or paraclinical feature, then actively rule out competing explanations rather than defaulting to a primary psychiatric label.
To learn more about the neurobiological intricacies of Autoimmune Encephalitis and how to apply the AE diagnostic criteria in practice, click the link below and check out our course on Autoimmune Psychosis & Autoimmune Encephalitis inside The Academy:
psychscene.co/4sZTzyr
psychscene.co/4sZTzyr
• • •
Missing some Tweet in this thread? You can try to
force a refresh