
physician-scientist, author of SUPER AGERS https://t.co/ZEdooyyJpP
and Ground Truths: https://t.co/YhatcBT0hA
How to get URL link on X (Twitter) App


 In 2010, when @GoogleDeepMind was formed, well before deep learning was taking hold, @demishassabis predicted AGI by 2030 and called out the use of agents
In 2010, when @GoogleDeepMind was formed, well before deep learning was taking hold, @demishassabis predicted AGI by 2030 and called out the use of agents
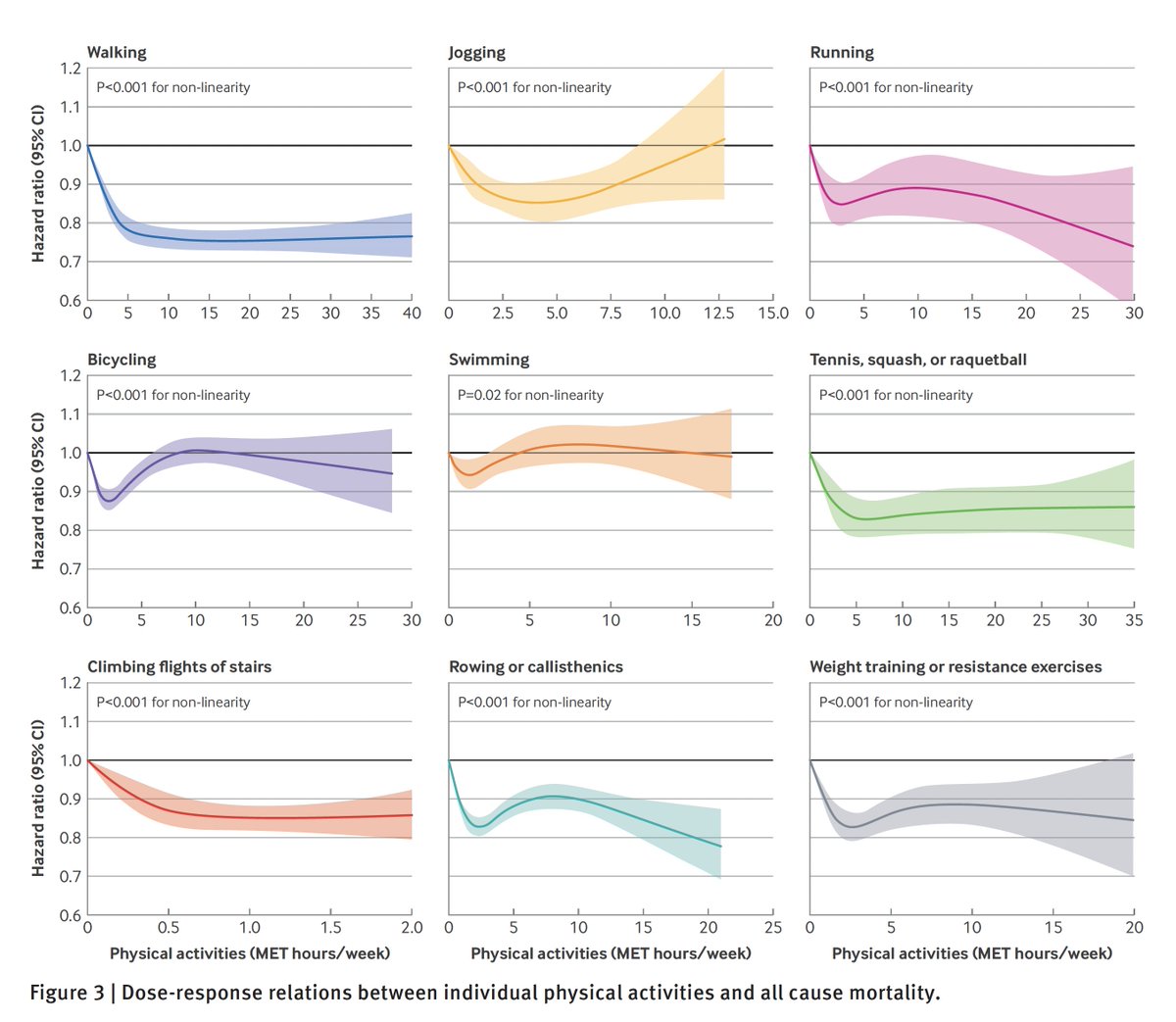
 2. Engaging in > 1 type of physical activity was generally correlated with better outcomes compared with 1 type (T1,2,3)
