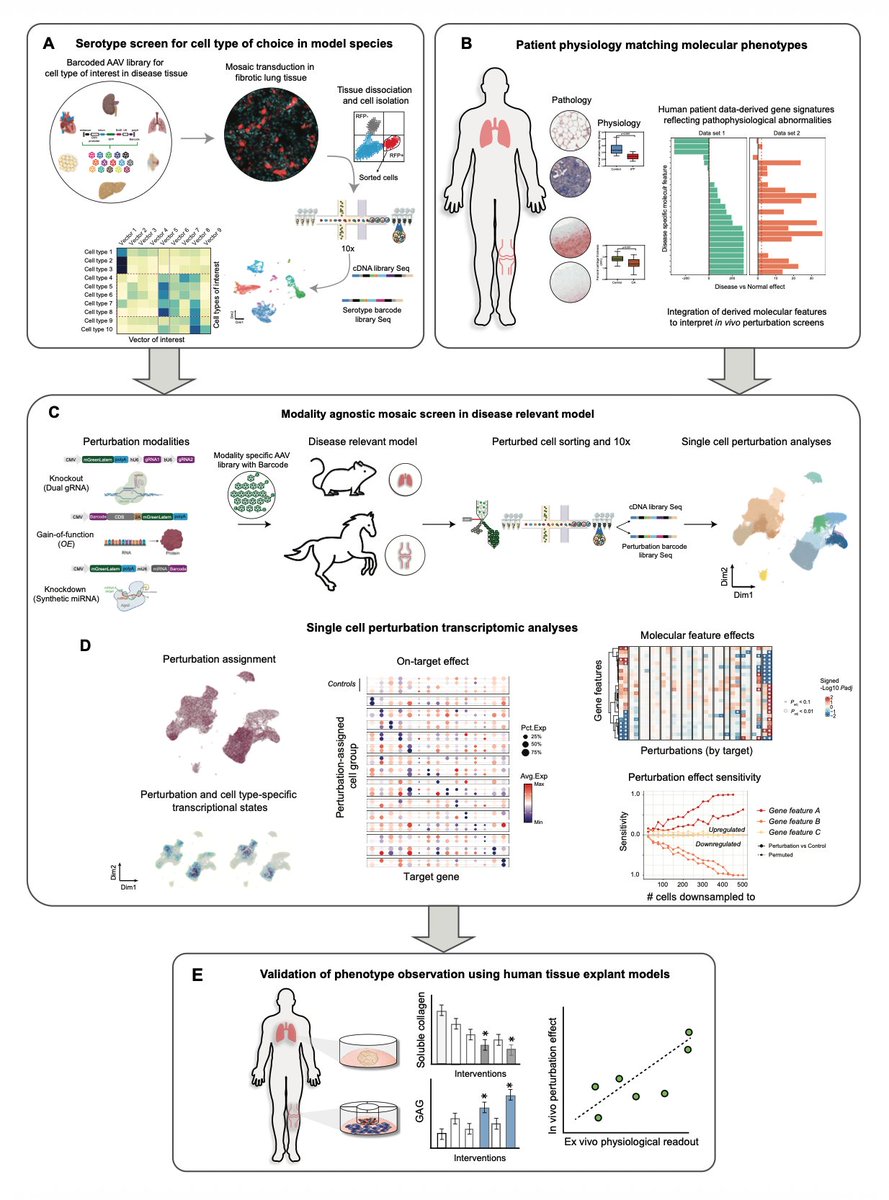
In the midst of #AcademicPatentGate, @Nicole_Paulk and I started talking more broadly about the ideal relationship between #Academia and for-profit in the field of #biotechnology. Should ties be stronger or weaker? What happens when #OpenAccess meets the global economy?
One side: For-profit companies license academic innovations that used public research #funding, then charge the public for the products.
Other side: Public research funding has the goal of fueling innovation. Restricting discoveries to the academic community is not beneficial.
Other side: Public research funding has the goal of fueling innovation. Restricting discoveries to the academic community is not beneficial.
But #Academia is also seen as an objective source of truth (increasingly important these days). At its best, scientists engage in a disinterested search for truth, and ideas are made to be tested. This has been very good for civilization so far.
Professors now often have financial interests based on research in their labs, particularly in fields which are very translational (like #GeneTherapy). The degree to which this compromises neutrality depends on their character, but objectivity can be doubted.
There are advantages to making #BasicResearch seem translational. Industry may supplement your lab’s funding. Grant review panels view translation research positively, even if it means repeating model organism work in a “questionable but best available” mouse model.
With these incentives in place, disinterested truth-seeking and curiosity-driven research feel beleaguered. Science has hardly been corrupted, but researchers are leaving poorly paid academic jobs, and finding unbiased observers is hard.
There is nothing wrong with researchers commercializing their findings, and we should still support this. But the current trend towards a continuous spectrum of commercial interests has the potential to deplete the wellspring of commercialization.
A proposed solution: funders, including the NIH, could delineate ‘Basic’ and ‘PreTranslational’ research, and allocate funding separately. #BasicResearch should receive much more funding (2/3), and pay higher salaries (by 30-50%).
The purpose of this skew is to counterbalance the natural #PreTranslational incentives of royalties, equity, sponsorship, consulting fees … and cementing #BasicResearch as intrinsically valuable reduces the motivation to feign translatability.
Drawing such a line is non-trivial. But since the goal is balanced incentives, we can just look at whether there’s a healthy equilibrium between curiosity-driven and pre-translational research efforts. If scientists flock to one side of the equation, incentives are imbalanced.
(Note that neither of us would benefit from this funding structure)
• • •
Missing some Tweet in this thread? You can try to
force a refresh