
Anti-platelet glycoprotein autoantibodies after ChAdOx1 vaccination are as common in VITT as in non-VITT thrombocytopenic patients. tinyurl.com/2y2na7wu #VITT 1/3 

Of greater importance is the observation that in mice, intravenous but not intramuscular administration of ChAdOx1 (AZ vaccine) leads to platelet-adenovirus aggregates and platelet activation. tinyurl.com/2y2na7wu #VITT 2/3 
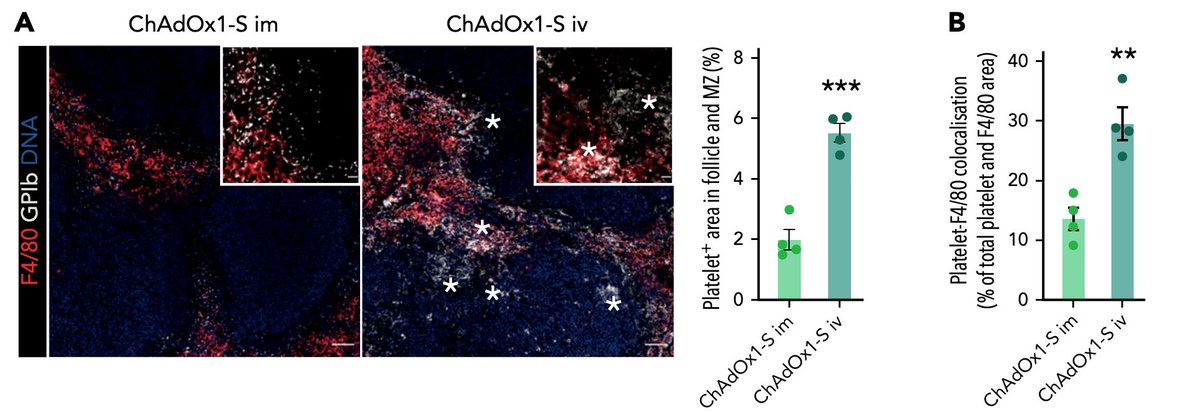
Although this is an animal study, it is nevertheless important because it shows the differential response to intramuscular and intravenous exposure to ChAdOx1 vaccine. #VITT 3/3
• • •
Missing some Tweet in this thread? You can try to
force a refresh







