New #preprint out on mapping effects of DBS for cervical vs. generalized dystonia to the connectome. A brief thread.
researchgate.net/publication/35…
researchgate.net/publication/35…
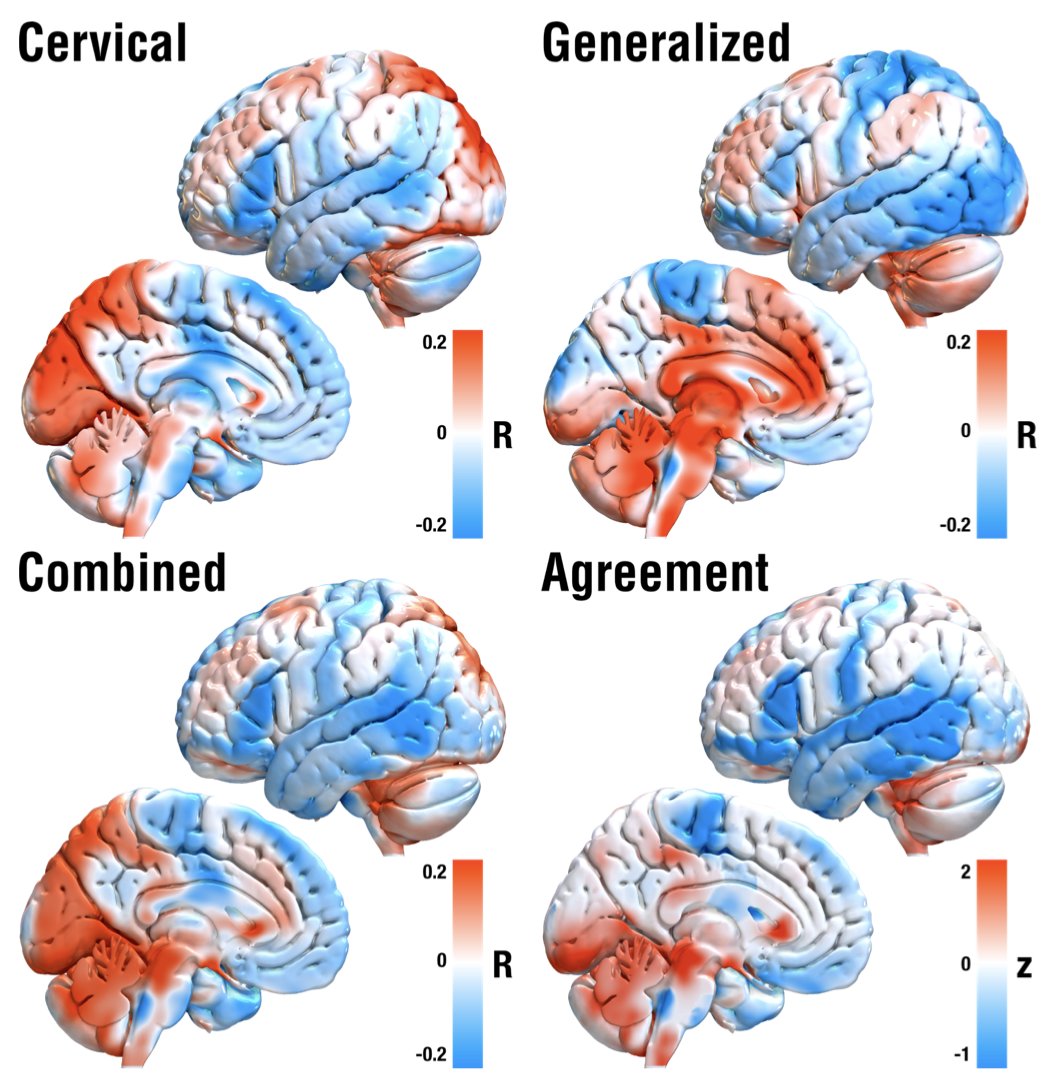
Based on N = 80 patients from five DBS centers, we mapped deep brain stimulation sites and looked at whether their structural and functional connectivity profiles could inform clinical outcomes in cervical vs. generalized dystonia. 
