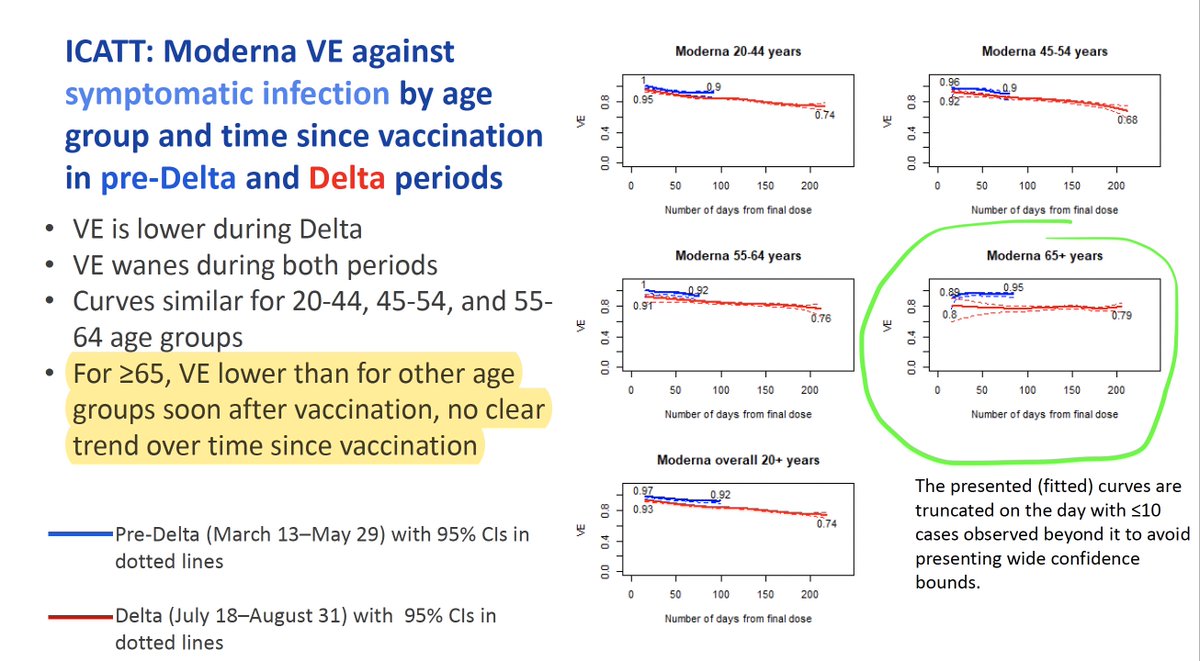
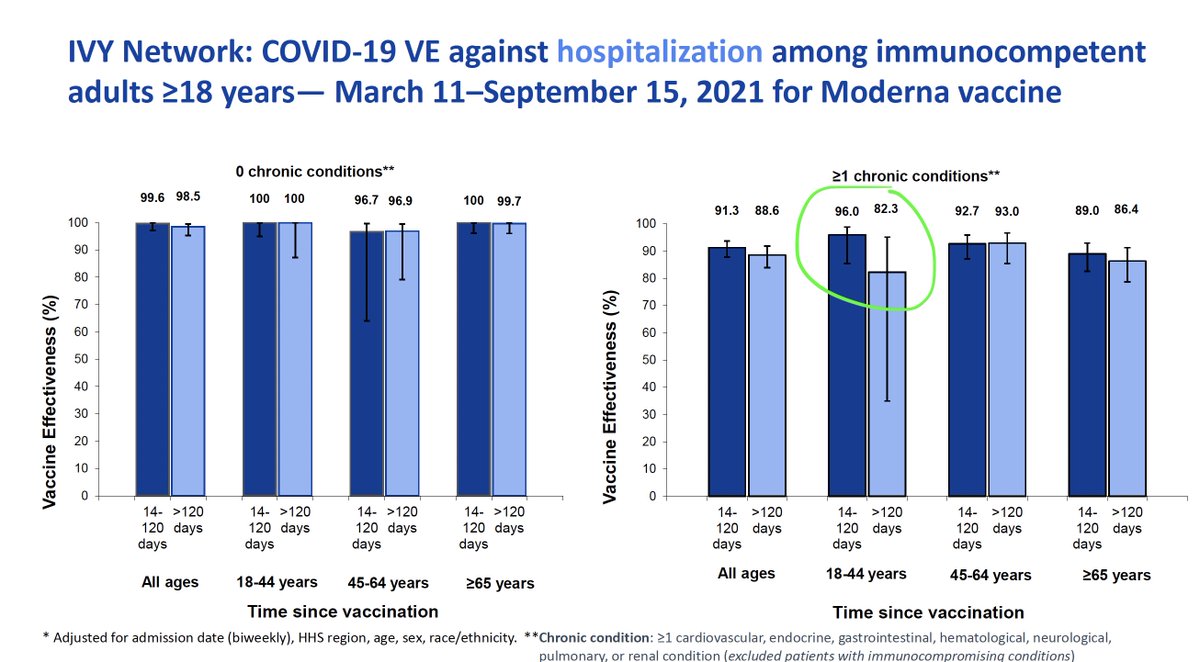
1/ Important new research by @KizzyPhD & other @NIAIDNews scientists on the Moderna vaccine in non-human primates.
They gave the primates 2 doses of the Moderna vaccine 4 weeks apart.
And challenged them with Delta variant virus 1 year later.
biorxiv.org/content/10.110…
They gave the primates 2 doses of the Moderna vaccine 4 weeks apart.
And challenged them with Delta variant virus 1 year later.
