
1/n
Stop the abuse: plea for more principled approach to analysis of AEs.
A (long) thread. Please RT.
@tim_friede @ADAlthousePhD @VR_Cornelius @RachPips @statsepi
How are AEs typically reported in an RCT? Along the lines of the table below.
From: nejm.org/doi/full/10.10…
Stop the abuse: plea for more principled approach to analysis of AEs.
A (long) thread. Please RT.
@tim_friede @ADAlthousePhD @VR_Cornelius @RachPips @statsepi
How are AEs typically reported in an RCT? Along the lines of the table below.
From: nejm.org/doi/full/10.10…

2/n
So what is the purpose of such tables? I see two:
1) "Signal detection" --> find AEs that differ between arms.
2) Estimate true probability of an AE of interest, P(AE).
I will focus on 2) below.
So what is the purpose of such tables? I see two:
1) "Signal detection" --> find AEs that differ between arms.
2) Estimate true probability of an AE of interest, P(AE).
I will focus on 2) below.
3/n Assume we are interested in P(AE) - this is our estimand.
What do proportions in above table estimate? The incidence proportion:
IP = P(AE happening in [0, t] and that this AE is observed).
What do proportions in above table estimate? The incidence proportion:
IP = P(AE happening in [0, t] and that this AE is observed).
4/n
Assumptions:
- Interpretable only if all patients have ~ same follow-up.
- If we have (administratively) censored patients then IP *underestimates* true AE probability.
Challenges for estimation of P(AE):
1) Unequal follow-up in both arms.
2) What is AE hazard?
Assumptions:
- Interpretable only if all patients have ~ same follow-up.
- If we have (administratively) censored patients then IP *underestimates* true AE probability.
Challenges for estimation of P(AE):
1) Unequal follow-up in both arms.
2) What is AE hazard?
5/n
3) Competing events (CE) such as death before AE or treatment discontinuation leading to end of AE recording.
Simply censoring CEs gives biased estimates of AE probabilities P(AE).
Incidence proportion accounts for 2) and 3).
What are other estimators?
3) Competing events (CE) such as death before AE or treatment discontinuation leading to end of AE recording.
Simply censoring CEs gives biased estimates of AE probabilities P(AE).
Incidence proportion accounts for 2) and 3).
What are other estimators?
6/n
See published SAP of SAVVY project (Survival analysis for AdVerse events with VarYing follow-up times) for list of alternative estimators:
doi.org/10.1002/bimj.2…
Their behaviour w.r.t. to above 3 criteria are indicated in Table 1 in this paper: arxiv.org/abs/2008.07883
See published SAP of SAVVY project (Survival analysis for AdVerse events with VarYing follow-up times) for list of alternative estimators:
doi.org/10.1002/bimj.2…
Their behaviour w.r.t. to above 3 criteria are indicated in Table 1 in this paper: arxiv.org/abs/2008.07883
7/n
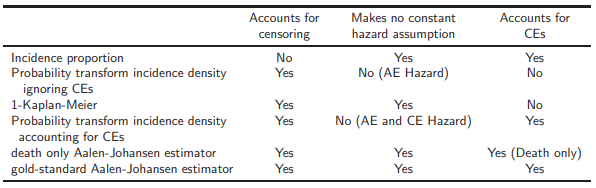
In absence of CEs (1 - Kaplan-Meier) is unbiased.
However, in presence of CEs (1 – KM) *overestimates* true P(AE).
Aalen-Johansen is only estimator of P(AE) that accounts for censoring, CEs, and makes no constant hazard assumption --> can be considered gold standard.
In absence of CEs (1 - Kaplan-Meier) is unbiased.
However, in presence of CEs (1 – KM) *overestimates* true P(AE).
Aalen-Johansen is only estimator of P(AE) that accounts for censoring, CEs, and makes no constant hazard assumption --> can be considered gold standard.
8/n
But how large can bias of other estimators become, notably IP and (1 - KM) with simple censoring of CEs?
SAVVY: academia + industry collaboration exploring bias on data of 17 RCTs from various indications.
Results:
- Estimation of P(AE) in 1 arm: arxiv.org/abs/2008.07883
But how large can bias of other estimators become, notably IP and (1 - KM) with simple censoring of CEs?
SAVVY: academia + industry collaboration exploring bias on data of 17 RCTs from various indications.
Results:
- Estimation of P(AE) in 1 arm: arxiv.org/abs/2008.07883
9/n
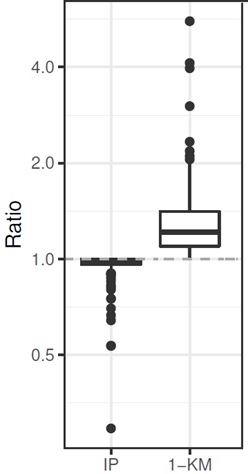
- Comparison of P(AE) in 2-arm RCT: arxiv.org/abs/2008.07881
--> IP underestimates P(AE) up to factor 3!
--> (1 - KM) overestimates P(AE) up to factor 5!
Papers have many more analyses, e.g. discrepancies of estimators when categorizing AE risk acc. to @iqwig guidelines.
- Comparison of P(AE) in 2-arm RCT: arxiv.org/abs/2008.07881
--> IP underestimates P(AE) up to factor 3!
--> (1 - KM) overestimates P(AE) up to factor 5!
Papers have many more analyses, e.g. discrepancies of estimators when categorizing AE risk acc. to @iqwig guidelines.
10/n
Empirical quantification illustrates how large bias can be.
Goal of SAVVY: raise awareness that when estimating P(AE):
- Start with scientific question: Signal detection or estimation of P(AE)?
- Censoring? Competing events?
- Appropriate estimator --> Aalen-Johansen.
Empirical quantification illustrates how large bias can be.
Goal of SAVVY: raise awareness that when estimating P(AE):
- Start with scientific question: Signal detection or estimation of P(AE)?
- Censoring? Competing events?
- Appropriate estimator --> Aalen-Johansen.
11/n
Aalen-Johansen is available in any standard software.
Code and references to all papers and preprints: numbersman77.github.io/AEprobs/SAVVY_…
Comments welcome!
The end.
Aalen-Johansen is available in any standard software.
Code and references to all papers and preprints: numbersman77.github.io/AEprobs/SAVVY_…
Comments welcome!
The end.
@threadreaderapp unroll
• • •
Missing some Tweet in this thread? You can try to
force a refresh