2. Engaging in > 1 type of physical activity was generally correlated with better outcomes compared with 1 type (T1,2,3) 
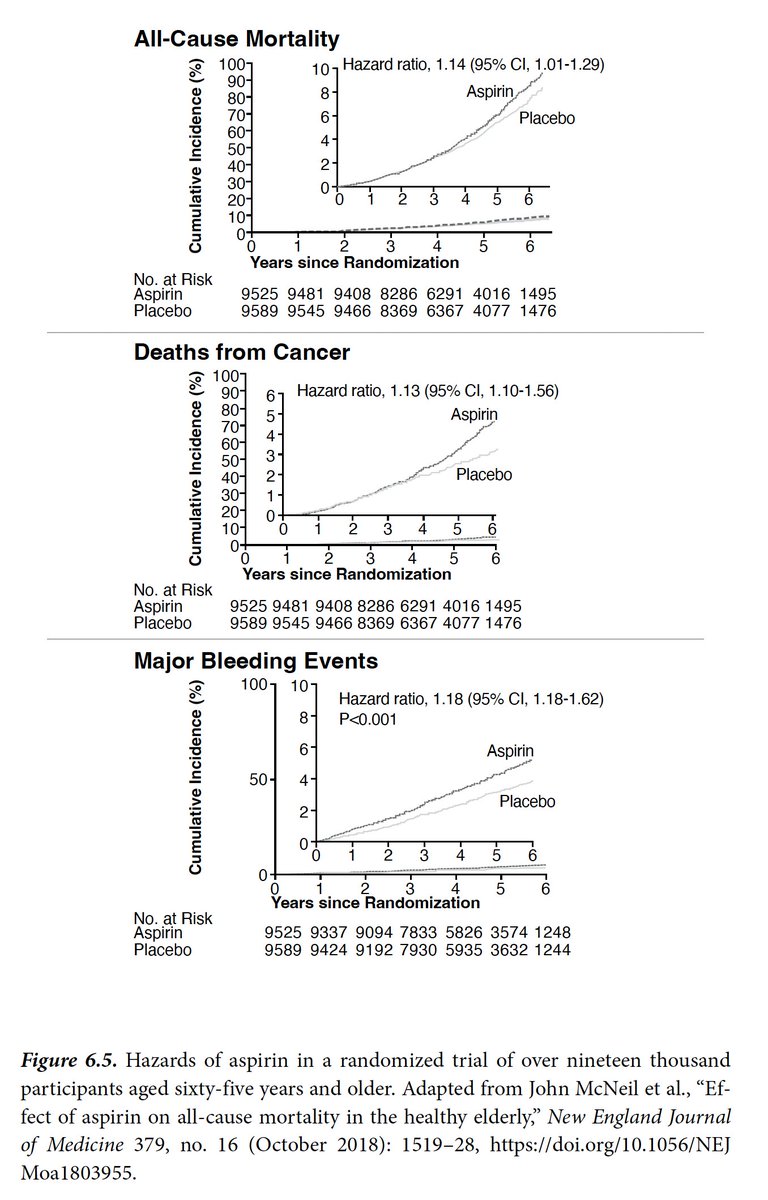
 President Trump takes 325 mg aspirin/day. The randomized trials tested 75-81 mg/day. His doctors recommended low-dose aspirin for heart event prevention. That recommendation is ill-founded based upon best evidence in older individuals.
President Trump takes 325 mg aspirin/day. The randomized trials tested 75-81 mg/day. His doctors recommended low-dose aspirin for heart event prevention. That recommendation is ill-founded based upon best evidence in older individuals.