biorxiv.org/content/10.110…
2/
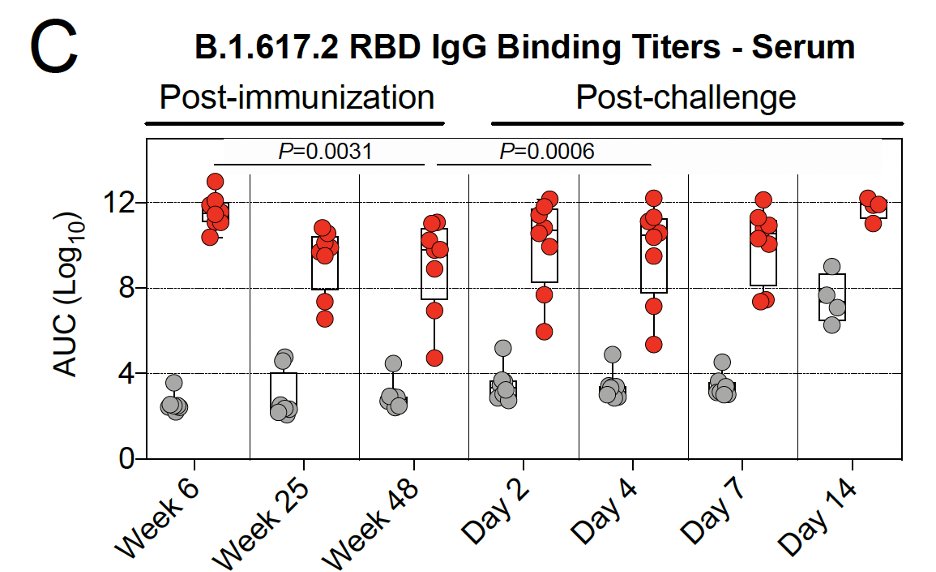
Gray: unvaccinated
Red: vaccinated
After vaccination, binding antibody levels:
- peaked at 6 weeks
- dropped weeks 6-25
- rate of decline slowed weeks 25-48
Gray: unvaccinated
Red: vaccinated
After vaccination, binding antibody levels:
- peaked at 6 weeks
- dropped weeks 6-25
- rate of decline slowed weeks 25-48
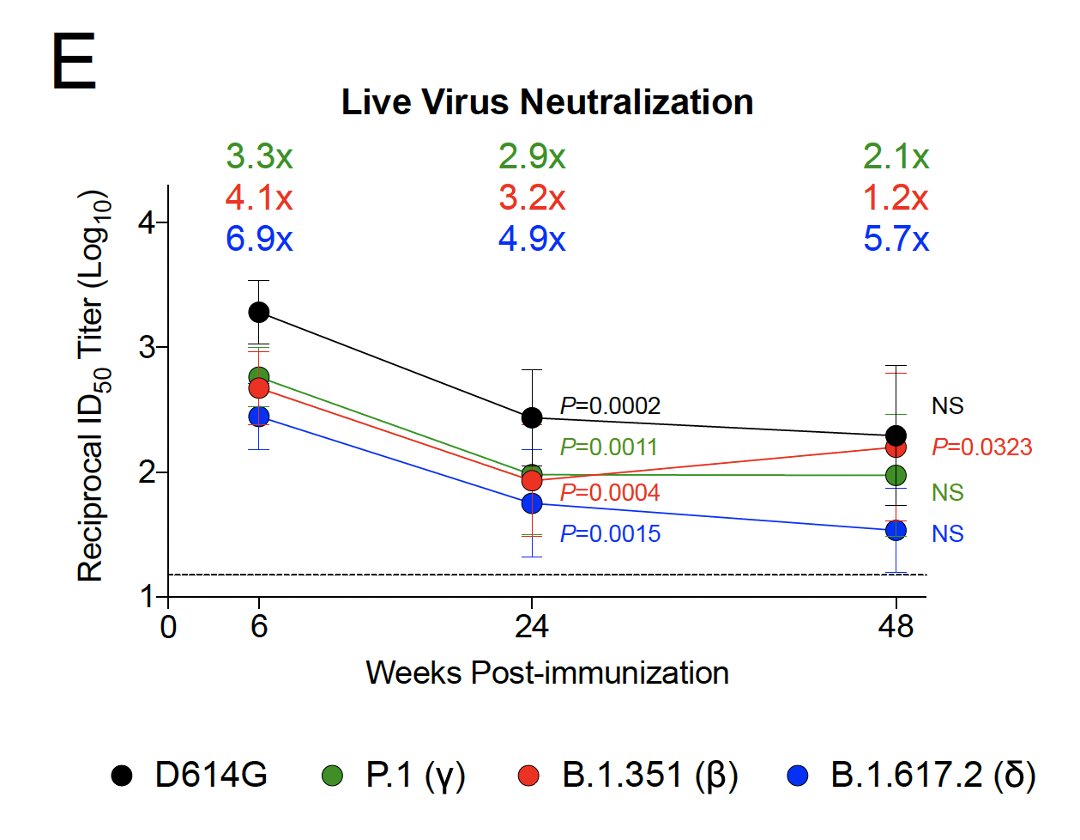
3/
Serum neutralizing antibody titers:
- dropped weeks 6-24
- rate of decline slowed weeks 24-48
- were highest vs D614G (early variant)⚫️>Gamma🟢>Beta🔴>Delta🔵
Serum neutralizing antibody titers:
- dropped weeks 6-24
- rate of decline slowed weeks 24-48
