"These are some of the most important clinical trials in history, affecting a vast majority of the planet’s population. It’s hard to imagine how much higher the stakes can be to get this right. Cutting corners should not be an option." 1/
nytimes.com/2020/09/22/opi…
@nytopinion
nytimes.com/2020/09/22/opi…
@nytopinion
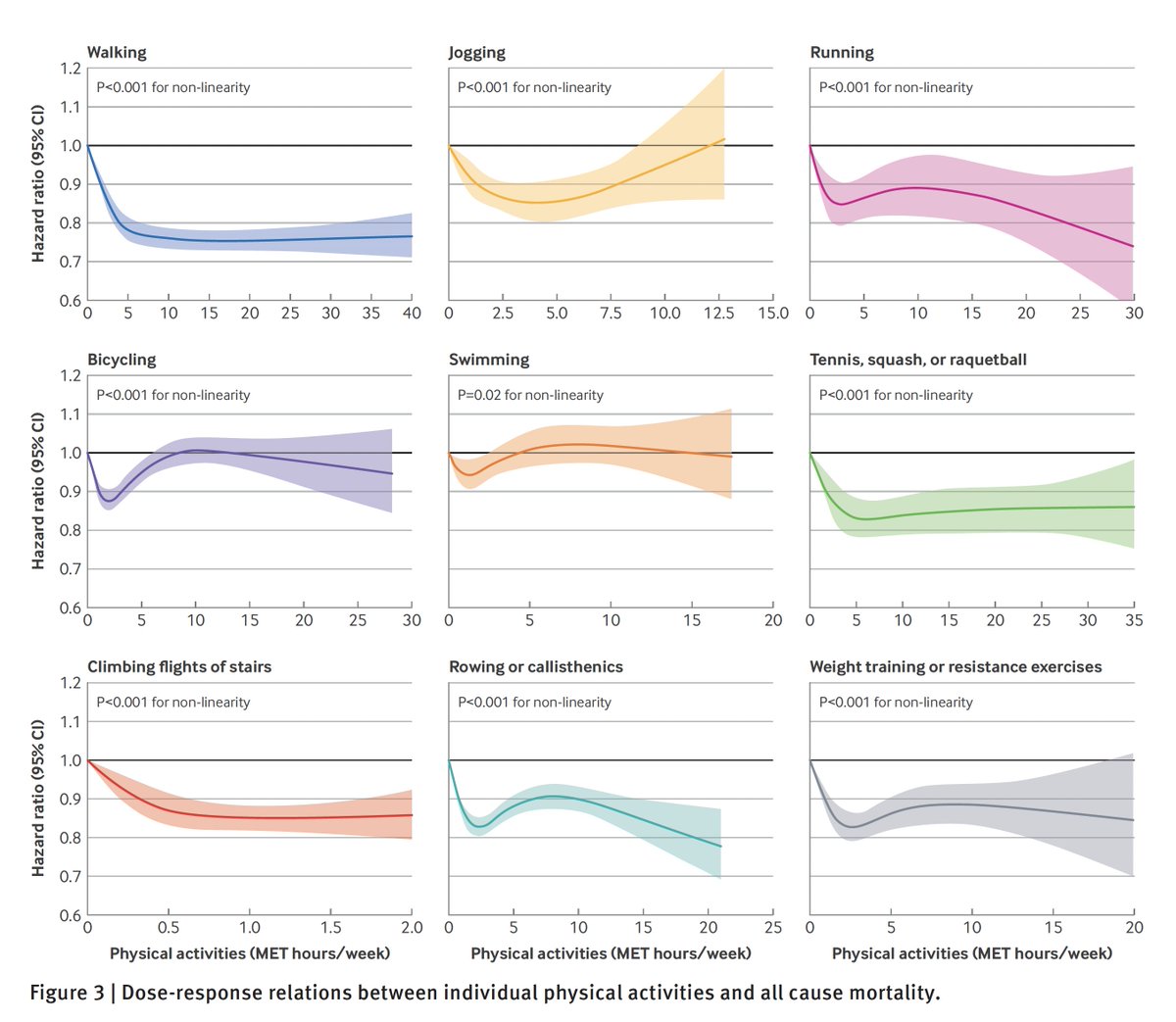
The primary endpoint in these trials— infections—will likely be mostly mild. For example, in the @pfizer trial, a sore throat and a + PCR test counts as an event. Are these the infections that we want to suppress with a vaccine? Is that a good proxy of effectiveness? 2/
There are interim analyses, some w/ stopping rules during the trials
—4 looks for Pfizer, 1st is at 32 events
—2 looks for Moderna, 1 look in Astra Zeneca (US) trial
—A trial could be stopped on the basis of very limited number and severity of such events, declaring efficacy 3/
—4 looks for Pfizer, 1st is at 32 events
—2 looks for Moderna, 1 look in Astra Zeneca (US) trial
—A trial could be stopped on the basis of very limited number and severity of such events, declaring efficacy 3/
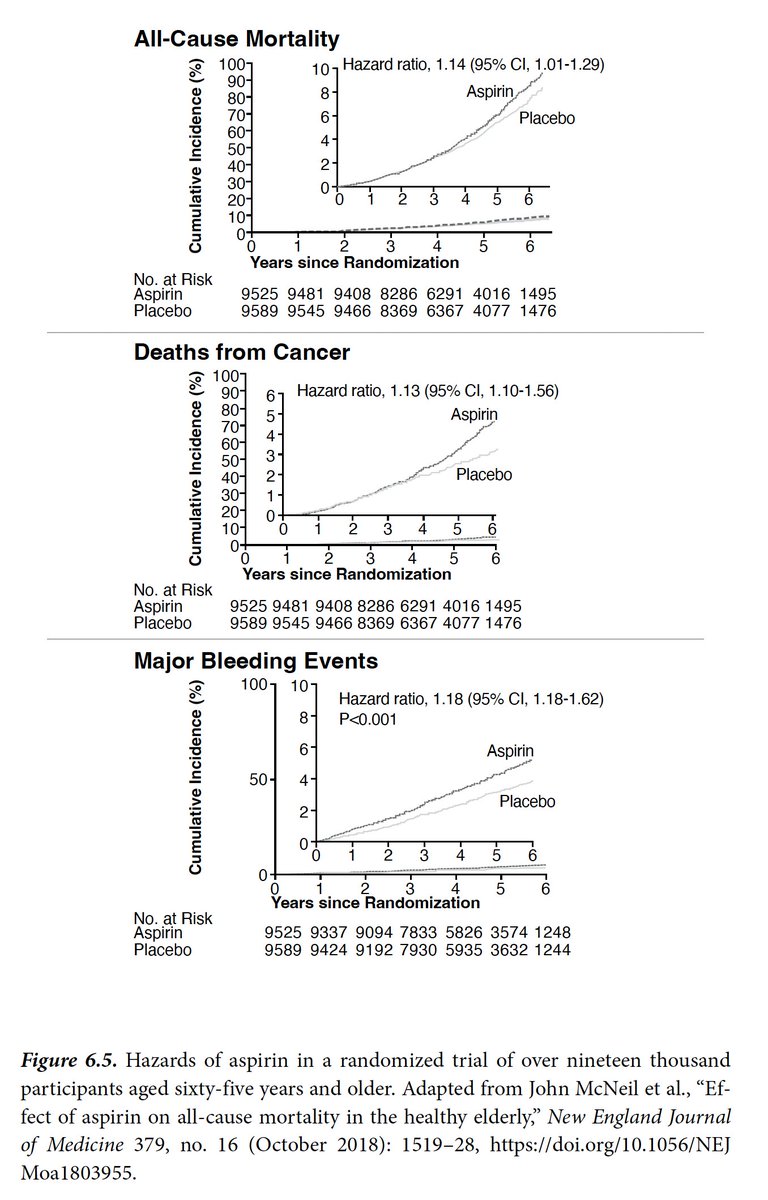
"Giving a vaccine to hundreds of millions of healthy people based on such limited data requires a real leap of faith."
Each trial has 30K-44,000 participants.
There's a difference of fulfilling a statistical endpoint vs extrapolation to the masses. Compelling evidence req'd 4/
Each trial has 30K-44,000 participants.
There's a difference of fulfilling a statistical endpoint vs extrapolation to the masses. Compelling evidence req'd 4/
