
Prof Darrel Francis ☺ Mk CardioFellows Great Again
14 Sep,
56 tweets, 11 min read
Here are 4 choices
When will they stop taking the statin?
At 12 votes, 2/3 of people are recommending "take a detailed history".
That is correct for Medical School Finals, as a general waffle answer, but this is a scientific thread, where we can't handwave away questions by saying "each patient is an individual".
That is correct for Medical School Finals, as a general waffle answer, but this is a scientific thread, where we can't handwave away questions by saying "each patient is an individual".
In this "detailed history" which you people are so keen to be acquiring
(Remind me never to invite you a dinner party as you will be a pretty boring conversationalist. At least remind me not to ask you, "How are you?" except on a zoom call where I can switch off...)
(Remind me never to invite you a dinner party as you will be a pretty boring conversationalist. At least remind me not to ask you, "How are you?" except on a zoom call where I can switch off...)
In this "detailed history" of the side-effects, what will you focus on? (Assuming you are focussing on anything in particular, and not just parroting a non-answer that you have been trained to give, in Medical School Finals)
The more you ask about the symptoms, what happens to the patient?
If you doubt your answer, have you ever noticed how by the time people show up for angiography, their symptoms have gradually progressed to the centre of the chest?
When people first show up at the front door, their chest pains are all over the place, described in all sorts of ways.
But gradually they funnel, after each medical student, nurse or doctor they speak to.
But gradually they funnel, after each medical student, nurse or doctor they speak to.

By the time the Old Buffer ProfDFrancis shows up, positions his monocle, and enquires, "So tell me what happened, in your own words, Mrs Jones," they announce in well-rehearsed tones:
"I got this crushing pain in my central chest, radiating ..."
"I got this crushing pain in my central chest, radiating ..."
Anyway, even if you are a detailed-history fascist, I can ignore your tedious enumeration of the patient's presence or absence of burning, twisting, thumping, scratching, stabbing etc nature of the pain,
and focus on what I agree is most interesting: the time course.
and focus on what I agree is most interesting: the time course.
Suppose the patient has stopped tablets because the symptoms were so intense.
When would they try restarting?
When would they try restarting?
OK, here is the plan our community has recommended. It is current best practice:
1. Take a "detailed history" (whatever that means).
Then tell them:
2. "If symptoms very bad, stop taking."
3. "Once symptoms have resolved, restart."
4. "Come back and report how you got on."
1. Take a "detailed history" (whatever that means).
Then tell them:
2. "If symptoms very bad, stop taking."
3. "Once symptoms have resolved, restart."
4. "Come back and report how you got on."
When they come back in a few months' time, how much detail can the provide about their symptomatic progress during their informal experimentation?
Typically.
Typically.
Yup. Whatever the tablet is, and whatever the side effect is.
That is not because they are stupid or trying to deceive you.
It is because you set them up to screw themselves over.
THEY DIDN'T SYSTEMATICALLY DOCUMENT SYMPTOMS IN AN ANALYSABLE WAY.
That is not because they are stupid or trying to deceive you.
It is because you set them up to screw themselves over.
THEY DIDN'T SYSTEMATICALLY DOCUMENT SYMPTOMS IN AN ANALYSABLE WAY.
OK let's fix that. We give them a diary. And make them write down contemporaneously how they are feeling. 