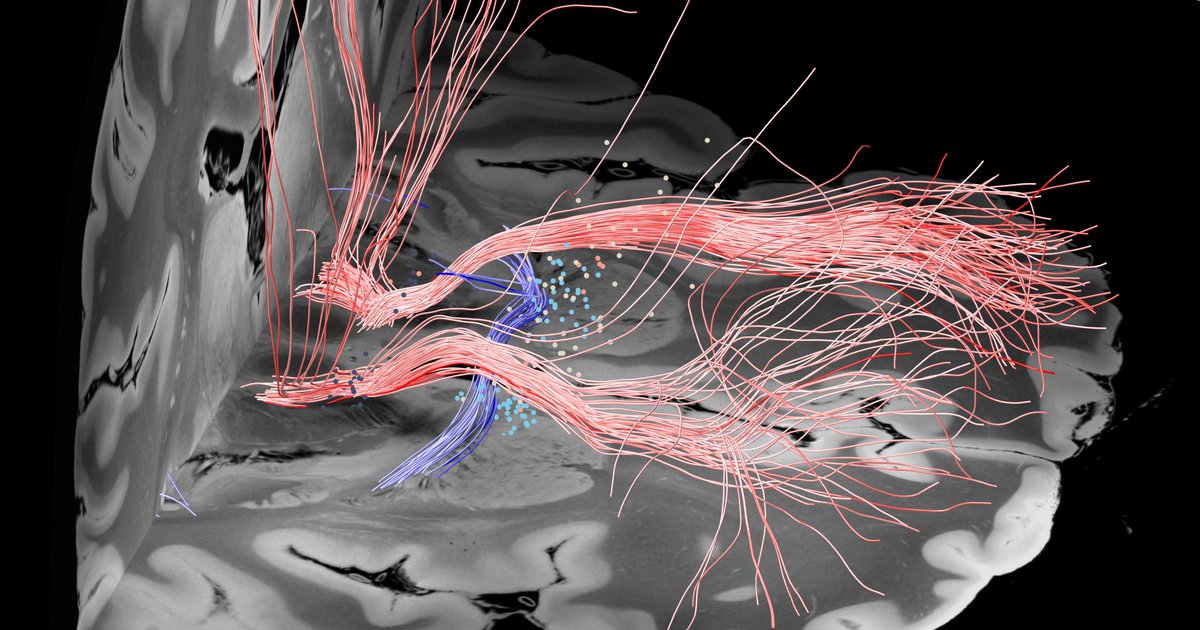
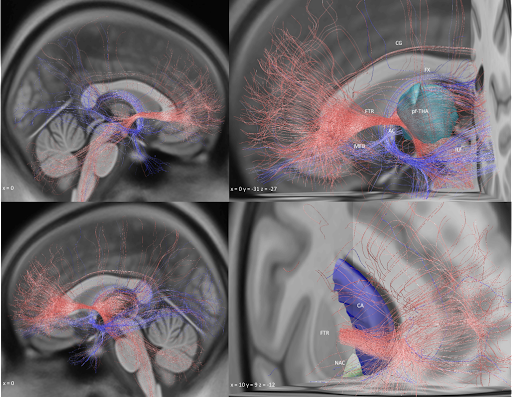
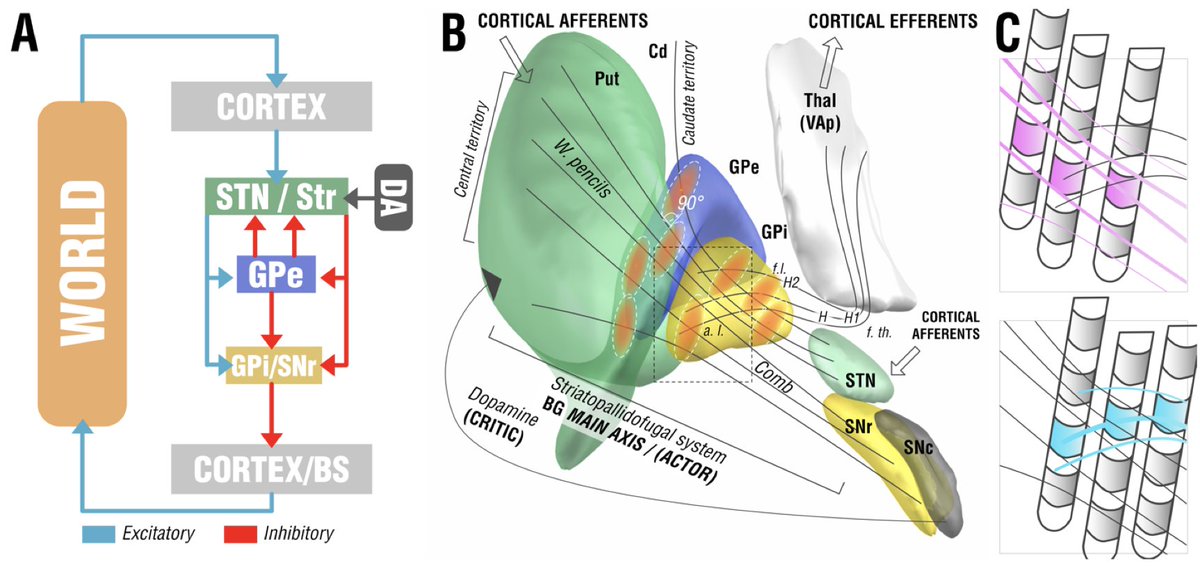
To derive at a testable hypothesis, we looked at microstructure of the internal pallidum (stimulation site) which is made up from two large processing streams that traverse *orthoginally* from each other. i) the pallidofugal bundle and ii) the pallidothalamic tracts. 

We hypothesized that we could leverage this anatomical feature of the pallidum to test whether stimulation of either system (panel C) would be associated with optimal outcomes in cervical vs. generalized dystonia. 
