Thus initial state is T1 = T2 = Tavg.
1/
Just separate the plates so that g = 1/2 - epsilon.
2/
200 = g ( R1 + R2 )
and for thermal equilibrium of Plate #2:
(1-g) R1 = R2
since only the radiant energy from plate 1 is heating plate 2.
3/
200 = g R1 + g (1-g) R1 = R1 g (2 - g)
and then
200 = k T1^4
= g(2-g) k T1'^4
T1' = T1 / (g(2-g))^0.25
T2' = T1' (1-g)^0.25 = T1 (1-g)^0.25 / (g(2-g))^0.25
Tavg' = (1/2) T1 (1 + (1-g)^0.25) / (g(2-g))^0.25
4/
Tavg' = (1/2) Tavg (1 + (1-g)^0.25) / (g(2-g))^0.25
and the final equilibrium is described by a geometric factor in g
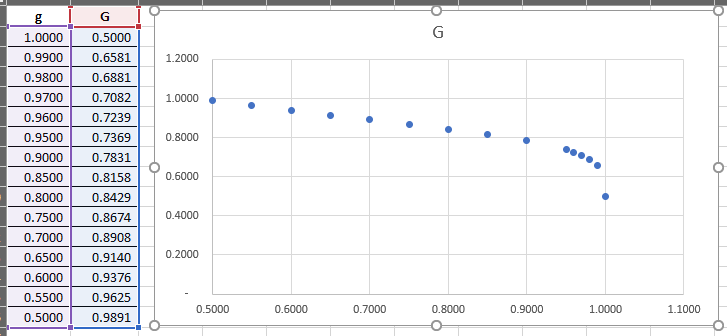
G = (1/2) (1 + (1-g)^0.25) / (g(2-g))^0.25
with
Tavg' = G Tavg
where
g = 1 => G = 0.50
g = 1/2 => G ~ 0.99
/5
/6
1) The usual analysis "introduces" the second plate into the closed system. This is thermodynamically unacceptable - the second plate must be in the closed system from the start.
/7
a) at a very great distance from Plate #1, which will be approximated as infinity;
b) initially in contact with Plate #1, forming a starting "double plate".
/8
a) g = 1 => G = 0.50
b) g = 1/2 => G ~ 0.99
/9
Q: Does this require a resistant internal force to said motion?
A: Unsure, but I think so. My Thermo is really rusty.
/10
After "just separating" Tavg' drops to Tavg', and drops further as the separation increases (g increasing).
/11
Q: Another resistant force?
A: ??
If true that there is a resistant force in both cases - it is a frictional force not a potential one since opposing motion in both cases.
Interesting!?!?
/12
From above
T1' - T2' = T1 / (g(2-g))^0.25 - T1 (1-g)^0.25 / (g(2-g))^0.25
= T1 ( 1 - (1-g)^0.25 ) / (g(2-g))^0.25
/13