Remember, YOU said that they would stop taking tablets when their symptoms were maximal.
What always happens (by definition!) after something is maximal?
What always happens (by definition!) after something is maximal?
So, regardless of what the tablet is, or what the symptom is, what will happen to the symptoms if they systematically stop the tablet when symptoms are maximal?
It's actually guaranteed. No need to do the experiment! You have shafted them by FORCING them to have a symptom regression when the tablet is stopped.
Meanwhile, you have also said that they will restart the tablets when the symptoms have resolved. You don't need to tell them that: that is obvious and only a madman would voluntarily restart a drug when the bad symptoms are still there.
But over a series of weeks and months, if the tablets are only restarted when symptoms are zero, what MUST happen to symptoms when the tablets are restarted?
So what *must* you do if you want to avoid this self-inflicted false-positive phenomenon?
OK so we have debunked 2 things in the standard management plan of "informal experiments", as actively harmful:
A. No formal documentation.
B. No pre-set time schedule.
But don't worry, "detailed history" to the rescue!
A. No formal documentation.
B. No pre-set time schedule.
But don't worry, "detailed history" to the rescue!
Compared to the time course of the symptoms induced by placebo, how do symptoms genuinely induced by statins develop?
Right now in this quiz people seem to be more interested in the OFFSET. In a way, it makes sense, in the same way that we focus on the offset of SVT symptoms (because people have their attention on it).
So what are the OFFSET kinetics of symptoms from statin versus from placebo?
So what are the OFFSET kinetics of symptoms from statin versus from placebo?
Today James Howard and Frances Wood (congrates to her for being a Psychologist, Nurse, and now PhD!) have published the full results of the SAMSON trial
jacc.org/doi/pdf/10.101…
whose results had previously only been shown as an AHA abstract and associated Research Letter.
jacc.org/doi/pdf/10.101…
whose results had previously only been shown as an AHA abstract and associated Research Letter.
Grey are days with "no tablet", and the two colours represent days with either statin or placebo. Which days are statin, and which placebo?
I have to do a meeting for a couple of hours but will be back afterwards to discuss further.
I have to do a meeting for a couple of hours but will be back afterwards to discuss further.
Hmmm, so the onsets are pretty identical, between onset of symptoms on STATIN (red) and onset of symptoms on PLACEBO (blue).
But most people seemed more interested in the OFFSET, yeh?
Here is EVERY day of data, from EVERY offset from tablet to no-tablet, in EVERY patient in the trial.
(As in the onset data, they are sorted by how "impressive" they are.)
Here is EVERY day of data, from EVERY offset from tablet to no-tablet, in EVERY patient in the trial.
(As in the onset data, they are sorted by how "impressive" they are.)
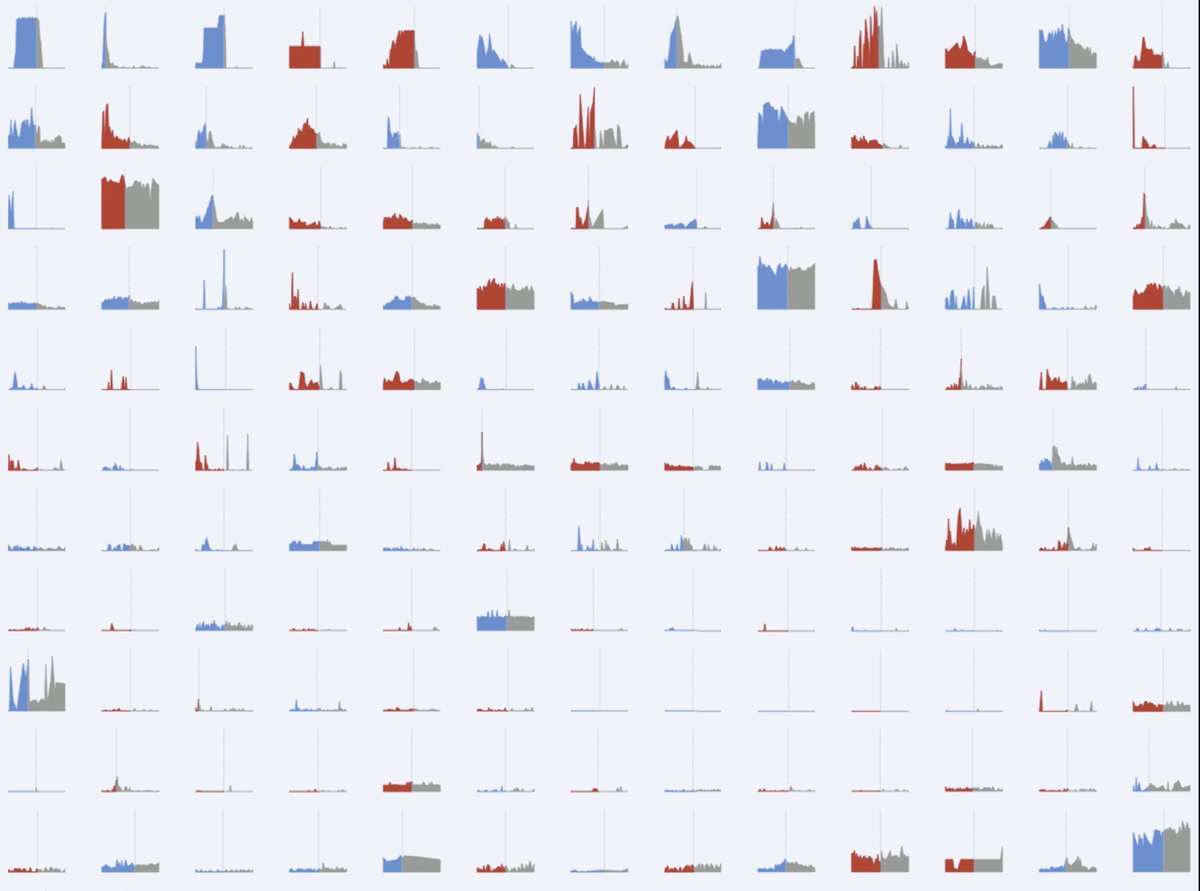
Having reviewed all the individual data points, how can you tell from the pattern of onset that it is statin and not placebo?
Nor can you tell from the pattern of the offset.
More interesting and upsetting, though is the implication of all of this for current best practice.
More interesting and upsetting, though is the implication of all of this for current best practice.
When you
1. Take a "detailed history" and note it all down, with a furrowed brow,
2. Get them to do informal experiments WITHOUT ANALYSABLE DOCUMENTATION,
3. STARTING tabs when symptoms are minimal.
4. STOPPING tabs when symptoms are maximal
What are you doing for the patient?