And indeed, patients with cervical dystonia responded best when modulating the striatopallidofugal bundle entering the internal capsule (the comb system) in its motor domain. Generalized dystonia patients instead responded best when modulating the pallidothalamic tracts. 
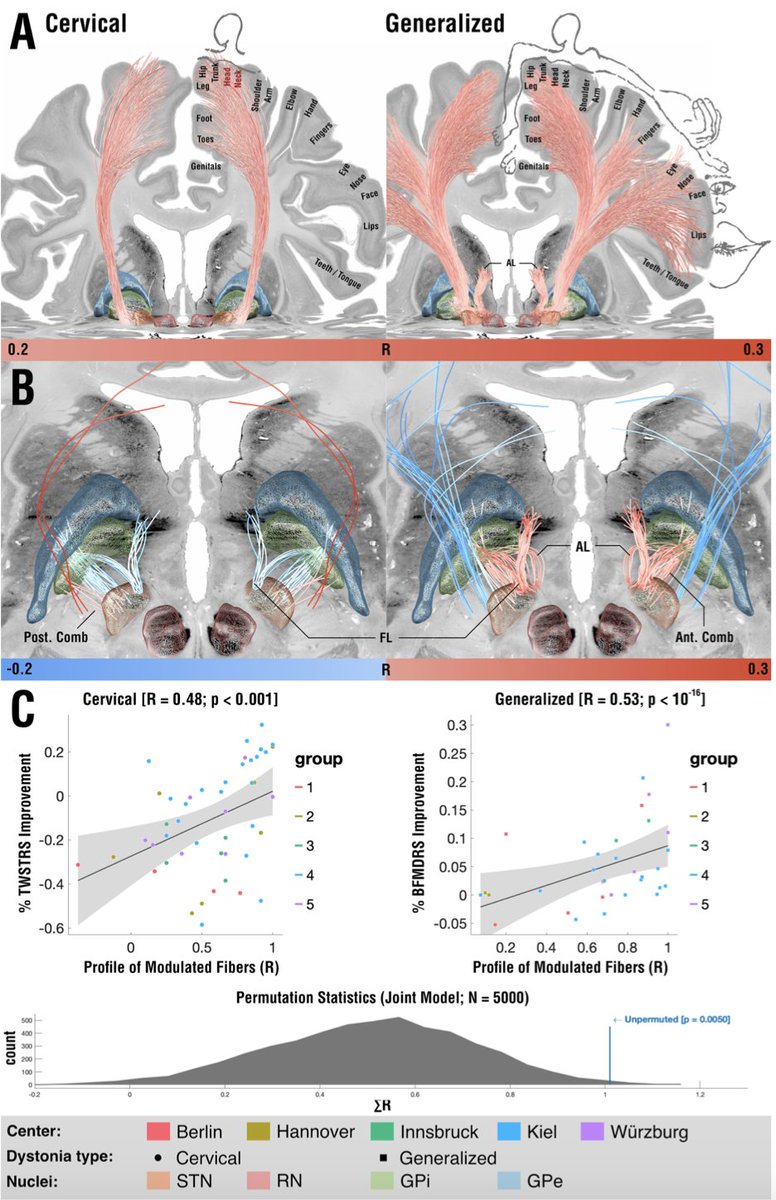
On a localized level, optimal stimulation sites for cervical dystonia mapped to the head/neck region of the pallidal homunculus, while optimal stimulation sites for generalized dystonia mapped to a broader, more diffuse area within the pallidum. 