 Inflammaging: with aging comes untoward inflammation, a summary graphic
Inflammaging: with aging comes untoward inflammation, a summary graphic 

 Our immune system as we age: the path to immunosenesence and inflammaging
Our immune system as we age: the path to immunosenesence and inflammaging 

 I review 4 new reports, summarized here:
I review 4 new reports, summarized here: 
 Here's the response to exercise
Here's the response to exercise 
https://x.com/EricTopol/status/1901957986727117069

 These are the specific foods that were linked with healthy aging and other outcomes. Green-favorable; Red-unfavorable
These are the specific foods that were linked with healthy aging and other outcomes. Green-favorable; Red-unfavorable 


 The human brain:
The human brain: 

 2. Report of the 13-year-old Canadian girl who developed acute respiratory failure. Sequence of her H5N1 revealed 3 mutations that may be linked to higher virulence and adaptation to human cell binding
2. Report of the 13-year-old Canadian girl who developed acute respiratory failure. Sequence of her H5N1 revealed 3 mutations that may be linked to higher virulence and adaptation to human cell binding 


 Such as Programming T cells with brain-specific proteins and payloads
Such as Programming T cells with brain-specific proteins and payloads 
 In other GLP-1 new publications today
In other GLP-1 new publications today



 science.org/toc/stm/16/773
science.org/toc/stm/16/773


 A clip from our conversation. Unknowingly, @AkassoglouLab was gearing up for understanding this complex pathophysiology for many years before Covid hit
A clip from our conversation. Unknowingly, @AkassoglouLab was gearing up for understanding this complex pathophysiology for many years before Covid hit

 "A major finding from our analyses was that the risk
"A major finding from our analyses was that the risk
 It's the deletion 31/31 that makes the KP.3.1.1 spike different, but otherwise 2 mutations away from KP.2 (R346T and Q493E)
It's the deletion 31/31 that makes the KP.3.1.1 spike different, but otherwise 2 mutations away from KP.2 (R346T and Q493E) 



 This variant growth advantage plot by @BenjMurrell (H/T @siamosolocani) shows why this is the case. Note KP.3 is the one at far left w/ almost 3-fold advantage to JN.1.
This variant growth advantage plot by @BenjMurrell (H/T @siamosolocani) shows why this is the case. Note KP.3 is the one at far left w/ almost 3-fold advantage to JN.1.
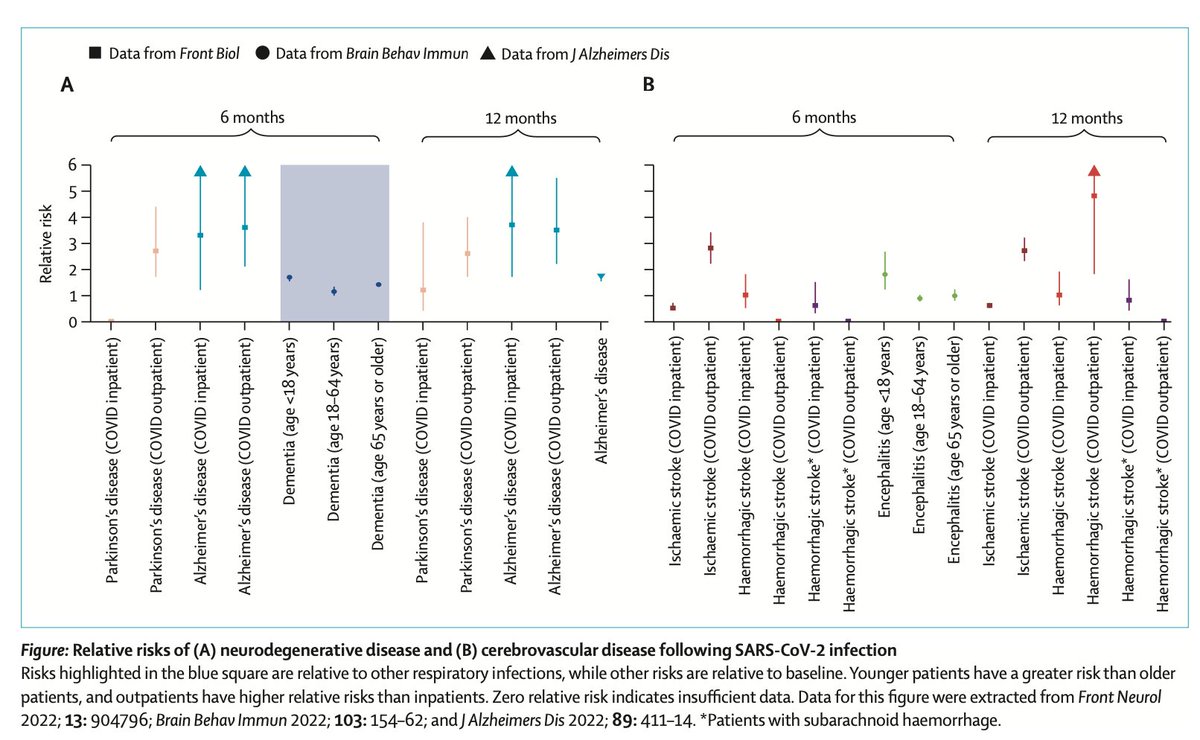
 2. Inflammation in patients with COVID-19, and controlled experiments show prolonged neuro-inflammation after mild SARS-CoV-2 infection
2. Inflammation in patients with COVID-19, and controlled experiments show prolonged neuro-inflammation after mild SARS-CoV-2 infection