@MBVanElzakker and I are excited that our new paper on biological factors that may contribute to the development of LongCovid/PASC was published today: frontiersin.org/articles/10.33…
2/ The paper details mechanisms by which RNA #viruses beyond just #SARS-CoV-2 have be connected to long-term health consequences.
3/ It also reviews literature on acute #COVID-19 and other virus-initiated chronic syndromes such as post-#Ebola syndrome or #ME/CFS to discuss different scenarios for #LongCovid/#PASC symptom development.
4/ Potential contributors to PASC symptoms include consequences from acute SARS-CoV-2 injury to organs, persistent reservoirs of SARS-CoV-2 in certain tissues, re-activation of neurotrophic #pathogens such as #herpesviruses under conditions of COVID-19 immune dysregulation...
5/ ....SARS-CoV-2 interactions with host #microbiome/virome communities, clotting/#coagulation issues, dysfunctional #brainstem/vagus nerve signaling, ongoing activity of primed immune cells, and #autoimmunity due to molecular mimicry between pathogen and host proteins.
6/ We are very thankful for the images that other research teams gave us permission to use in the paper. Thanks @ahandvanish for allowing us to adapt this graph of top #LongCovid/#PASC symptoms from your excellent patient-led paper on the topic 

7/ Thanks to Dr. John Chia at the #Enterovirus Medical Research Center for this incredible original image of enteroviral capsid protein 1 in the stomach biopsy of an #ME/CFS patient. 

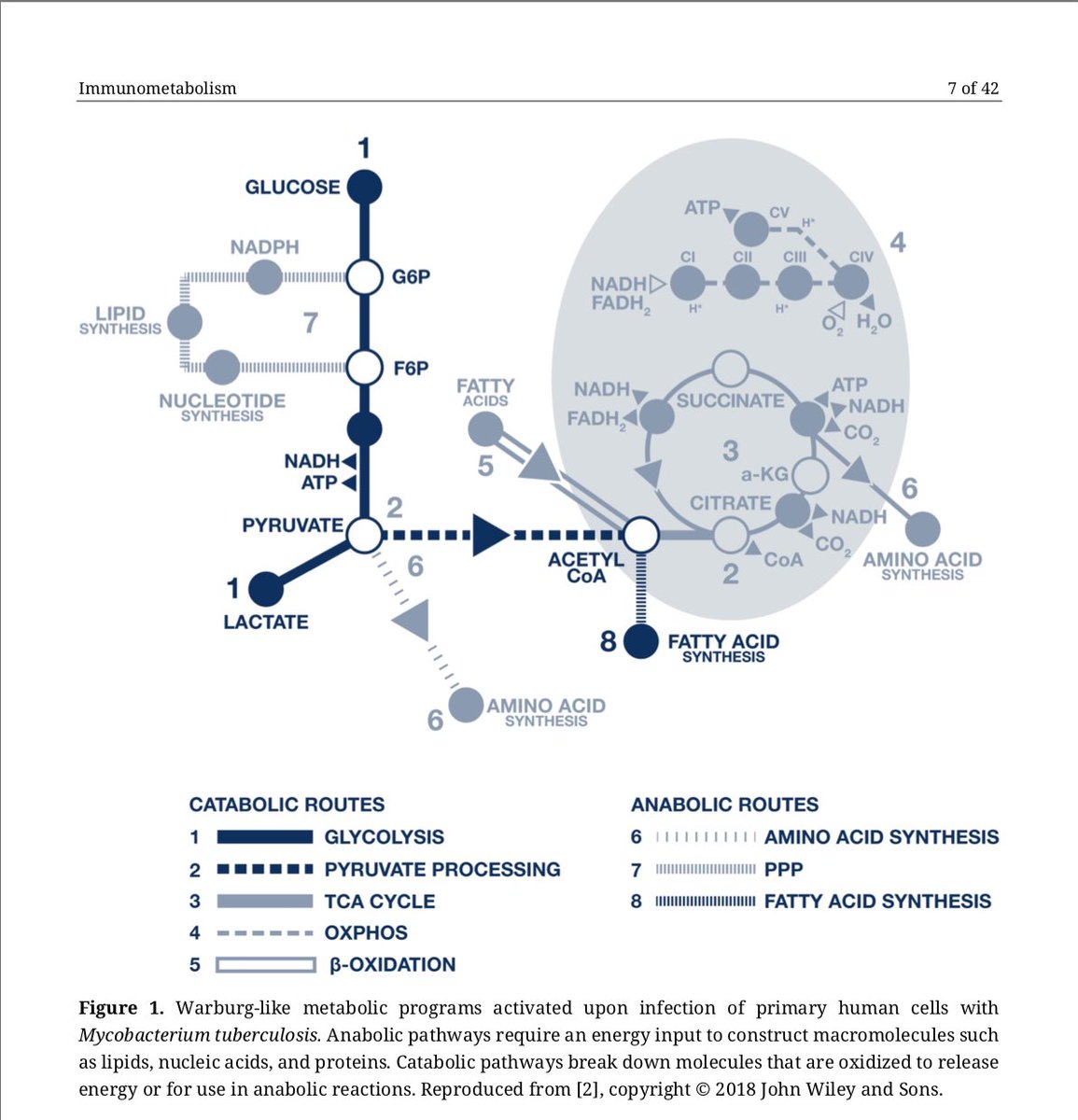
