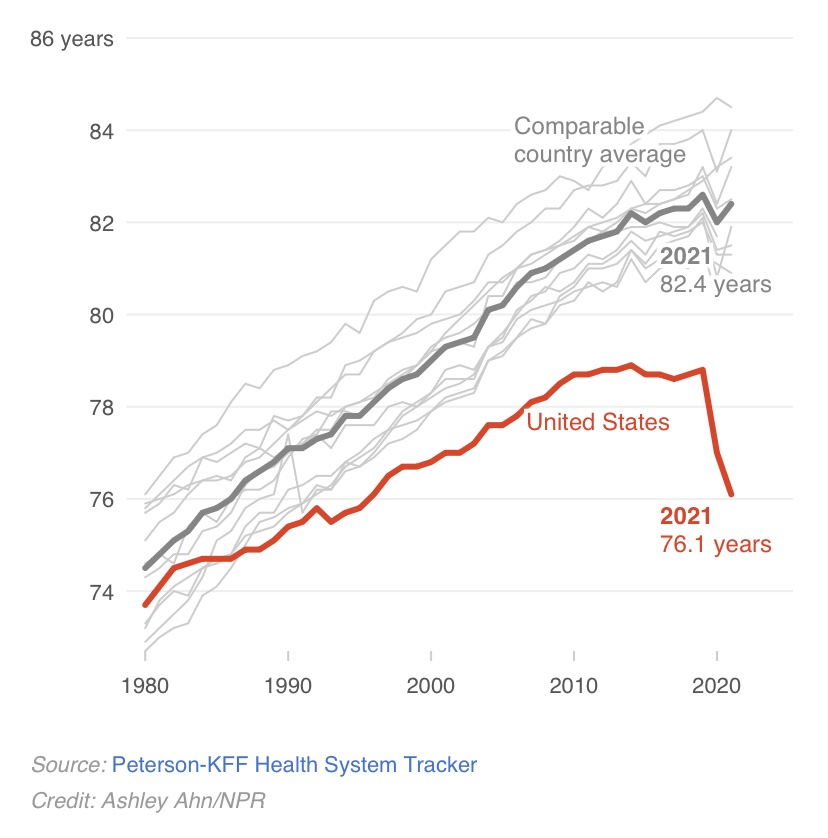
1- My current take on whether anti-amyloid therapies are likely to have meaningful impact on cognition, functioning & quality of life of people living w/ #dementia due to #Alzheimers disease:
Not likely.
For details, based on my recent Grand Rounds at Penn St, read on.
Not likely.
For details, based on my recent Grand Rounds at Penn St, read on.
2- History:
After finding that active immunization with Abeta42 in mice that overexpressed APP resulted in virtual elimination of amyloid deposits, there was great enthusiasm for anti-amyloid therapies - and for the amyloid hypothesis.
pubmed.ncbi.nlm.nih.gov/10408445/
After finding that active immunization with Abeta42 in mice that overexpressed APP resulted in virtual elimination of amyloid deposits, there was great enthusiasm for anti-amyloid therapies - and for the amyloid hypothesis.
pubmed.ncbi.nlm.nih.gov/10408445/

3- More history:
Unfortunately, active immunization with Abeta42 in humans resulted in unacceptable side effects.
Passive immunization with various anti-amyloid monoclonal antibodies failed in multiple studies.
It seemed like this was the end of the line, until ...
Unfortunately, active immunization with Abeta42 in humans resulted in unacceptable side effects.
Passive immunization with various anti-amyloid monoclonal antibodies failed in multiple studies.
It seemed like this was the end of the line, until ...

4- Despite an advisory panel voting against approval, @US_FDA granted accelerated approval for aducanumab.
What did they base this on?
What did they base this on?

5- Aducanumab is extremely good at clearing amyloid.
As are many of the other anti-amyloid monoclonal antibodies.
Which should not be surprising because that is exactly what they are designed to do.
jpreventionalzheimer.com/5919-two-rando…
As are many of the other anti-amyloid monoclonal antibodies.
Which should not be surprising because that is exactly what they are designed to do.
jpreventionalzheimer.com/5919-two-rando…

6- But aducanumab had very little effect on cognition.
In ENGAGE, no sign of benefit relative to placebo.
There was slight benefit w/ high-dose aducanumab in EMERGE, though as @LonSchneiderMD pointed out, this could be due to cognition worsening more in placebo group.
In ENGAGE, no sign of benefit relative to placebo.
There was slight benefit w/ high-dose aducanumab in EMERGE, though as @LonSchneiderMD pointed out, this could be due to cognition worsening more in placebo group.