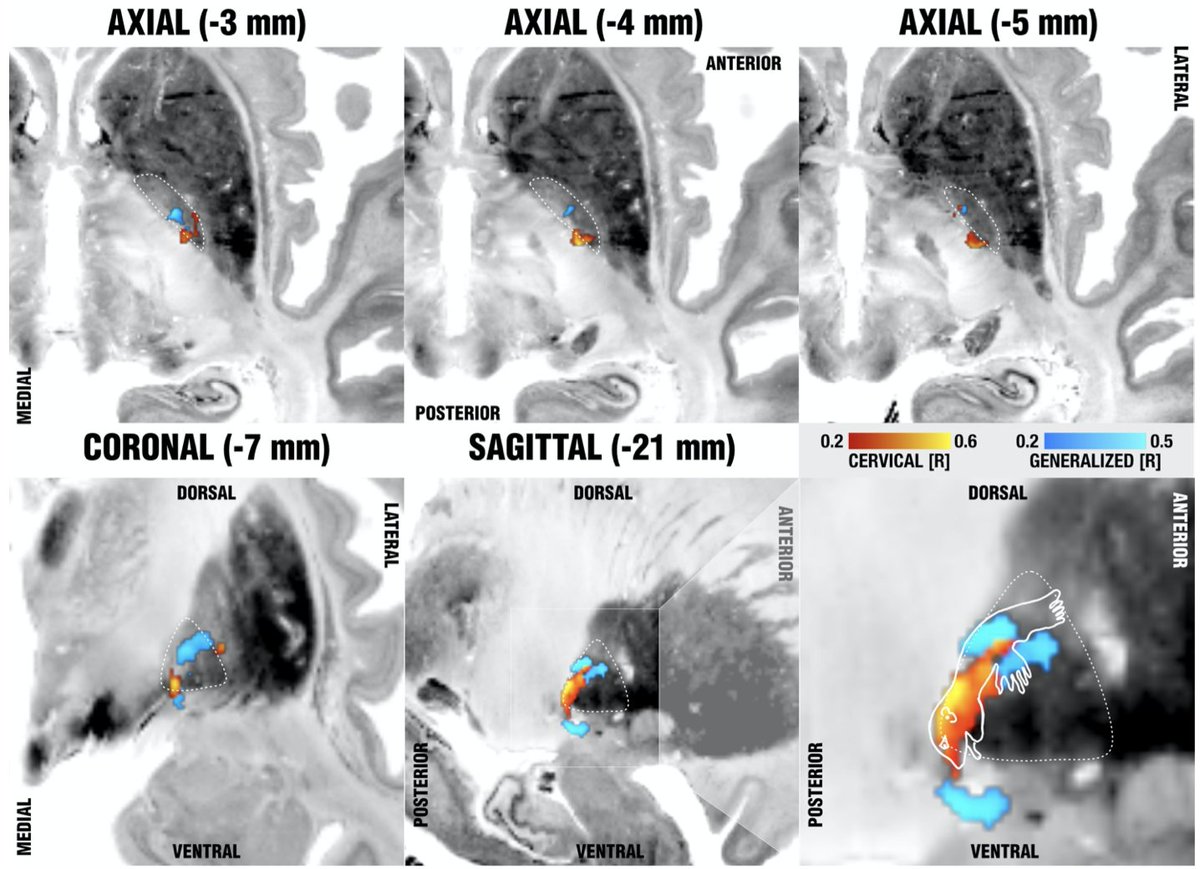
So does this mean we should modulate distinct networks in cervical vs. generalized dystonia? Well, not really. On a whole brain level, using normative rs-fMRI data, inter alia, things converged on sites in the sensorimotor cortex and cerebellum, as priorly described. 