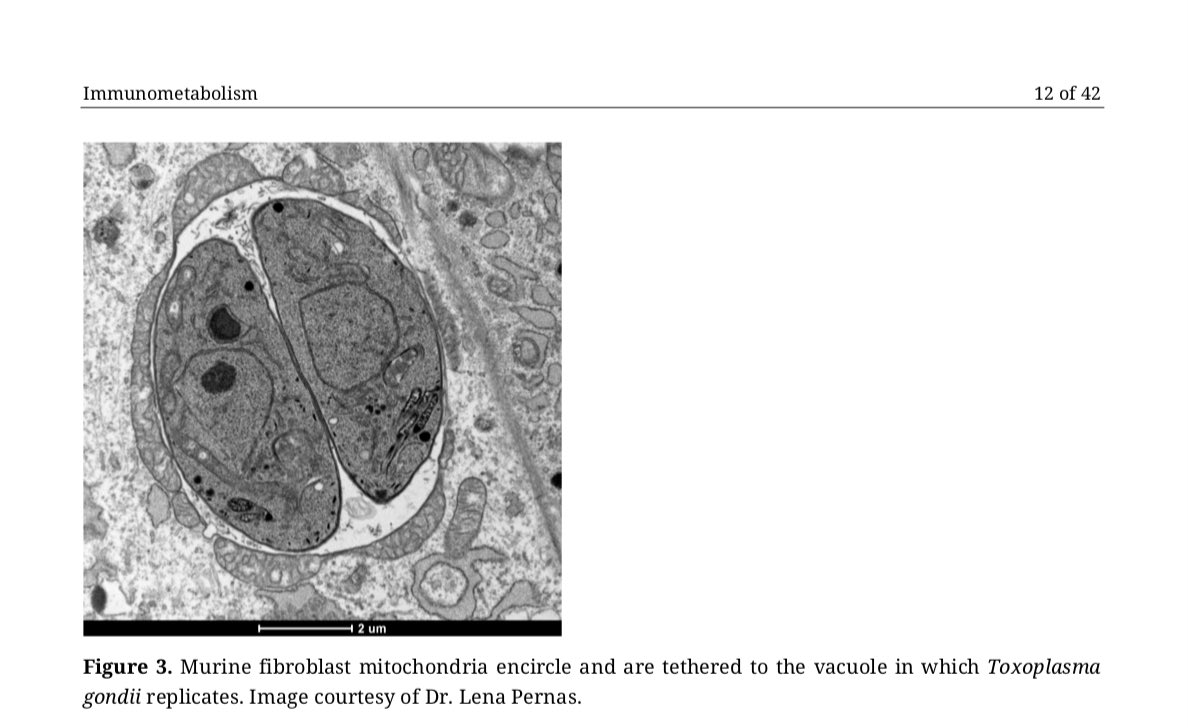
8/ Thanks to @lenapernas for this beautiful image of murine embryonic fibroblasts infected with #Toxoplasma gondii tachyzoites (the paper discusses how persistent pathogens such as Toxoplasma may reactivate during acute #COVID-19) 

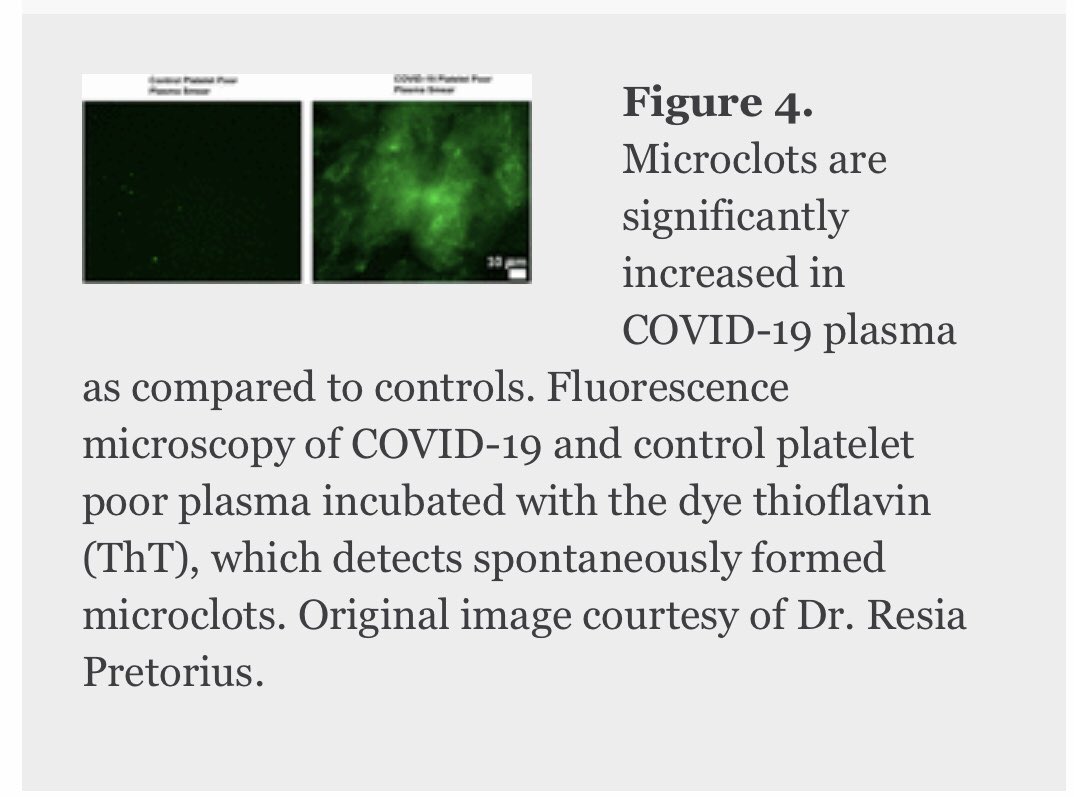
9/ Thanks to @resiapretorius for this image of microclots significantly increased in #COVID-19 plasma. She has also identified similar microclots in the plasma if patients with LongCovid/PASC: medrxiv.org/content/10.110… 
10/ Thanks to @RScloccoPhD, Dr. Zahra Mona Nasiriavanaki, and @PaulitaLara2 from the @MGHMartinos for help creating this original image of nuclei in the human brainstem 

• • •
Missing some Tweet in this thread? You can try to
force a refresh