- were highest vs D614G (early variant)⚫️>Gamma🟢>Beta🔴>Delta🔵
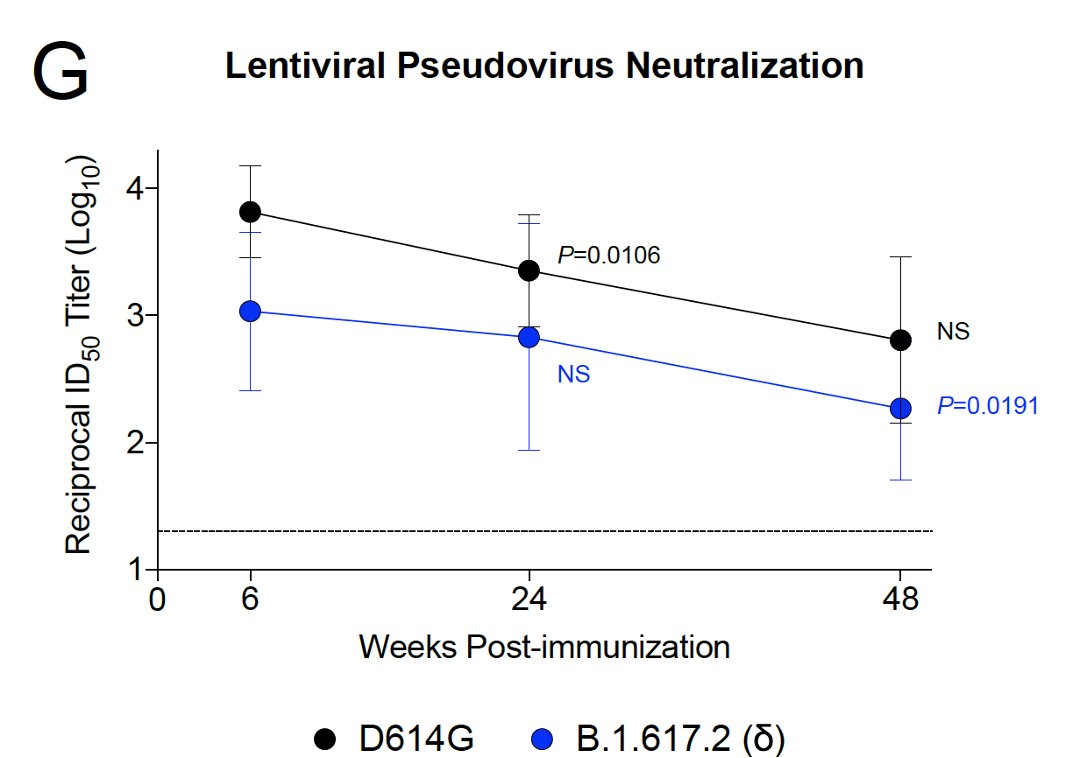
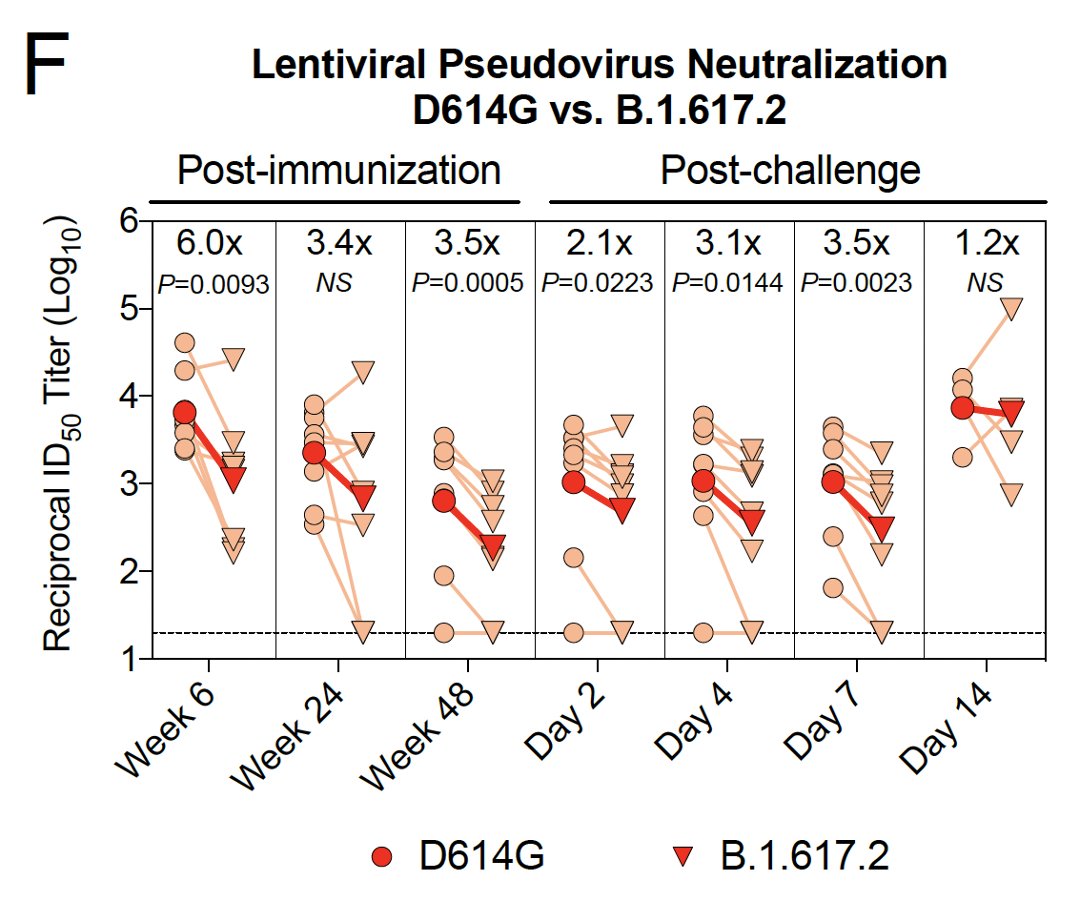
4/ This same pattern was observed with a pseudovirus assay:
- decline in neutralizing antibody titers over time
- lower neutralizing antibody titers against the Delta variant vs the early D614G variant
- decline in neutralizing antibody titers over time
- lower neutralizing antibody titers against the Delta variant vs the early D614G variant
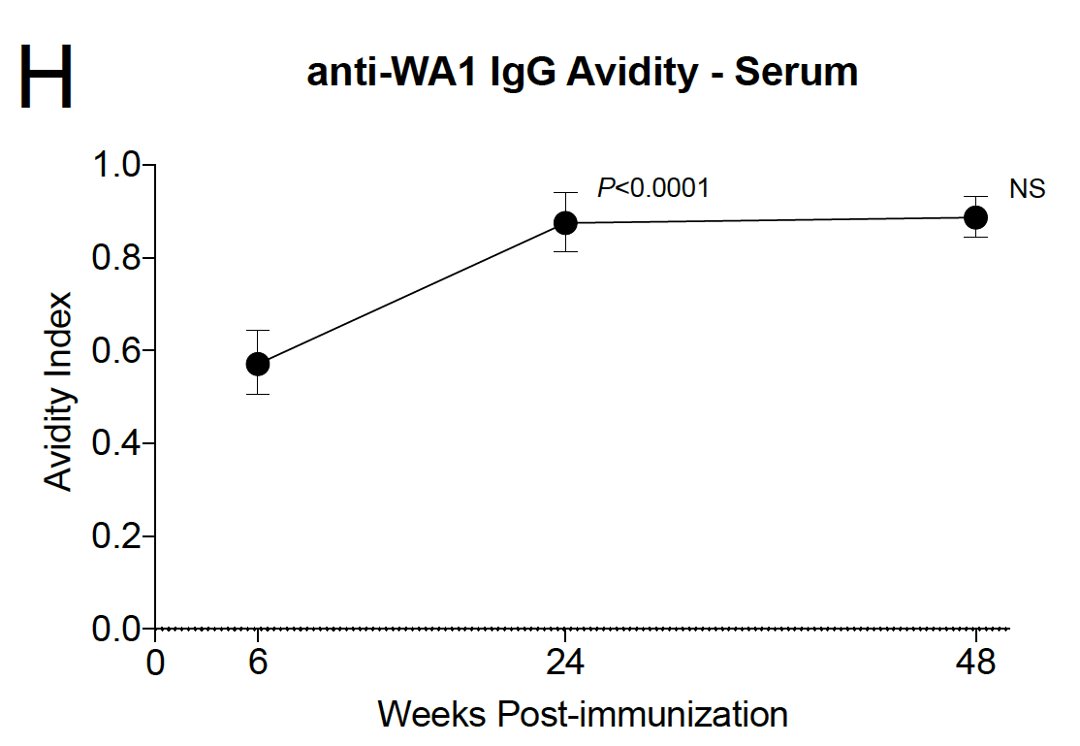
6/ Over time, antibody avidity (i.e. the strength of binding to Spike protein) got better 6-24 weeks after vaccination.
