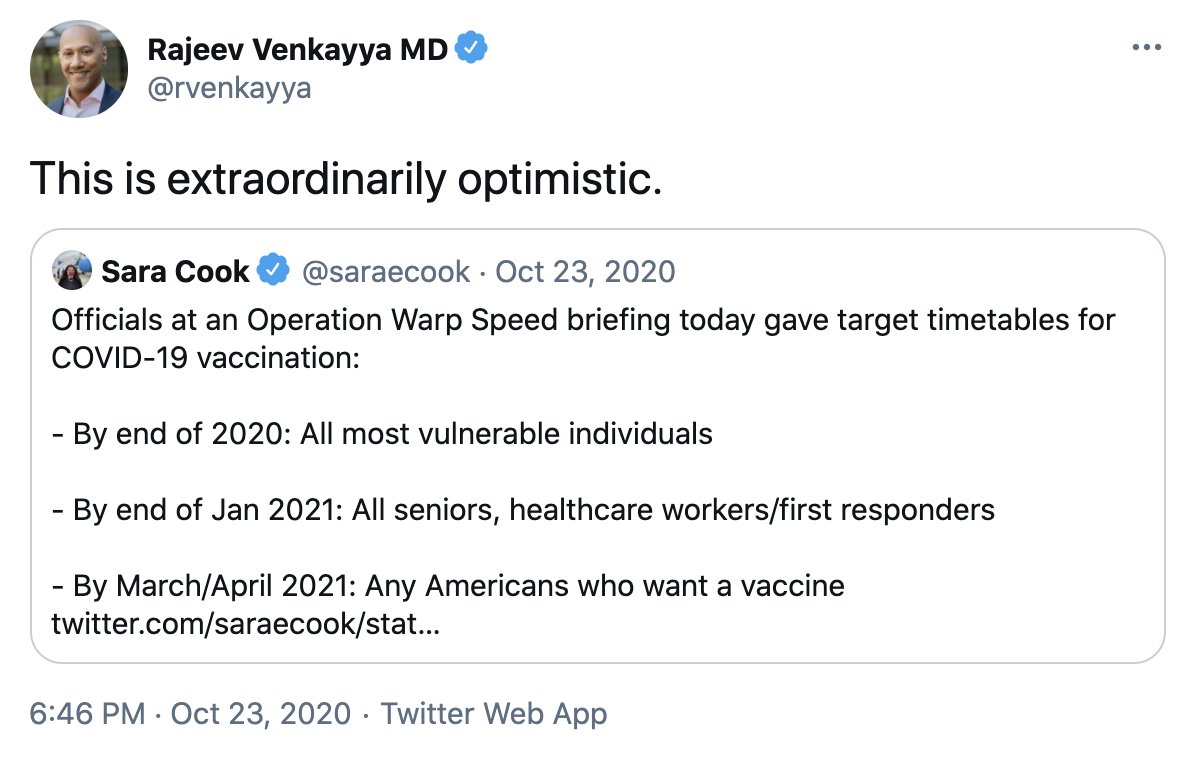
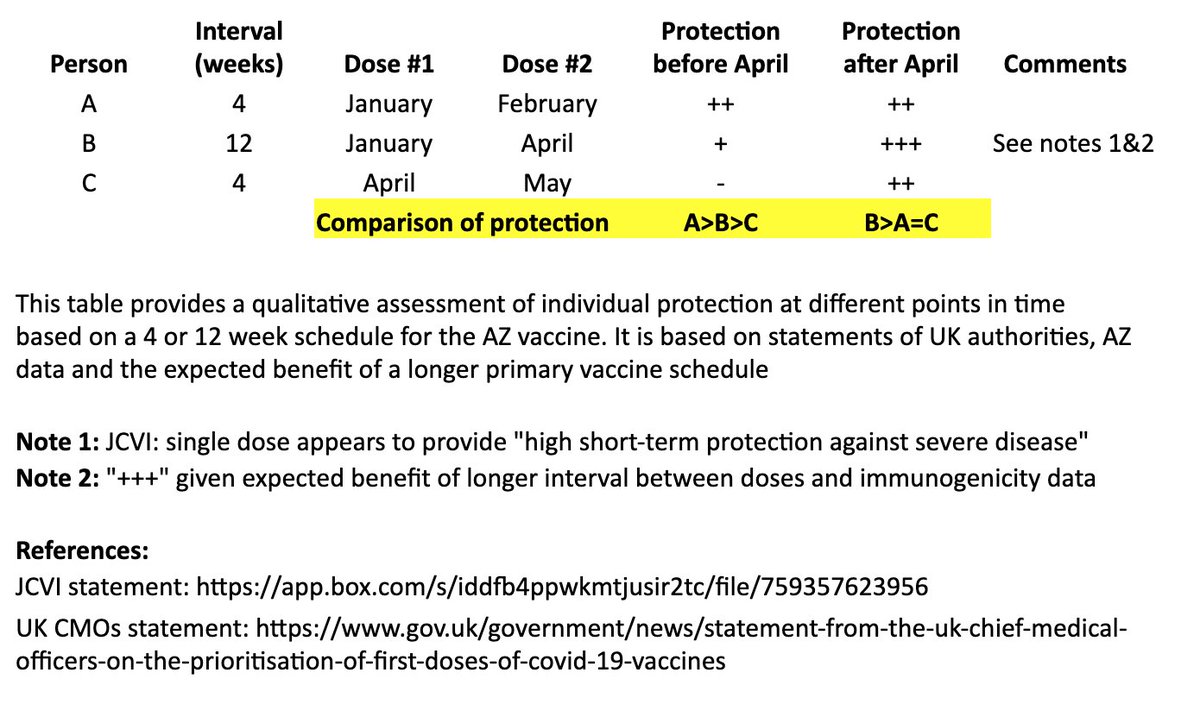
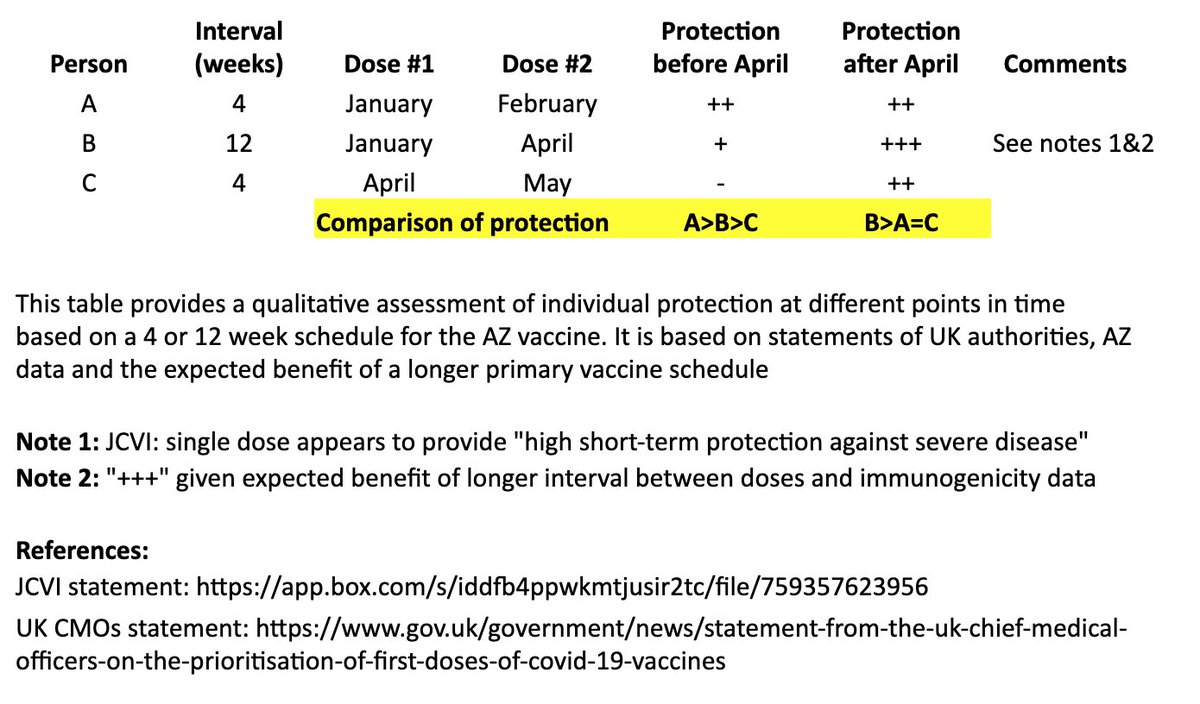
This is an excellent overview of the complexity of mRNA vaccine manufacturing.
An mRNA batch currently takes 110 days from start-finish.
“Traditional” vaccines (recombinant, vectored, inactivated) can take months longer. 1/
@eweise @kweintraub
usatoday.com/in-depth/news/…
An mRNA batch currently takes 110 days from start-finish.
“Traditional” vaccines (recombinant, vectored, inactivated) can take months longer. 1/
@eweise @kweintraub
usatoday.com/in-depth/news/…
More about the complexity of "traditional" vaccine approaches below.
These timelines assume the process has been "scaled up" and everything is in place & ready to go: facility, equipment, raw materials, etc. Unexpected issues can add weeks or months. 2/
These timelines assume the process has been "scaled up" and everything is in place & ready to go: facility, equipment, raw materials, etc. Unexpected issues can add weeks or months. 2/
https://twitter.com/rvenkayya/status/1356286201309900800?s=20
Here’s a good reference on #vaccine #manufacturing from several colleagues for those interested in learning more. 3/
ncbi.nlm.nih.gov/pmc/articles/P…
ncbi.nlm.nih.gov/pmc/articles/P…
Many thanks to @eweise @kweintraub @USATODAY for writing this story. Much-needed insight when the spotlight is on global supply. 4/
h/t @Merz @jonemo
usatoday.com/in-depth/news/…
h/t @Merz @jonemo
usatoday.com/in-depth/news/…
• • •
Missing some Tweet in this thread? You can try to
force a refresh