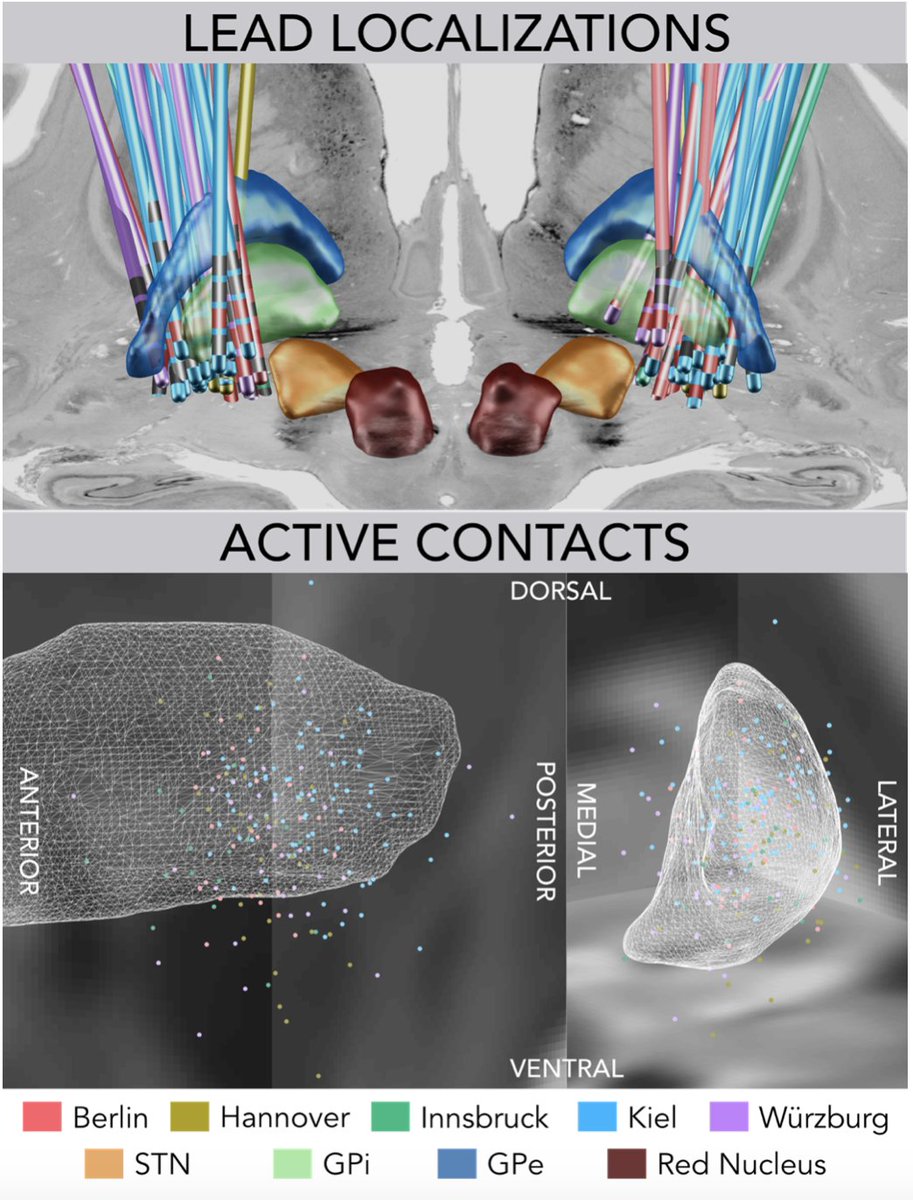
This work was possible based on the great efforts of Martin Reich and @volkmann_jens to pool together a large cohort of DBS patients and all centers that collaborated (list too long – so a picture is attached).