This likely reflects somatic hypermutation & affinity maturation.

This likely reflects somatic hypermutation & affinity maturation.
https://twitter.com/celinegounder/status/1417550246142529536?s=20
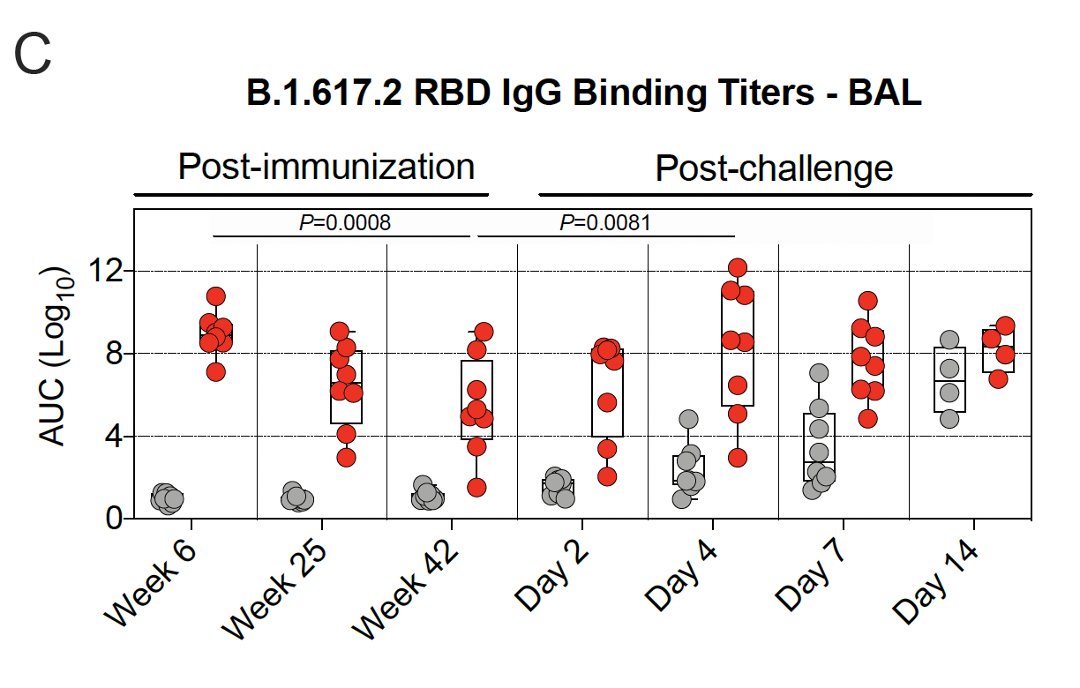
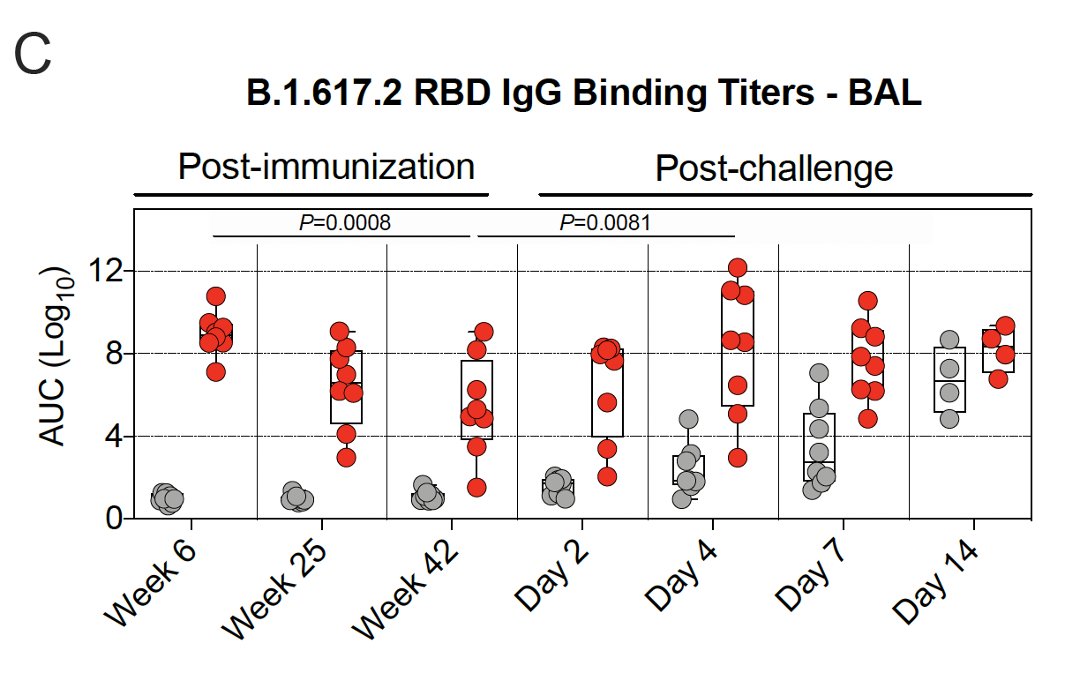
7/ They also looked at antibody titers in the lungs (bronchoalveolar lavage, BAL) & nose (nasal washes).
Binding antibody titers in the lungs dropped weeks 6-25, & then the rate of decline slowed.
Binding antibody titers in the lungs dropped weeks 6-25, & then the rate of decline slowed.
8/ Among vaccinated primates challenged with Delta virus, antibody titers in the lungs⬆️ & peaked by day 4.
Among unvaccinated primates challenged with Delta virus, antibody titers in the lungs⬆️ days 4-14.
Among unvaccinated primates challenged with Delta virus, antibody titers in the lungs⬆️ days 4-14.