Compelling evidence would be to power the trials on moderate to severe covid illness, which is a secondary endpoint of the trials. That adjustment to the 1° endpoint could easily be made by an amendment to the protocols. 5/
It is worth taking the extra weeks to get this right—to ice it—so that we have unequivocal proof of efficacy and a longer temporal window into safety.
Further, it is essential these trial go to completion of the 150-164 events. 6/
Further, it is essential these trial go to completion of the 150-164 events. 6/
Any shortcuts to "declare a winner" (which has major ramifications👇) would be based on a soft endpoint and incomplete ascertainment of safety and efficacy. There should be no emergency approval (EUA) by @US_FDA until a/the trial(s) are completed 7/ 

We are well aware of the company & political interests to obtain an EUA ASAP. And that @HHSgov can override FDA.
It will take all of us to stand up for proper completion of the trials, avoiding shortcuts, getting this right. This will promote public trust, which is critical. 8/
It will take all of us to stand up for proper completion of the trials, avoiding shortcuts, getting this right. This will promote public trust, which is critical. 8/
And this FDA announcement today is most welcome, moving in the right direction. #NoRushJob
washingtonpost.com/health/2020/09…
w/ @PeterHotez @califf001 @DrPaulOffit
by @lauriemcginley2 and @Carolynyjohnson 9/

washingtonpost.com/health/2020/09…
w/ @PeterHotez @califf001 @DrPaulOffit
by @lauriemcginley2 and @Carolynyjohnson 9/


• • •
Missing some Tweet in this thread? You can try to
force a refresh