If you've already had COVID-19, would a *single* dose of Pfizer-BioNTech/Moderna provide a boost?
This brief report brings antibody titer data: yes.
This suggests a possible dose-sparing strategy to improve vaccine rollout... 1/4
medrxiv.org/content/10.110…
This brief report brings antibody titer data: yes.
This suggests a possible dose-sparing strategy to improve vaccine rollout... 1/4
medrxiv.org/content/10.110…

If vax & infection provided equivalent immunity, a dose-sparing strategy targeting seronegatives could be thought of as increasing vax supply (pic).
Ex: se 96.6%, sp 99.9% (Roche), 25% seroprev. = 32% increase in vax supply. 2/4
Modeled in this paper: science.sciencemag.org/content/early/…
Ex: se 96.6%, sp 99.9% (Roche), 25% seroprev. = 32% increase in vax supply. 2/4
Modeled in this paper: science.sciencemag.org/content/early/…

But taking the idea from the medrxiv report cited above, and using *one* dose for each seropositive and *two* doses for each seronegative, one can derive a similar formula (pic).
Ex: se 96.6%, sp 99.9% (Roche), 25% seroprev. = 16% bonus vax supply. 3/4
Ex: se 96.6%, sp 99.9% (Roche), 25% seroprev. = 16% bonus vax supply. 3/4
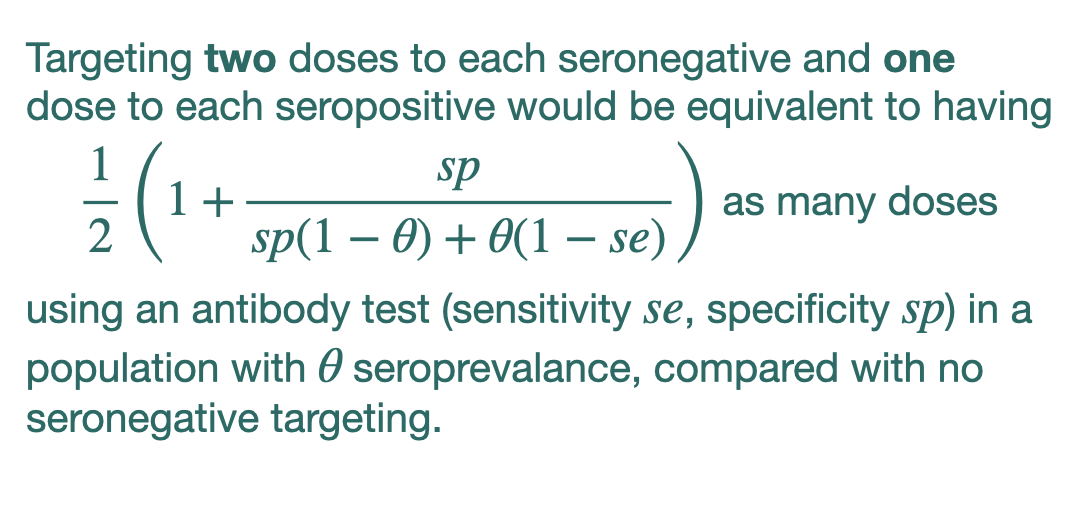
When I write "32% bonus vax supply" or "16% bonus vax supply", I think of that as a theoretically possible increase in the accumulation of protection in the population.
In other words, an increase in rollout speed (for the same # of vaccines).
In other words, an increase in rollout speed (for the same # of vaccines).
Of course, there are costs too: price of antibody tests, logistics of it all, headaches of messaging.
Reality: Probably too difficult to do broadly, but there may be situations where this dose sparing would be valuable. 4/4
h/t @mlipsitch for pointing out the medrxiv. 👍
Reality: Probably too difficult to do broadly, but there may be situations where this dose sparing would be valuable. 4/4
h/t @mlipsitch for pointing out the medrxiv. 👍
A 2nd study also preprinted today draws similar conclusions re antibodies!
"the antibody response to the 1st vaccine dose in individuals with pre-existing immunity is equal to or even exceeds the titers found in naive individuals after the 2nd dose" 5/4
medrxiv.org/content/10.110…
"the antibody response to the 1st vaccine dose in individuals with pre-existing immunity is equal to or even exceeds the titers found in naive individuals after the 2nd dose" 5/4
medrxiv.org/content/10.110…

• • •
Missing some Tweet in this thread? You can try to
force a refresh