@NingfeiL, @Bassam_Al_Fatly, @SiobhanEwert, @simonoxen, @AA_Kuhn
@NingfeiL, @Bassam_Al_Fatly, @SiobhanEwert, @simonoxen, @AA_Kuhn

Precision of the analyses was possible due to two methodological advancements: First, the holographic basal ganglia pathway atlas published by the McIntyre lab that they graciously made openly available (@mv_petersen et al. @NeuroCellPress 2019). 
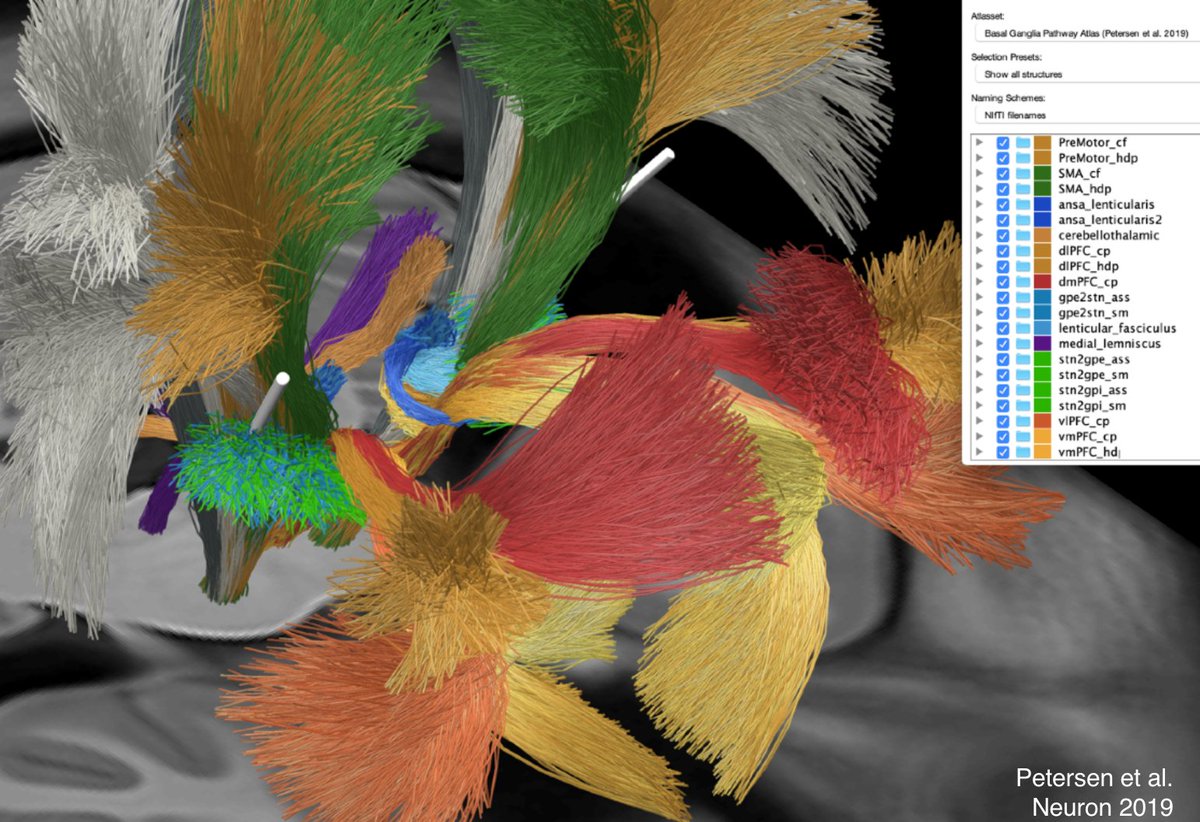
Second, @simonoxen's WarpDrive method to manually refine ANTs-based spatial normalization warp fields – see a demo here: vimeo.com/465316910
Repo here: github.com/netstim/Slicer…
Repo here: github.com/netstim/Slicer…
• • •
Missing some Tweet in this thread? You can try to
force a refresh