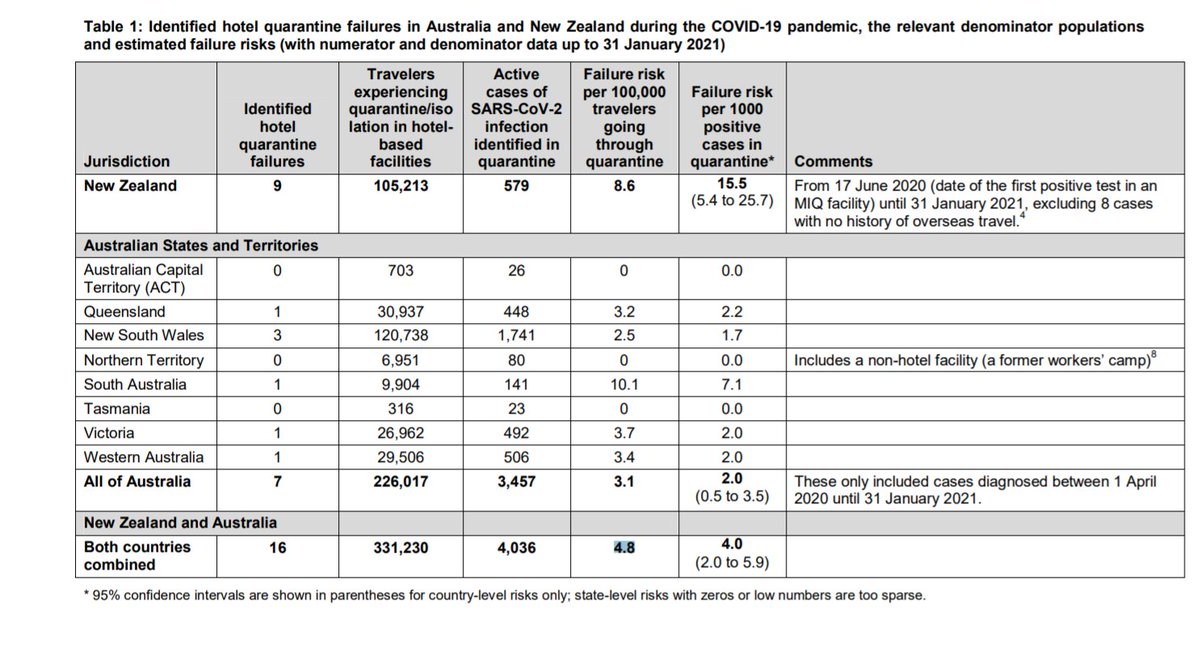
JCVI extension of vax dosing interval to 12 weeks is a great public health decision.
3.5X higher peak antibody levels w/ Pfizer if delayed boost vs 3 week.
Really important for protection against variants and duration esp in vulnerable🧵
1/
nature.com/articles/d4158…
3.5X higher peak antibody levels w/ Pfizer if delayed boost vs 3 week.
Really important for protection against variants and duration esp in vulnerable🧵
1/
nature.com/articles/d4158…
We kept seeing 1 dose vax after prior infxn boosting Ab tires 3X higher than 2 doses at the standard 3 week interval. Some of us wondered if this was from optimised prime-boost interval at the time...
It now looks like a probable explanation.
2/