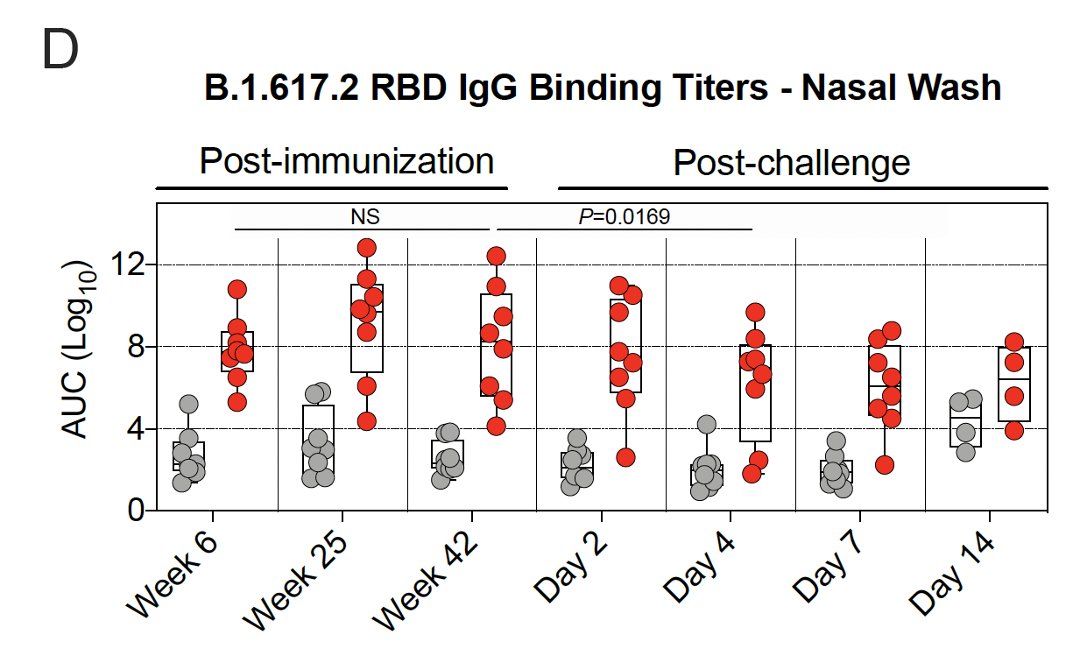
9/ Binding antibody titers in the👃🏾peaked later than in the🫁at 25 weeks & then remained stable out to week 42. 
10/
Among vaccinated primates challenged with Delta virus, antibody titers in the👃🏾⬇️by day 4.
Among unvaccinated primates challenged with Delta virus, antibody titers in the👃🏾⬆️ by day 14.
Antibodies in the👃🏾may be cleared differently than in the 🫁.
Among vaccinated primates challenged with Delta virus, antibody titers in the👃🏾⬇️by day 4.
Among unvaccinated primates challenged with Delta virus, antibody titers in the👃🏾⬆️ by day 14.
Antibodies in the👃🏾may be cleared differently than in the 🫁.
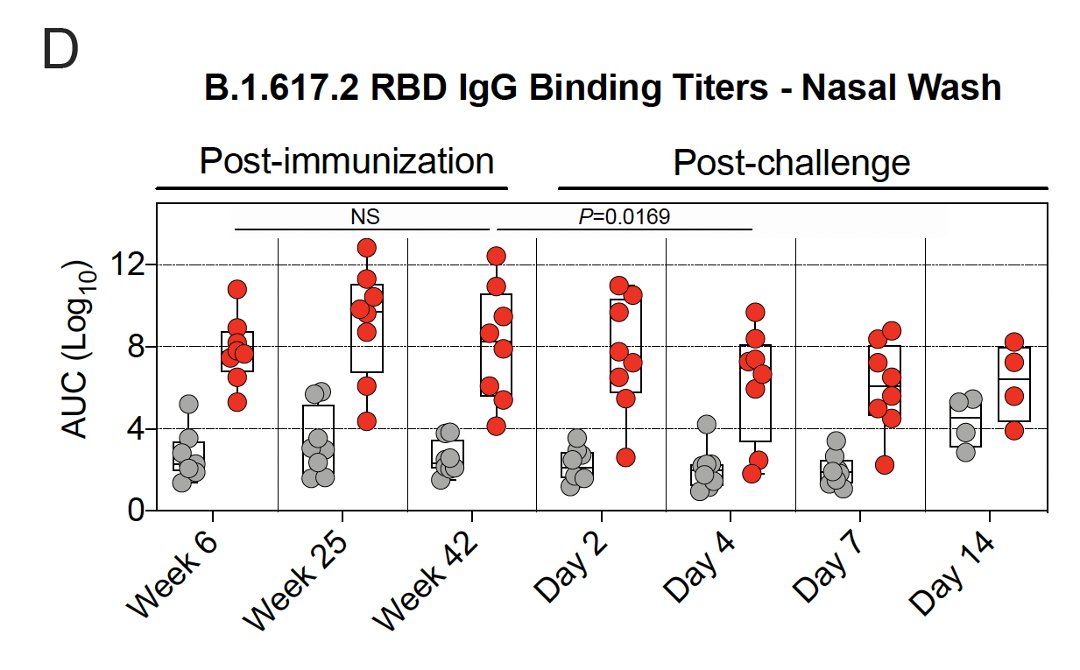
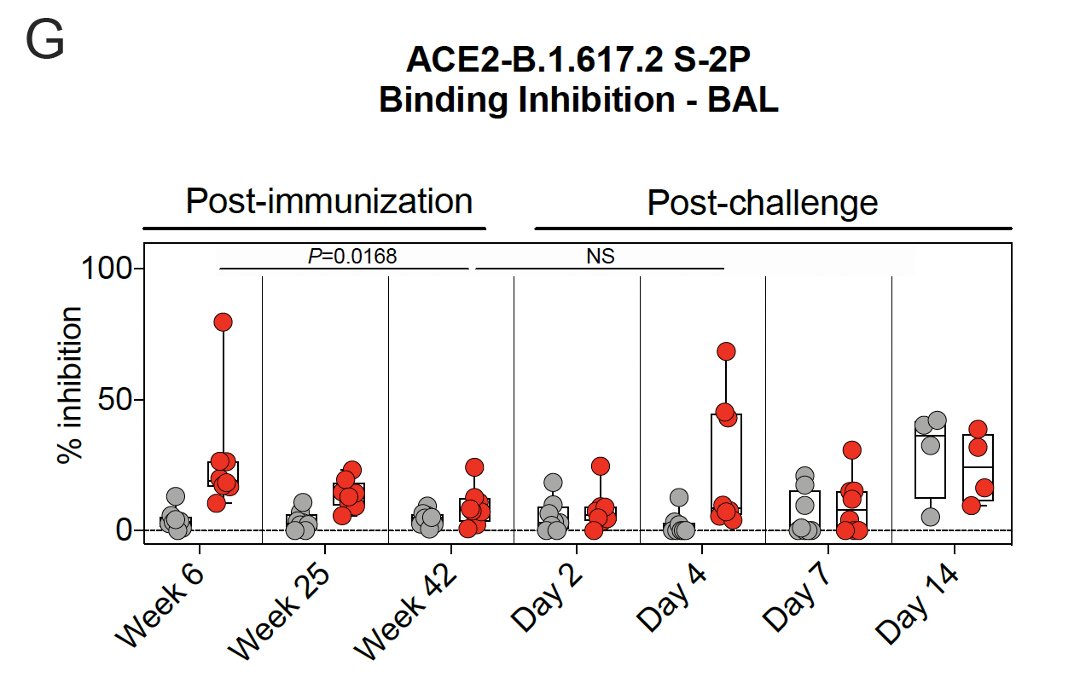
11/ They also used a Spike protein-ACE2 binding inhibition assay to assess antibody function.
