Investigating the extent of acceptance worldwide of the role of @Bioconlimited's #Itolizumab in the treatment of #Psoriasis - a disease that afflicts upto 2% of the population. I conducted a search on PubMed.
molecules (apremilast, tofacitinib, ponesimod), anti-TNF alpha (etanercept, infliximab, adalimumab, certolizumab), as well as..
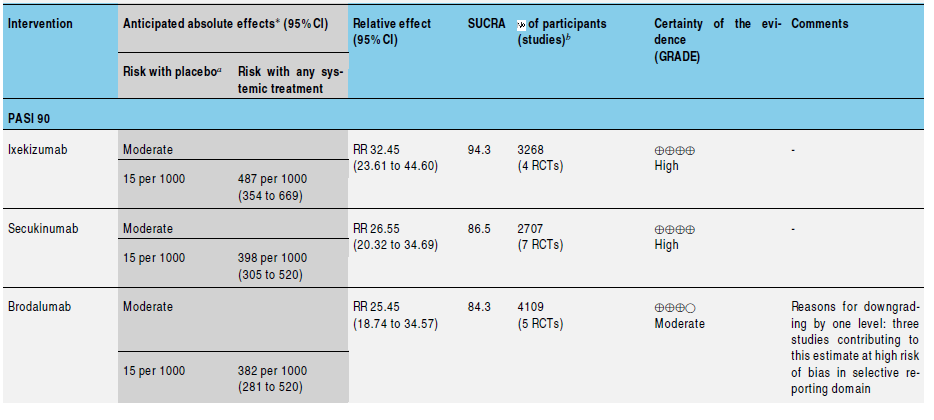
(ustekinumab), anti-IL17 (secukinumab, ixekizumab, brodalumab), anti-IL23 (guselkumab, tildrakizumab), and other biologics (alefacept, itolizumab) for patients with moderate to severe psoriasis and to rank these treatments according to their efficacy
and safety.