It now looks like a probable explanation.
2/
https://twitter.com/DevanSinha/status/1375468203070132226
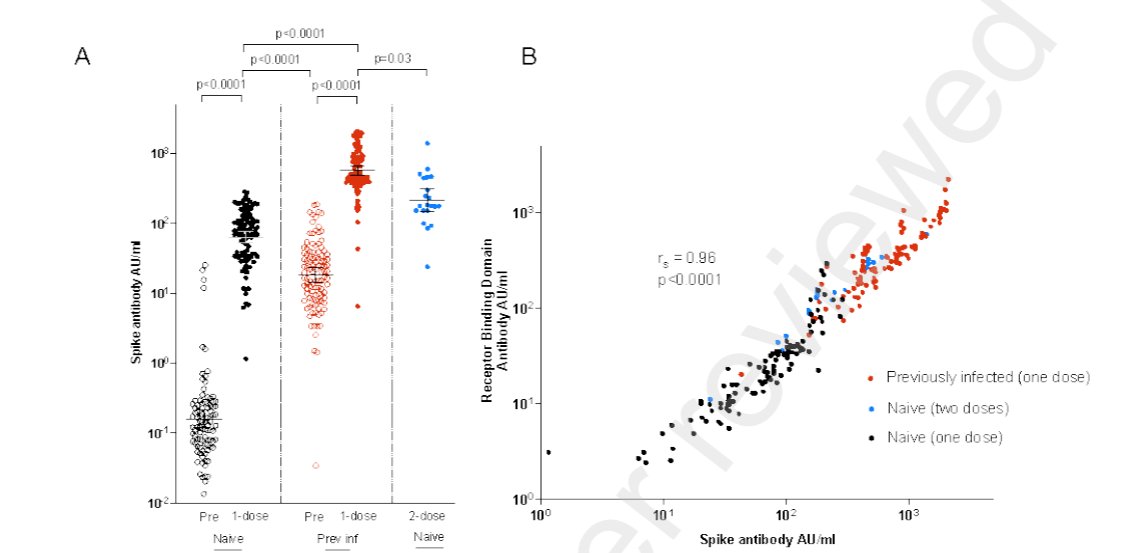
3 weeks on short side of intervals, where immune response hasn't completely matured after prime stimulus.
Delay may selectively expand population of long lived plasma cells (B cell derivatives) too.
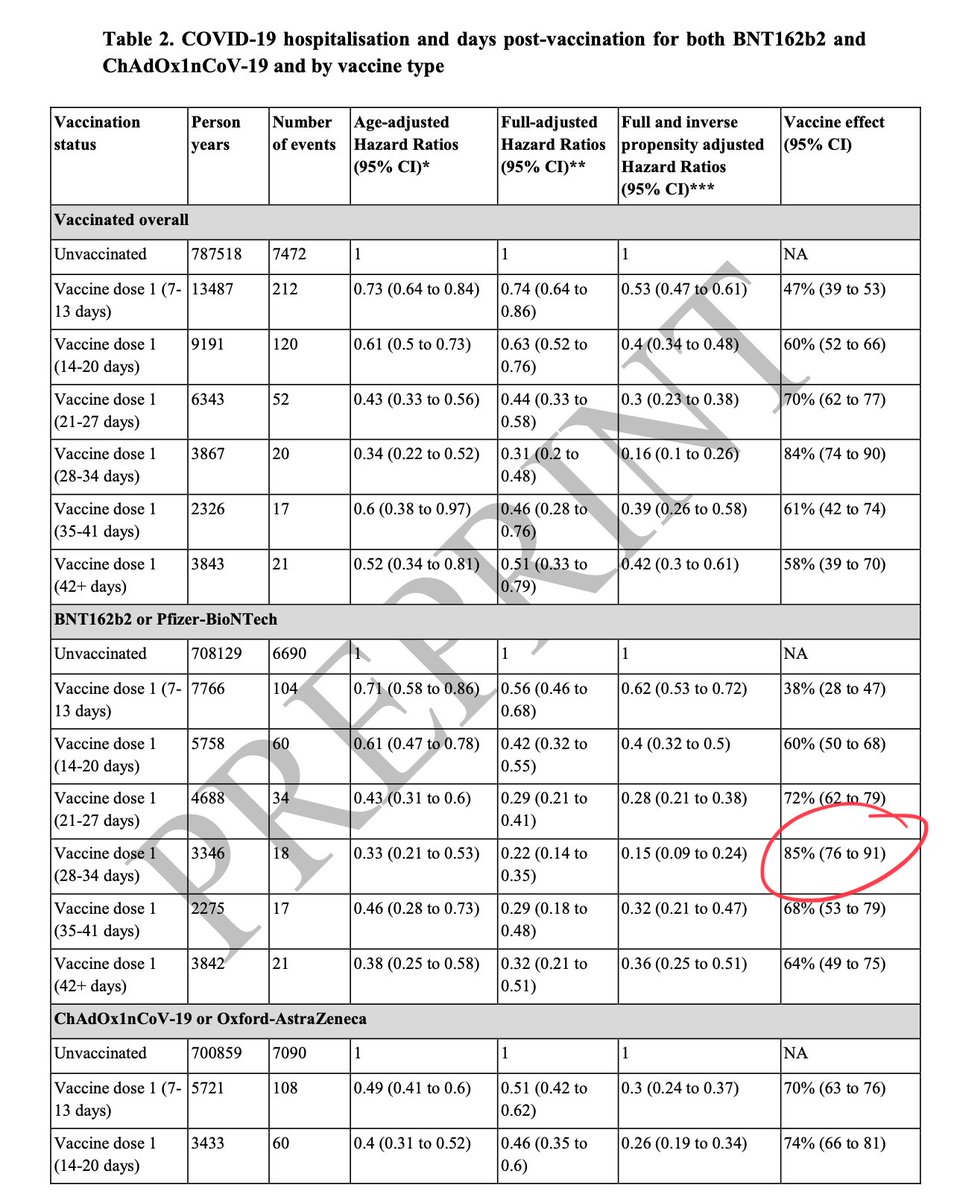
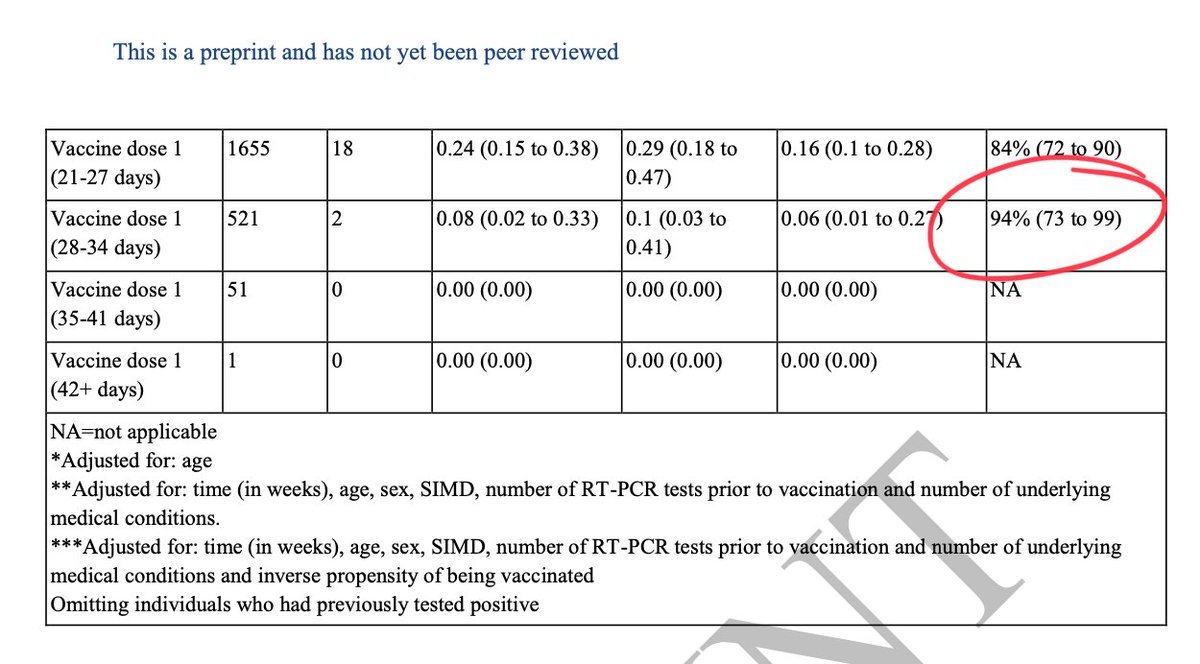
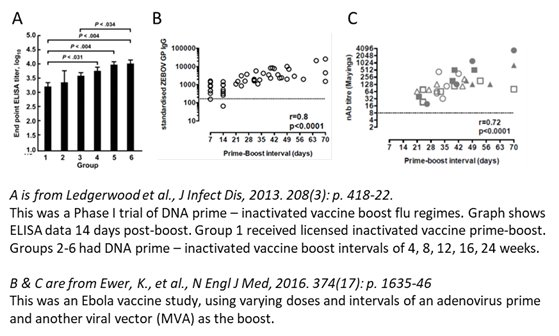