Binding in🫁 peaked at 6 weeks after vaccination & declined by 42 weeks.
Binding in🫁 peaked at 6 weeks after vaccination & declined by 42 weeks.
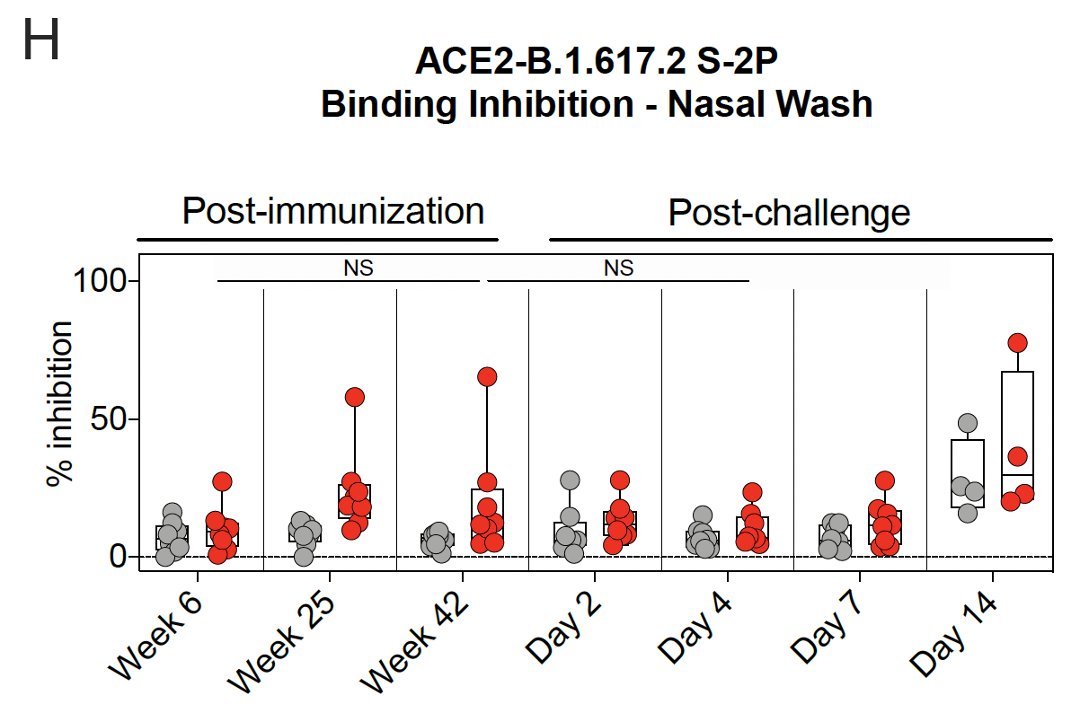
12/ The Spike protein-ACE2 binding inhibition assay found that binding in👃🏾increased until week 25 & remained elevated at week 42 after vaccination. 
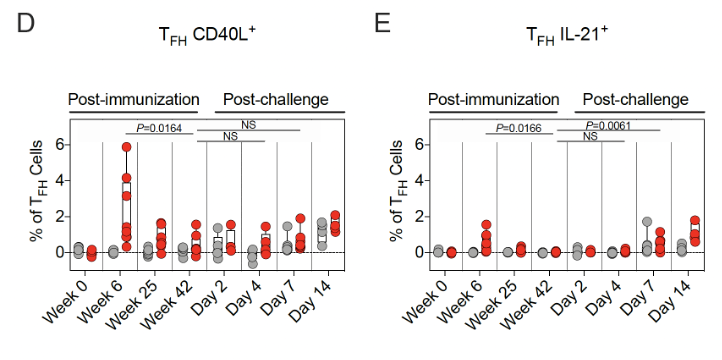
13/ They assessed S protein-specific memory B-cells after vaccination with flow cytometry.
The % of S-protein specific memory B-cell responses peaked at 6 weeks after vaccination & then decreased by week 25.
The % of S-protein specific memory B-cell responses peaked at 6 weeks after vaccination & then decreased by week 25.
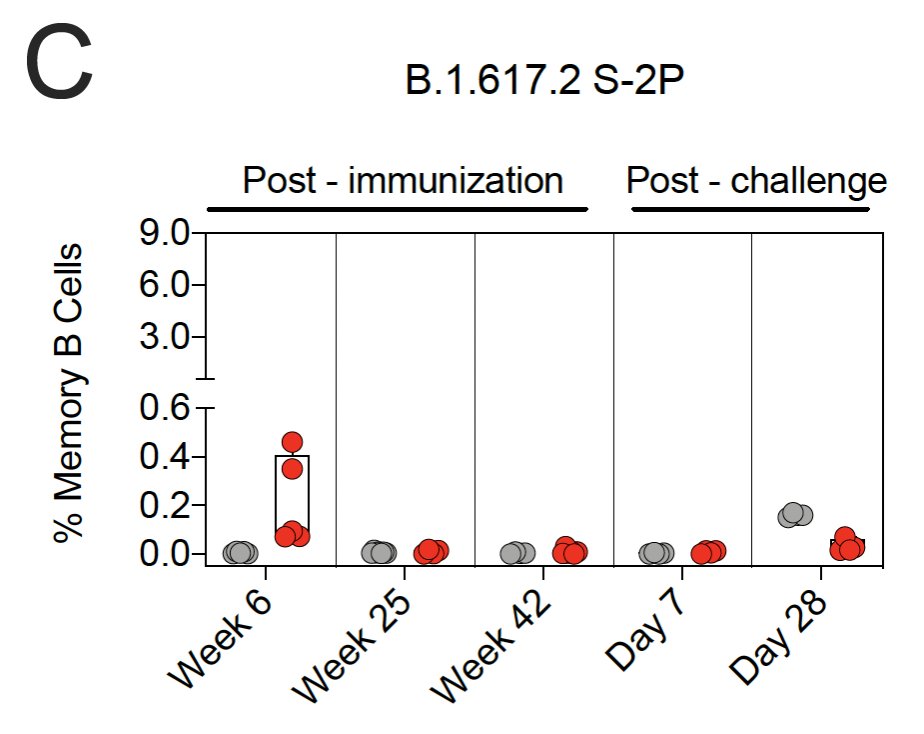
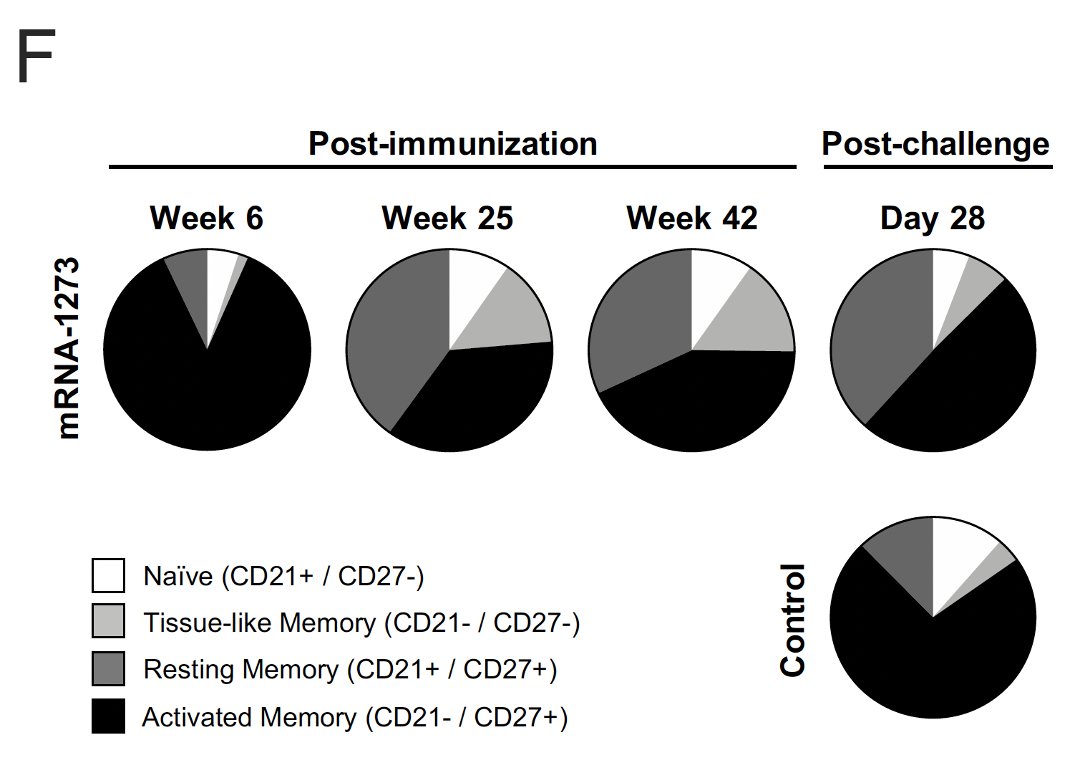
