
I'll document this case of aggressive dishonesty from @AviBittMD as a reference for why I will no longer be engaging him in the future, as he's heavy on the insults and light on the substance.
So, he claims that my claim was false because remdesivir succeeded in a similar trial.
So, he claims that my claim was false because remdesivir succeeded in a similar trial.
https://twitter.com/AviBittMD/status/1495259818227126275
He contests this claim I made about the JAMA study:
"In brief, the study design was such that any other antiviral, such as Paxlovid or Molnupiravir, would also have failed."
"In brief, the study design was such that any other antiviral, such as Paxlovid or Molnupiravir, would also have failed."
And further down, I summarized the issue:
https://twitter.com/alexandrosM/status/1495066510942879744?t=nIaTZNQmvCNe8VXcFYA_AQ&s=19
To disprove my claim, he brings up a study that had even later start of treatment, and indeed succeeded at its primary endpoint. But that endpoint was very different (time to recovery) so not really the same study design.
@Grinman helpfully noted this, and got abuse back.
@Grinman helpfully noted this, and got abuse back.

In his abusive response, he notes another study he referenced. Patients with 5 days from symptom onset, and title mentions "progression to severe disease".
It looks like the identical study design.
To someone who isn't paying attention. nejm.org/doi/full/10.10…
It looks like the identical study design.
To someone who isn't paying attention. nejm.org/doi/full/10.10…
When we try to see what they mean by "progression to severe disease", we see their endpoint:
"The primary efficacy end point was a composite of Covid-19–related hospitalization or death from any cause by day 28"
"The primary efficacy end point was a composite of Covid-19–related hospitalization or death from any cause by day 28"

The I-TECH study had a super-early endpoint, that in the ivm group triggered in 3.2. days after start of treatment. So, the average patient didn't even have time to complete the treatment. 

In fact, the I-TECH definition of severe disease classifies only as "mild disease" by the standards of the WHO.