Delayed boost shown to have better Ab response across prev vax types👇
3/
Delay may selectively expand population of long lived plasma cells (B cell derivatives) too.
Delayed boost shown to have better Ab response across prev vax types👇
3/
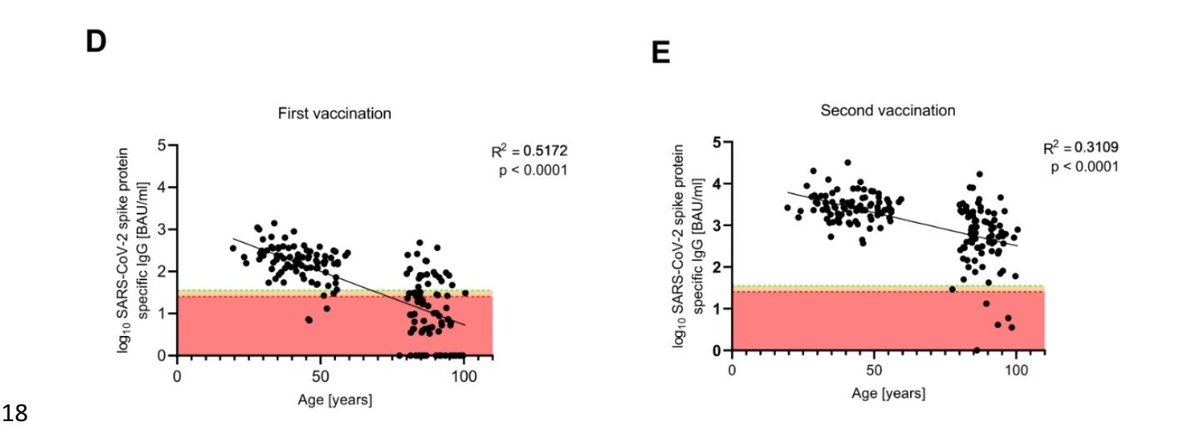
This is particularly important in the vulnerable elderly population where immune response is slower with later detectable seroconversion, and lower final Ab level too.
4/