1. Take a "detailed history" and note it all down, with a furrowed brow,
2. Get them to do informal experiments WITHOUT ANALYSABLE DOCUMENTATION,
3. STARTING tabs when symptoms are minimal.
4. STOPPING tabs when symptoms are maximal
What are you doing for the patient?
Suppose, from today, you make a firm commitment to never do this making-worse of patients.
"Secundum non nocere, after a few decades of nocere'ing."
ALWAYS enforce an analysable diary
ALWAYS set prespecified start/stop dates
How good will that be?
"Secundum non nocere, after a few decades of nocere'ing."
ALWAYS enforce an analysable diary
ALWAYS set prespecified start/stop dates
How good will that be?
Because ... you will see what SAMSON saw
DAILY documentation on an App
PRESET start and stop dates
Yielded clear onset and offset of symptoms when tablets are started and stopped.
DAILY documentation on an App
PRESET start and stop dates
Yielded clear onset and offset of symptoms when tablets are started and stopped.
The only problem was,
this "clear onset and offset"
was the same for statin and placebo.
If you don't have placebo, you and (more importantly) the patient will misinterpret this as evidence that the STATIN is the cause.
this "clear onset and offset"
was the same for statin and placebo.
If you don't have placebo, you and (more importantly) the patient will misinterpret this as evidence that the STATIN is the cause.
What we really want to know is:
"Within the STATIN TABLET,
Is it the STATIN
or the TABLET
that is the cause of the symptoms?"
"Within the STATIN TABLET,
Is it the STATIN
or the TABLET
that is the cause of the symptoms?"
In SAMSON you can see how often it was each of those, from the full individual patient data we have published in the full paper in JACC.
The trial was funded by the British Heart Foundation, which pays to have its research open-accessed:
jacc.org/doi/pdf/10.101…
The trial was funded by the British Heart Foundation, which pays to have its research open-accessed:
jacc.org/doi/pdf/10.101…
What stops people from doing SAMSON themselves is the lack of easy access to high-quality placebo as in SAMSON, i.e. manufactured to be identical in appearance, rather than just over-encapsulated which is easy to defeat for curious patients.
We now offer an NHS service for patients with statin-tablet intolerance, where they do a protocol like SAMSON's, and find out for themselves whether the statin or the tablet is the problem.
Only 1 London visit needed
You or the patient can contact us on d.francis@imperial.ac.uk
Only 1 London visit needed
You or the patient can contact us on d.francis@imperial.ac.uk
The reason for the visit to London is so we can give you the tablets.
https://twitter.com/susanbconnolly/status/1437737924775366659?s=20
I don't want to go to prison for mailing people tablets that include placebos. Someone told me it could be illegal?
If anyone knows how we can do it as purely distance, i.e. no visits to London, with nobody going to prison, I am very keen to hear!
If anyone knows how we can do it as purely distance, i.e. no visits to London, with nobody going to prison, I am very keen to hear!
Any clinician or institution willing to do the getting of the placebos and packaging up the sets of real and placebos and giving to patients locally, we are delighted to provide the App side of things virtually.
Let us know on the above email, many thanks.
Let us know on the above email, many thanks.
International sites VERY welcome to join. Again, we will provide the App work, if you can acquire the placebo and give to the patient.
By the way, I think the correct answers to the contentious questions are as follows.
This one:
All options are wrong.
They all leave the patient worse off.
This one:
All options are wrong.
They all leave the patient worse off.