14/ Over the course of a year, Spike protein-binding B-cells shifted from activated memory phenotype to activated/resting/tissue-like memory cells. 
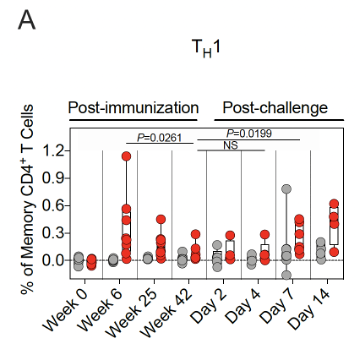
15/ Spike protein-specific T-cell responses were also assessed.
Th1 responses were detected at week 6 & ⬇️by week 25. They were low-undetectable by week 42.
Th1 responses were detected at week 6 & ⬇️by week 25. They were low-undetectable by week 42.
18/ After Delta virus challenge, there was significantly less viral replication in 🫁 by day 4 among vaccinated vs unvaccinated primates. 
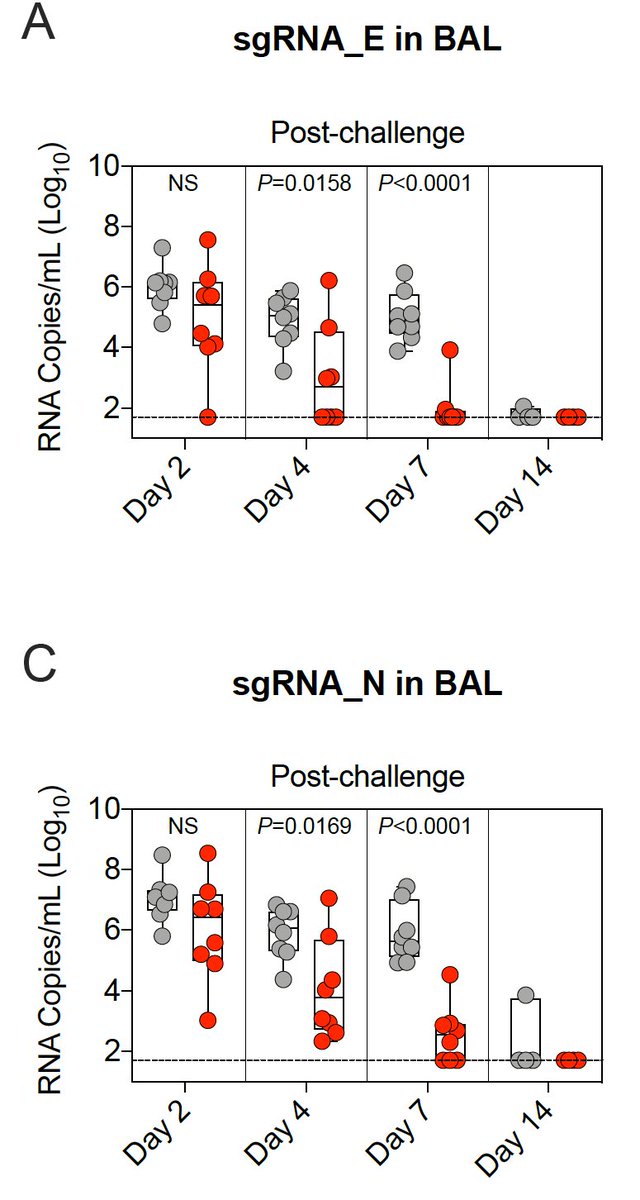
19/ After Delta virus challenge, there was significantly less viral replication in👃🏾by day 2 among vaccinated vs unvaccinated primates, but the overall reduction in viral replication was less in👃🏾than in 🫁. 
