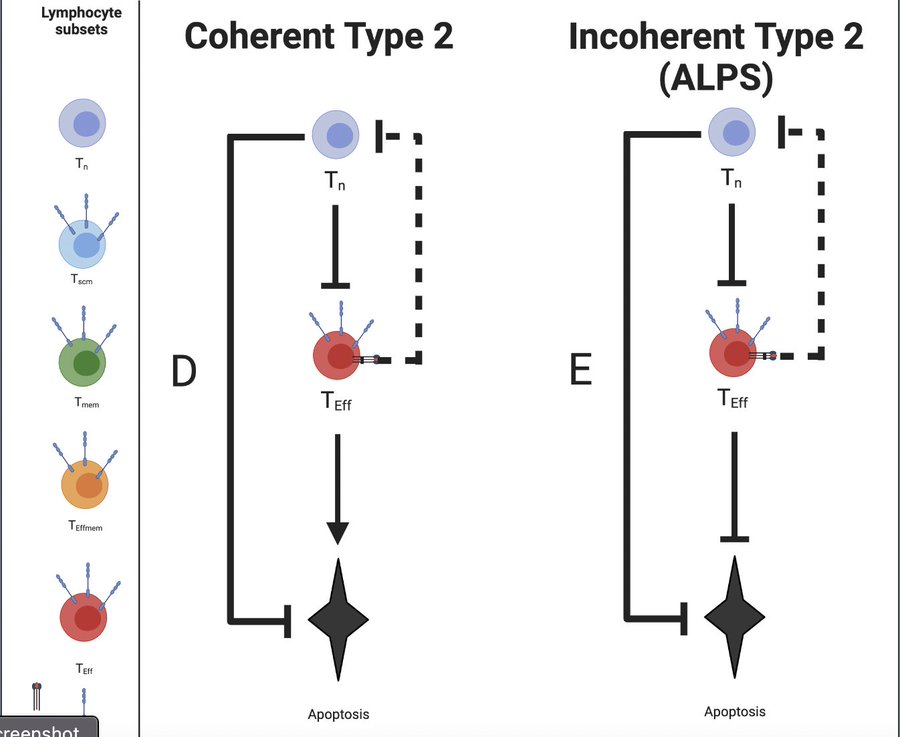
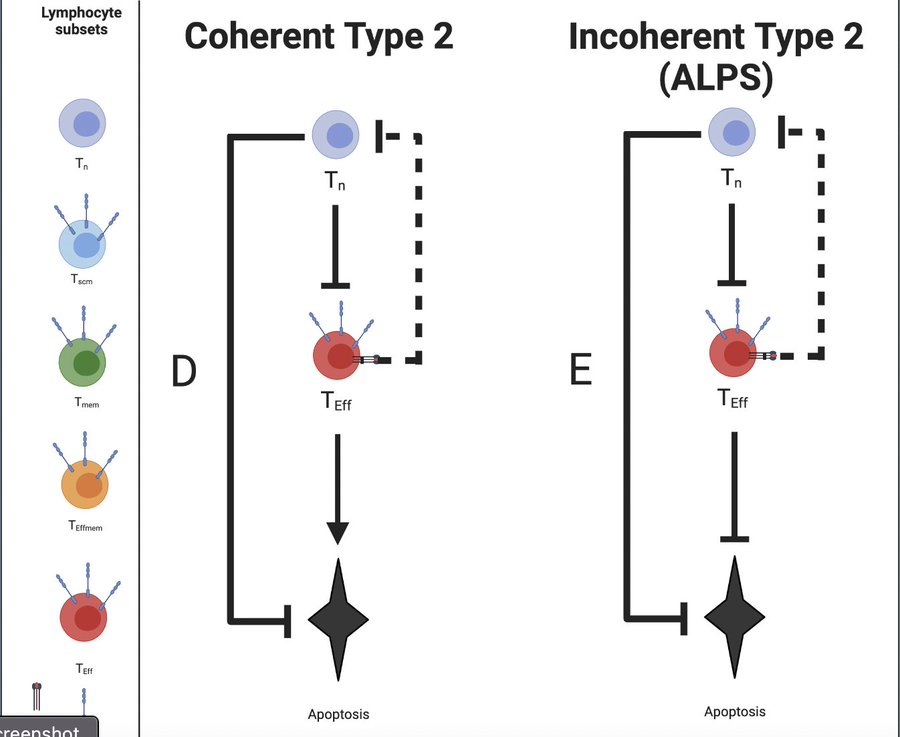
🤖 Good evening humans. I'm making an exception to my @fitterhappieraj-only rule and have translated this important thread by @VirusesImmunity. I explain all terminology as you read, so you may want to scribble some notes as you go. It's a long thread:
The study looks at how #SARS2 variants of concern (VOCs) suppress the genes (MHC I) that help killer T cells recognise the virus on cell surfaces. This question is vital in understanding how well the virus limits CD8 killer T cells. 1/
https://twitter.com/VirusesImmunity/status/1522939444856193024?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
CD8 T cells help fight off viral infection by detecting and killing infected cells. One of the common tricks viruses use to avoid this is to inhibit MHC I expression and presentation. 2/
https://twitter.com/VirusesImmunity/status/1522939446970126336?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
A previous study showed that the ancestral #SARS2 decreased the amount of MHC I in infected cells. They found the virus’s rapidly evolving protein (ORF8) played a vital role in this process. 3/
https://twitter.com/VirusesImmunity/status/1522939448215834624?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
To see whether VOCs also suppress MHC I, they looked at the surface expression of MHC I in human cells infected with #SARS2 variants. All variants significantly reduced MHC I levels compared to a mock-infected control, but no more than the Wuhan strain. 4/
https://twitter.com/VirusesImmunity/status/1522939451290177542?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
This study showed that #SARS2 suppresses MHC I expression by targeting genes involved in the cytokine response. All variants, except Gamma, decreased the amount of MHC I. But VOCs did no better than the ancestral variant. 5/
https://twitter.com/VirusesImmunity/status/1522939457355149312?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Next, the scientists investigated the role of #SARS2’s most variable gene (ORF8) in decreasing MHC I. By comparing the VOCs, they found unique ORF8 mutations in many circulating variants. 6/
https://twitter.com/VirusesImmunity/status/1522939461629145088?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
In the lab, they saw that ORF8-expressing cells had reduced expression of MHC I. The exception was Alpha with its Q27stop mutation that renders ORF8 inactive. 7/
https://twitter.com/VirusesImmunity/status/1522939465538236416?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