Finally, and most crushingly, this is the answer to how good is it to do
Experiments on Symptom Causation
Without Placebo
Experiments on Symptom Causation
Without Placebo

Without placebo, you will get an answer that is
Simple
Clear
and
Wrong.
Don't like the bluntness of reality?
Sorry.
Move to another universe.
Simple
Clear
and
Wrong.
Don't like the bluntness of reality?
Sorry.
Move to another universe.

Excellent point made by Holly Gonazales @HollyGHeartMed! 
That is definitely the right instinct for us as healthcare workers: we are in this vocation because we care about people and their experiences.
But there is a downside too.
We and the patient are both human, and voicing our curiosity tends to erode a channel in both our minds.
But there is a downside too.
We and the patient are both human, and voicing our curiosity tends to erode a channel in both our minds.

Just as tourists interested in experiencing a site, unwittingly damage that site,
we as clinicians interested in understanding a patient's suffering, may unwittingly
(a) enhance that suffering and, more insidiously,
(b) create a false belief:
we as clinicians interested in understanding a patient's suffering, may unwittingly
(a) enhance that suffering and, more insidiously,
(b) create a false belief:
The dangerous false belief is that
"by considering in great detail the nature and temporal pattern of the symptoms, we can find out the true cause"
Actually we can't.
"by considering in great detail the nature and temporal pattern of the symptoms, we can find out the true cause"
Actually we can't.
Yes there are published guidelines on how to tell if symptoms are from the statin or not.
But there are also published guidelines on how to assess personality from feeling the bumps on people's heads.
They are both mumbo jumbo.
When tested scientifically, they don't work.
But there are also published guidelines on how to assess personality from feeling the bumps on people's heads.
They are both mumbo jumbo.
When tested scientifically, they don't work.

Yes we have been doing this for decades.
But people have been dowsing for water with rods for decades. So what, it is still bullshit.
Its time we stop the pseudoscience of guessing the origin of statin-TABLET symptoms without matched placebo control.
But people have been dowsing for water with rods for decades. So what, it is still bullshit.
Its time we stop the pseudoscience of guessing the origin of statin-TABLET symptoms without matched placebo control.

• • •
Missing some Tweet in this thread? You can try to
force a refresh














