ICYMI: We recently announced results that expand our knowledge of NVX-CoV2373 and its ability to help protect from COVID19 over a longer period of time. Let's talk about what it means. Thread: 

Our new data build on findings from our #UK Phase 3 #clinicaltrial (randomized, placebo-controlled, double-blind, with crossover), where we examined vaccine performance in 15K adults. Publication: nejm.org/doi/full/10.10…
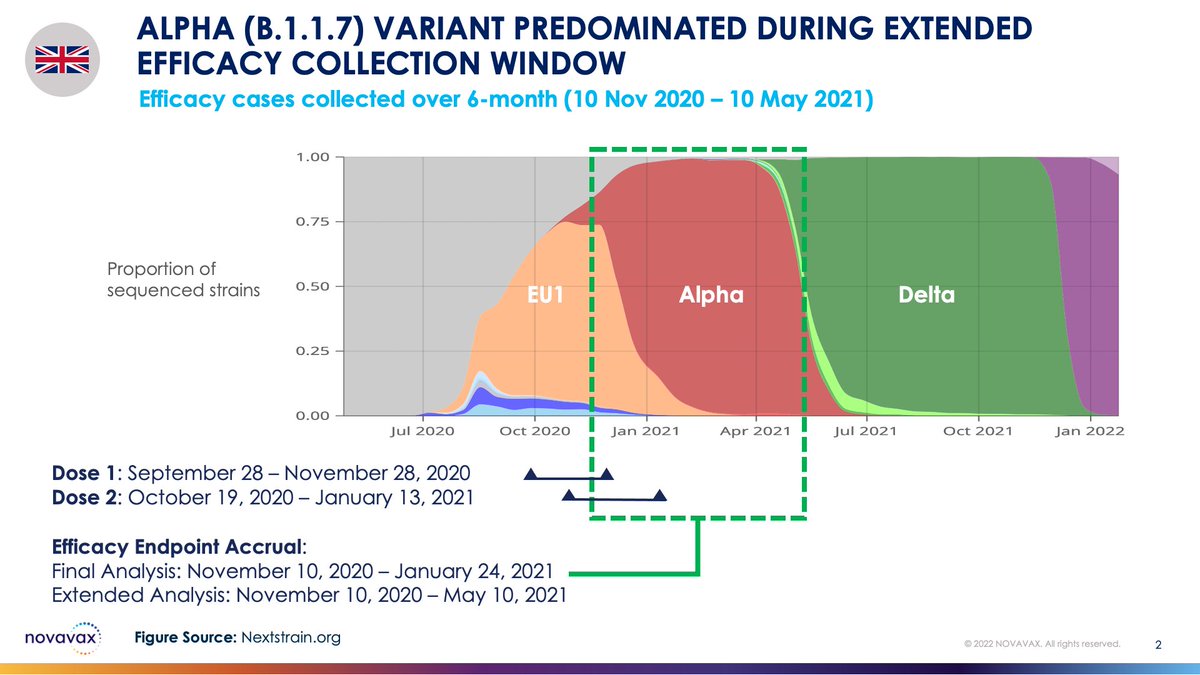
There, we found an overall vaccine efficacy of 89.7%. A median of 55 days of surveillance was used for the final analysis. The Alpha (B.1.1.7) strain predominated during this time, with 86.3% efficacy vs Alpha. 

Now, we're sharing data from an extended efficacy collection window from the UK Ph3 study: the 6-month period of 10 Nov 2020 - 10 May 2021, with a median of 101 days of surveillance. Alpha continued to dominate. 
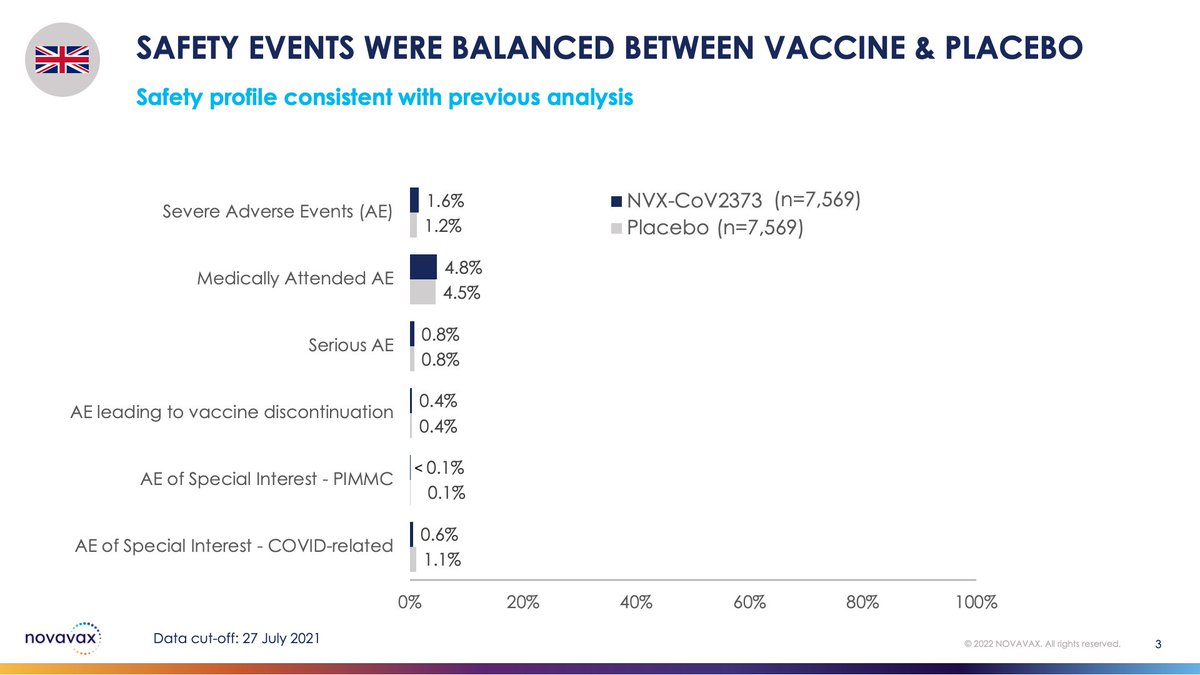
Our new analysis shows that the vaccine continues to demonstrate a reassuring top-line safety profile. Adverse events during this window were infrequent and balanced b/w vaccine & placebo groups. 
In the primary efficacy analysis of this expanded timeframe, 158 cases were observed: 24 in the vaccine group, 134 in placebo group, for a vaccine efficacy of 82.7%. 

A high level of vaccine efficacy was maintained over a 6-month period of surveillance, following the analysis used for EUA authorization (a median of 55 days of surveillance). 

Further, in the expanded timeframe, all severe cases were in the placebo group, resulting in 100% efficacy against severe disease.
In examining protection against all COVID infection - symptomatic or asymptomatic (measured by PCR+ or anti-N seroconversion), we saw a vaccine efficacy of 82.5%. 

Summary: Reassuring top-line safety profile. Clinical efficacy maintained over 6 months of surveillance. 100% efficacy against severe disease. 82.5% protection against all infection. 

These encouraging findings suggest that NVX-CoV2373 may have a positive impact on disease transmission and protection over a longer period of time.
Thank you. Novavax is grateful to the thousands of people around the world who are volunteering for our vaccine studies. We thank the U.S. Government, @CEPIvaccines & other govts for their significant support of our work.
• • •
Missing some Tweet in this thread? You can try to
force a refresh