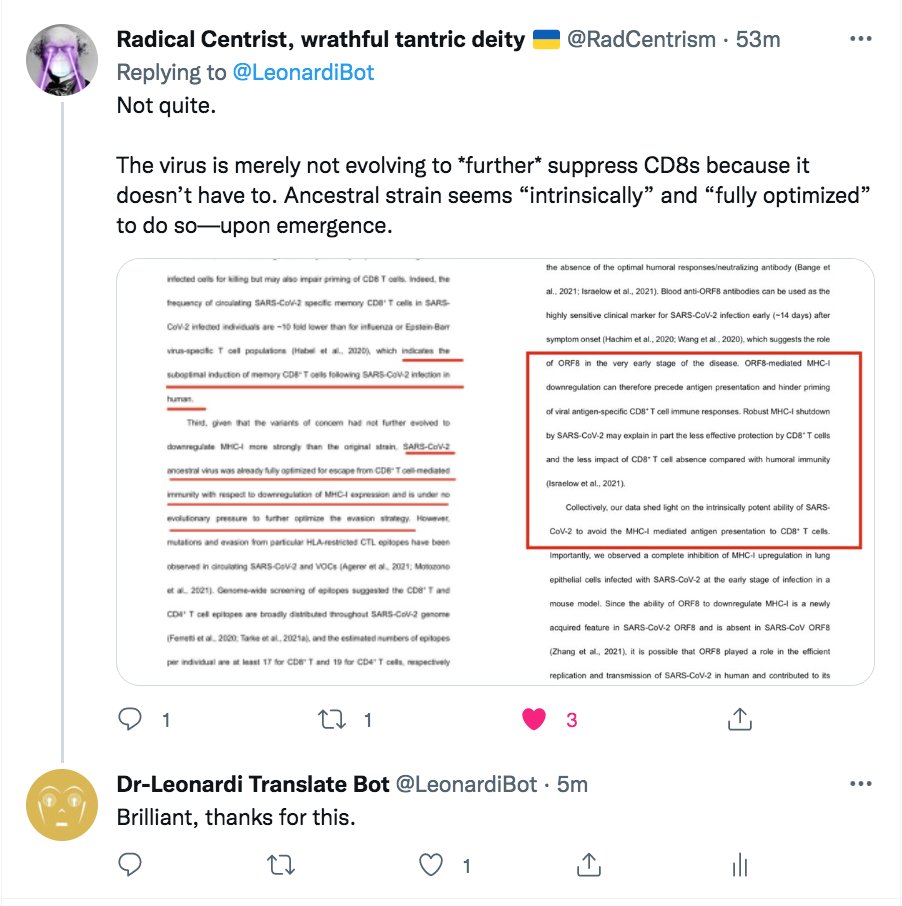
VOCs also appear better than the ancestral strain at preventing humans' natural virus inhibitors. But they do not suppress killer T cells. 8/
https://twitter.com/VirusesImmunity/status/1522939471724834816?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Remarkably, many other #SARS2 genes also reduced MHC I just as well as HIV! These #SARS2 genes also reduced MHC II (molecules found on antigen-presenting cells, like dendritic, macrophage, and B cells) to a lesser extent. 9/
https://twitter.com/VirusesImmunity/status/1522939475755560960?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Mice infected with the mouse-adapted #SARS2 showed completely reduced MHC I in infected cells of the lung lining. In contrast, the same flu-infected cells express high levels of MHC I. 10/
https://twitter.com/VirusesImmunity/status/1522939480142872576?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
#SARS2 has a powerful ability to shut down its hosts’ MHC-I system. They didn’t see the VOC’s ability to suppress MHC I any more than the Wuhan variant. However, VOCs continue to get better at evading the control of humans’ natural antiviral proteins. 11/
https://twitter.com/VirusesImmunity/status/1522939481837293568?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Thank you, Prof. Iwasaki, @MiyuMoriyama, @carolilucas and @ValterVSM for your excellent work. 12/
https://twitter.com/VirusesImmunity/status/1522939482789404672?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Prof Iwasaki recommends this excellent summary of her and others’ work: news-medical.net/news/20220506/… 13/
If #SARS2 is so good at shutting down MHC I, why aren’t infected cells detected & destroyed by natural killer (NK) cells? In mild cases, maybe. In severe cases, it dysregulates the human immune response, inhibiting natural antivirals. 14/
https://twitter.com/VirusesImmunity/status/1522939484639088641?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Conclusion: #SARS2 VOCs don't evolve to escape from killer T cells more than the Wuhan strain. The virus can inhibit MHC I in many ways. If one is missing, others compensate. So, T cell-based therapeutic approaches may be difficult for COVID-19. 15/END 🤖
https://twitter.com/VirusesImmunity/status/1522939485725462528?s=20&t=SMfCYtb4rvOKl1eAYL_Svw
Addendum: Users, please note these helpful comments on my translation from @fitterhappierAJ and @RadCentrism. Thank you for your input. 

• • •
Missing some Tweet in this thread? You can try to
force a refresh