4/

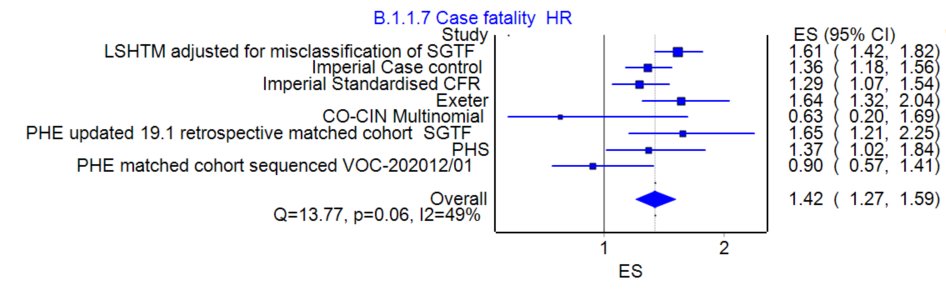
When it comes to variants the way we measure 'immune evasion' typically in labs (in vitro) is what concentration or dilution of blood serum containing Ab is needed to neutralise the virus to prevent growth on plates. (usually 50% of baseline viral plaque growth)
5/
5/
For Pfizer serum after 3 week dose interval compared to wild type SAR-COV-2 virus it takes approximately 3X higher serum concentration to achieve viral neutralisation for UK B1.1.7 and Brazilian P.1 and 8X higher serum concentration for South African B1.351.
6/
6/