https://twitter.com/compsciman/status/1494905161709498368?t=mG2pXWtv1qbp0kn19FVi_Q&s=19
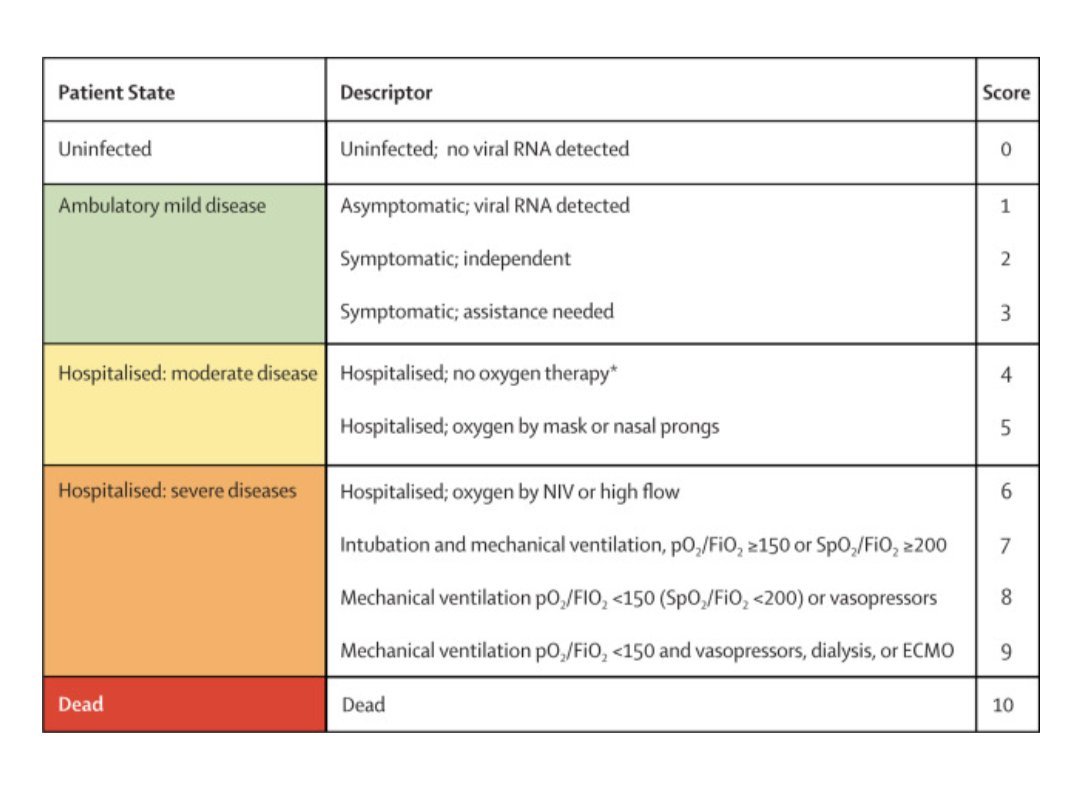
So what we have here is wordplay.
The 2nd remdesivir study used nonhospitalized patients, and the endpoint was, roughly "hospitalization or death"
The ivm study used mostly (I think) hospitalized patients, endpoint was "does a Dr think you need oxygen to not drop below 95%"
The 2nd remdesivir study used nonhospitalized patients, and the endpoint was, roughly "hospitalization or death"
The ivm study used mostly (I think) hospitalized patients, endpoint was "does a Dr think you need oxygen to not drop below 95%"
The definitions of "severe disease" are very very different in these two studies. Also, the nonhosp. patients in the remdesivir trial were obviously in better condition, as none of the control group died, whereas in the similar sized I-TECH study, 10 died in the control group.
My entire point is that patients that already have significant momentum, combined with an endpoint that is super close to their initial condition, will not give an antiviral enough time to act. Again, my figure: 
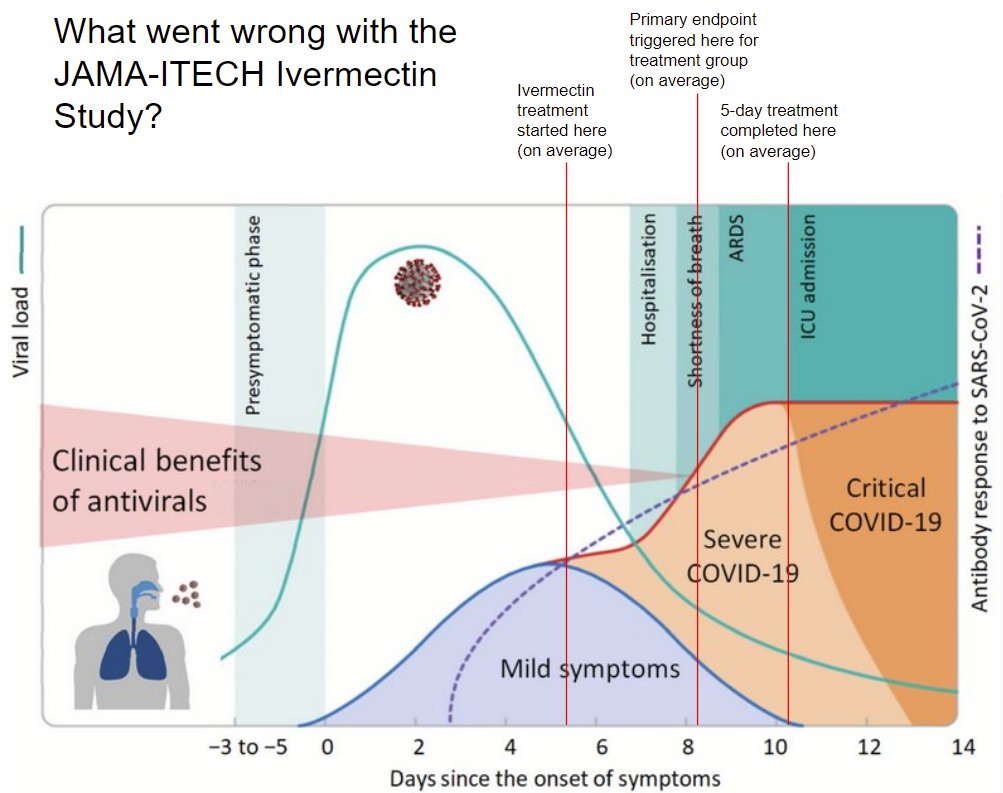
His response, other than vile insults, is a study with patients that are clearly in better shape, and an endpoint that is harder to trigger (and indeed, only 15 triggered it, even in the control group, vs 43 control group pts that triggered the endpoint in the ivm trial)
Now, everyone makes mistakes. That's not a problem.
The problem is his disgusting attitude, dripping with condescension and ridicule. Victory lapping and insults without even a hint of doubt or self reflection.
The problem is his disgusting attitude, dripping with condescension and ridicule. Victory lapping and insults without even a hint of doubt or self reflection.
https://twitter.com/AviBittMD/status/1495266226775412737?t=k99Yppn9qwQGTwpwSpi4Kw&s=19
Attempting to discuss with this person constantly puts me in situations where I have to deal with sophisticated sophistry, and the payoff for showing him to be in error is nothing of value to me.
I have tolerated him thus far as I do believe pushback is valuable.
I have tolerated him thus far as I do believe pushback is valuable.
This stuff isn't honest pushback though. It's grandstanding in the hopes I won't have time to dig in, or at best, without actually understanding my claim or the studies he cites.
This isn't useful, and I don't owe him my time.
This isn't useful, and I don't owe him my time.
Aaand responds with a classic goalpost shift.
I couldn't conclude our interaction with a better demonstration of this man's inability to admit an error.
I couldn't conclude our interaction with a better demonstration of this man's inability to admit an error.
https://twitter.com/AviBittMD/status/1496635958866939904?t=k99Yppn9qwQGTwpwSpi4Kw&s=19
• • •
Missing some Tweet in this thread? You can try to
force a refresh