7- There is reason to be skeptical of the hypothesis that reducing amyloid results in improved cognition.
To wit, this systematic review & meta-analysis found very weak correlation between amyloid reduction & improved cognition across 17 MAB studies:
pubmed.ncbi.nlm.nih.gov/33831607/
To wit, this systematic review & meta-analysis found very weak correlation between amyloid reduction & improved cognition across 17 MAB studies:
pubmed.ncbi.nlm.nih.gov/33831607/

7b - Take another look at this scatter plot. If you throw out the apparent outlier in the bottom left, do you think there would be any correlation between amyloid reduction & cognitive improvement? 

8- Back to aducanumab studies.
How representative were the 3000+ subjects wrt race & ethnicity?
Not at all.
That's both a scientific problem (how generalizable are these results or, for that matter, the amyloid hypothesis?) and a problem of health equity (how did this happen?)
How representative were the 3000+ subjects wrt race & ethnicity?
Not at all.
That's both a scientific problem (how generalizable are these results or, for that matter, the amyloid hypothesis?) and a problem of health equity (how did this happen?)

9- Then there are side effects, esp amyloid-related imaging abnormalities (ARIA).
31% of aducanumab subjects got ARIA-E (edema), more if higher dose or if apoE e4+.
It's usually mild & reverses w/ discontinuation. But, still, it's brain edema. And it requires MRI monitoring.
31% of aducanumab subjects got ARIA-E (edema), more if higher dose or if apoE e4+.
It's usually mild & reverses w/ discontinuation. But, still, it's brain edema. And it requires MRI monitoring.
9b - 18% of aducanumab subjects got ARIA-H (hemorrhage), with significant overlap with ARIA-E.
Usually asymptomatic. But, again, we will increase the risk of brain bleeding.
We will need to be very mindful of contraindications: being on blood thinner, h/o CNS bleed or stroke.
Usually asymptomatic. But, again, we will increase the risk of brain bleeding.
We will need to be very mindful of contraindications: being on blood thinner, h/o CNS bleed or stroke.
10- Of course, there's the cost. The drug itself will be $28,000 per year.
But a recent analysis found that aducanumab would need to be < $3000/y to be cost effective.
jamanetwork.com/journals/jaman…
But a recent analysis found that aducanumab would need to be < $3000/y to be cost effective.
jamanetwork.com/journals/jaman…
11- And there will be a lot of other expenses:
- biomarker to establish amyloid positivity
- baseline & surveillance MRIs
- apo E testing & genetic counseling
- infusion center
Plus expertise to interpret amyloid PET (for dx) and to identify & assess severity of ARIA.
- biomarker to establish amyloid positivity
- baseline & surveillance MRIs
- apo E testing & genetic counseling
- infusion center
Plus expertise to interpret amyloid PET (for dx) and to identify & assess severity of ARIA.
12- What does the future hold for monoclonal antibodies?
We are waiting for results of studies of
- donanemab
- lecanemab
- gantenerumab
- solanezumab (A4)
Biogen is required to complete another aducanumab study as part of FDA accelerated approval.
We are waiting for results of studies of
- donanemab
- lecanemab
- gantenerumab
- solanezumab (A4)
Biogen is required to complete another aducanumab study as part of FDA accelerated approval.
13- Many other types of agents in the pipeline.
Perhaps not surprisingly, since neurofibrillary tangles are within neurons, anti-tau antibody therapies have failed.
Perhaps not surprisingly, since neurofibrillary tangles are within neurons, anti-tau antibody therapies have failed.

14- Conclusion, part 1
1. I think it's unlikely that anti-amyloid antibodies will have an impact on the care of people with #Alzheimers disease.
2. Blood-based biomarkers are coming soon and could result in a major change in how we diagnose AD.
1. I think it's unlikely that anti-amyloid antibodies will have an impact on the care of people with #Alzheimers disease.
2. Blood-based biomarkers are coming soon and could result in a major change in how we diagnose AD.
15- Conclusion, part 2
Treatment of dementia requires a holistic approach:
- eliminating medications/substances that worsen cognition
- educating/supporting families & caregivers
- socializing, cognitive stimulation, exercise, diet
- supporting functioning
- addressing BPSD
Treatment of dementia requires a holistic approach:
- eliminating medications/substances that worsen cognition
- educating/supporting families & caregivers
- socializing, cognitive stimulation, exercise, diet
- supporting functioning
- addressing BPSD
• • •
Missing some Tweet in this thread? You can try to
force a refresh