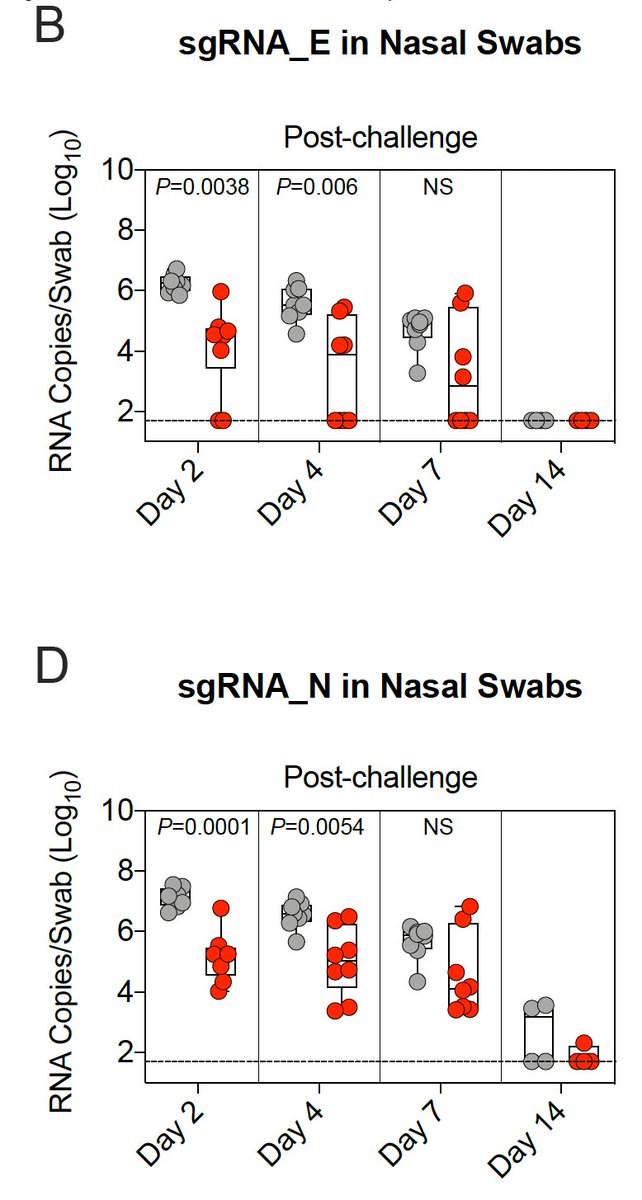
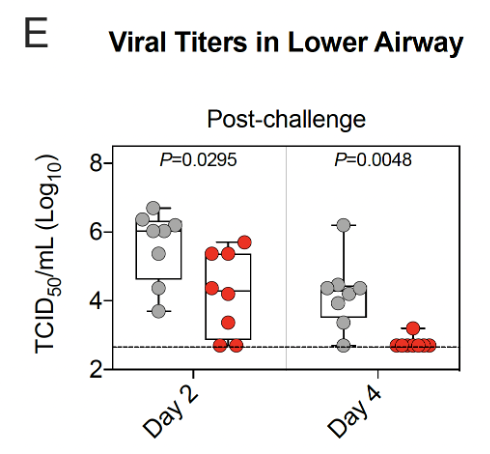
20/ Tissue culture infectious dose was also assessed from 🫁&👃🏾samples.
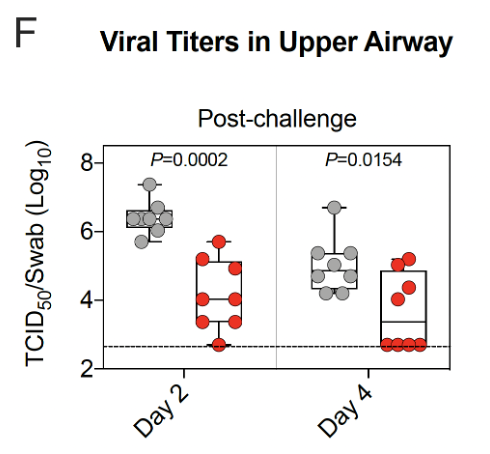
Viral replication in 🫁&👃🏾 of vaccinated primates was lower than for unvaccinated primates.
By day 4 after Delta virus challenge, replication in🫁was undetectable in almost all vaccinated primates.
Viral replication in 🫁&👃🏾 of vaccinated primates was lower than for unvaccinated primates.
By day 4 after Delta virus challenge, replication in🫁was undetectable in almost all vaccinated primates.
21/ Viral replication in the 👃🏾of vaccinated primates was lower than for unvaccinated primates, but not zero. 
22/ IN SUMMARY:
Unvaccinated and Moderna vaccinated primates were challenged with the Delta variant ~1 year after vaccination.
Four days after challenge, vaccinated primates had NO culturable virus in the lungs, but virus was culturable in the nose.
Unvaccinated and Moderna vaccinated primates were challenged with the Delta variant ~1 year after vaccination.
Four days after challenge, vaccinated primates had NO culturable virus in the lungs, but virus was culturable in the nose.
23/ Antibody-related protection in the lungs is durable but delayed.
It takes time for memory B-cells to rev up & make antibodies.
It takes time for memory B-cells to rev up & make antibodies.
24/ Note that enough virus made it into the 🫁 of vaccinated primates to elicit a local anamnestic antibody response and a bump in Spike protein-specific B- & T-cell populations.
25/🫁from vaccinated & unvaccinated primates were examined 7 days after Delta virus challenge.
Viral antigen was detected in🫁of all unvaccinated primates & none of the vaccinated primates.
Inflammation was:
- minimal-moderate among vaccinated
- minimal-severe in unvaccinated
Viral antigen was detected in🫁of all unvaccinated primates & none of the vaccinated primates.
Inflammation was:
- minimal-moderate among vaccinated
- minimal-severe in unvaccinated
26/ Binding & neutralizing antibody titers declined over one year after vaccination leaving the lungs susceptible to viral infection & replication in the first couple days after challenge.
But the local anamnestic response was sufficient to prevent severe disease.
But the local anamnestic response was sufficient to prevent severe disease.
27/ Neutralizing antibodies are a good correlate of protection soon after vaccination.
A year after vaccination, memory B-cells may be a better correlate of protection.
A year after vaccination, memory B-cells may be a better correlate of protection.
28/ If the goal is to prevent infection in the👃🏾&🫁, it’s likely primates (& people) will need repeated (yearly?) vaccine boosters.
If the goal is to prevent severe disease, protection from the mRNA vaccines appears durable (at least out to 48 weeks).
If the goal is to prevent severe disease, protection from the mRNA vaccines appears durable (at least out to 48 weeks).
• • •
Missing some Tweet in this thread? You can try to
force a refresh