This happens bc mutations in viral spike protein alter its amino acid residues and the protein shape our antibodies recognise and form hydrogen bonds with.
The mutations lower affinity (or binding association constant) between our generated antibodies and the spike protein.
7/
The mutations lower affinity (or binding association constant) between our generated antibodies and the spike protein.
7/

Spike mutations thus affect antibody binding equilibrium constant (Keq), the reaction of antigen (Ag) and antibody (Ab) binding to form antigen-antibody complex (Ab-Ag).
Shifting equilibrium to the left (unbound)
(Ka/Kd= Ka =Keq, where Kd is the dissociation constant)
8/
Shifting equilibrium to the left (unbound)
(Ka/Kd= Ka =Keq, where Kd is the dissociation constant)
8/

Since Ab bind less strongly to mutant spike antigen the virus is no longer trapped and free to infect cells and replicate.
But simply by increasing concentration of serum Ab we can mitigate this and shift antibody binding equilibrium back to where it was pre-mutation.
9/

But simply by increasing concentration of serum Ab we can mitigate this and shift antibody binding equilibrium back to where it was pre-mutation.
9/


Whilst not exactly linear nor necessarily at equilibrium in dynamic human system, serum w/ 3.5X higher Ab level should neutralise variants B1.1.7, P.1 and even Indian B1.617.2 similarly as serum from someone w/ 3 week Pfizer interval neutralising wild type virus.
10/
10/
In addition to providing likely better effectiveness against variants in the short term, delayed dosing will also increase durability of protection.
Ab levels decay w/ time and starting from a higher initial peak helps.
But decay rate itself is proportional to peak level!
11/
Ab levels decay w/ time and starting from a higher initial peak helps.
But decay rate itself is proportional to peak level!
11/
This is seen both in Ab longevity studies from natural SARS-COV-2 infection and also after vaccination of other viruses. Where higher initial Ab titres lower the rate of Ab decay.
So duration of protection is even longer than expected from using a static decay model.
12/

So duration of protection is even longer than expected from using a static decay model.
12/


In summary JCVI delayed 12 dose:
⬆️ Ab level, particularly valuable in vulnerable elderly
- will effectively neutralize variants as well as WT (except B1.351) if compared to 3 week interval
- longer protection at high level (area under Ab curve)
- maximised vax resources
13/
⬆️ Ab level, particularly valuable in vulnerable elderly
- will effectively neutralize variants as well as WT (except B1.351) if compared to 3 week interval
- longer protection at high level (area under Ab curve)
- maximised vax resources
13/
Puts UK in a strong position to deal with emerging variants in the coming months/years. More leeway for amount of 'immune escape' and time if boosters needed etc.
Canada following suit with 16 week interval now.
NB. Delayed boost increases Ab tires/VE for Oxford/AZ too👇
14/
Canada following suit with 16 week interval now.
NB. Delayed boost increases Ab tires/VE for Oxford/AZ too👇
14/

If accelerating dose interval <<12 weeks there should be real pressing need/risk of immediate infection...
2 doses definitely provides better protection against infxn, hospitalisation and death v 1 dose at any interval, but shorter interval will forgo longer term benefits.
End/
2 doses definitely provides better protection against infxn, hospitalisation and death v 1 dose at any interval, but shorter interval will forgo longer term benefits.
End/
• • •
Missing some Tweet in this thread? You can try to
force a refresh